-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Archaeology
p-ISSN: 2332-838X e-ISSN: 2332-841X
2021; 9(1): 68-72
doi:10.5923/j.archaeology.20210901.12

Can IBA Compete with Newer Analytical Methods in Archaeometry?
1Faculty of Mathematics and Physics, University of Ljubljana, Jadranska 19, SI, Ljubljana, Slovenia
2Jožef Stefan Institute, Jamova 39, SI, Ljubljana, Slovenia
Correspondence to: Ž. Šmit, Faculty of Mathematics and Physics, University of Ljubljana, Jadranska 19, SI, Ljubljana, Slovenia.
| Email: |  |
Copyright © 2021 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Ion beam analytical methods (IBA) are bound to the use of expensive electrostatic accelerators. During recent years, several analytical techniques were developed in the form of table-top instruments that can match or surpass the analytical capabilities of IBA. In the present study we compare IBA to several competing methods, notably XRF, LA ICP MS and neutron-based methods applied on different archaeological materials. In conclusion, IBA can still provide advantage, as several analytical techniques can be applied during single measurements, elemental profile information can be extracted and the irradiation is almost nondestructive.
Keywords: IBA, PIXE, PIGE, In-air beam, Metals, Glass
Cite this paper: Ž. Šmit, Can IBA Compete with Newer Analytical Methods in Archaeometry?, Archaeology, Vol. 9 No. 1, 2021, pp. 68-72. doi: 10.5923/j.archaeology.20210901.12.
Article Outline
1. Introduction
- Ion beam analytical methods are bound to the use of electrostatic accelerators and exploit radiation, induced by bombarding target with light ions, primarily protons, but also deuterons, alphas and lithium ions. The respective spectroscopies detect characteristic X-rays (PIXE), gamma-rays induced by inelastic nuclear scattering (PIGE), particles induced by nuclear reactions (NRA), Rutherford or elastically scattered ions (RBS), knocked-out light ions (elastic recoil spectroscopy – ERD), forward scattered projectiles and optical emission. Heavier ions are now used for the spectroscopies of secondary ions (MeV SIMS). These spectroscopies have been quite early employed for the analysis of the objects of cultural heritage, their main attraction being great sensitivity and almost non-destructive measurement. Pioneering work was done in Australia (J.R. Bird) [1], United States (C.P. Swan), Belgium (G. Demortier), Italy (P. Mando), Greece (A.A. Katsanos) and South Africa (M. Piesach). A decisive impetus was establishing a dedicated accelerator facility in the Louvre museum (1989) [2], which motivated such research in almost every lab having an electrostatic accelerator. In Ljubljana, for example, the first published work on analysis of archaeologic material appeared in 1984 [3], while intensive archaeometry research started since 1998. Archaeometry research further achieved support from European actions and IAEA [4].Parallel to IBA, alternative chemical analytical methods have been developed. Their main advantage are table-top dimensions of the instruments and much lower price, while their analytical abilities developed to be comparable or even better than those of IBA. It is then worth estimating how the analytical abilities of IBA are related to other competing methods and if they still have advantage. The criteria considered will be sample preparation, sample manipulation during measurement, destructive-nondestructive character of measurement, the range of detected elements, sensitivity and accuracy, surface-bulk analysis, and the range of eligible materials.
2. In-Air Setup
- IBA measurements on the objects of cultural heritage are typically performed by a proton beam extracted into air, which avoids putting the delicate objects into vacuum and allows simple manipulation of objects of unlimited dimensions (Fig. 1). The protons are extracted into air through a thin foil. Nowadays a sub-micrometer foil of silicon nitride (Si3N4) is popularly used, as it is characterized with good mechanical properties and it does not produce unwanted background in the spectra. Previously, foils of polyamide materials (sensitive to radiation damage), aluminum (produces strong background gamma lines) or tantalum (its L-lines appear in the X-ray spectra) have also been used. The number of impact particles is measured as a signal (RBS or X-ray) from the exit window or by a chopper intersecting the beam before the exit window. Different techniques are used to detect soft and hard X-rays. Soft X-rays are numerously produced, but they easily attenuate in the material. The detector for soft X-rays is then a thin-window type, in several labs it is equipped with a nozzle flushed with helium. Soft X-rays up to sodium are then easily detected, while the limit for a mere air path is around silicon. Hard X-rays are fairly less numerous due to low ionization cross section in heavy elements, so the respective detector is put close to the target to profit a larger solid angle, while the soft X-rays are suppressed by an absorber. An alternative is, however, a single detector using a pin-hole filter. The upper limit is around antimony for detecting K X-rays, while heavier elements are detected through their L X-rays. Technically, silicon drift detectors are now increasingly used for detection of X-rays, replacing the older Si(Li) or IGe detectors. Their advantage is, beside simpler cooling without liquid nitrogen, better energy resolution at low energies, but they have much lower counting efficiency at higher energies, as their crystals are only about 0.5 mm thick in comparison to 3 mm at a typical Si(Li). The target holder usually allows computer-controlled movement in XYZ directions, which allows mapping measurements.
 | Figure 1. In-air set up at the Tandetron accelerator of the Jožef Stefan Institute in Ljubljana |
2.1. Measurement of Archaeological Metals
- Archaeological metals are usually covered by a thick layer of patina that prevents analysis of the bulk material. The composition of the patina may show which elements are present in the alloy, but quantitative values of the metals are unreliable. For example, the patina of bronze is fairly enriched in tin, lead and iron, the latter element probably originating from the earth. For any measurement of IBA, it is then necessary to remove the patina on a small area until bulk material is reached. The size of the area may be several mm2, which is quite visible, and the object needs repair after the measurement by a restorer. On the other hand, for the analytical procedures based on the inductively-coupled plasma (ICP), such as atomic emission (AES) or mass spectrometry (MS) it is a standard procedure to sample material by drilling deep into the sample. Drilling leaves a rather small trace at the surface and the empty space is easily filled. However, the drilled material could be analyzed by IBA methods as well. Drilling may not be applied on the objects of thin metal sheets. (Semi) precious metals as silver and gold are usually without thick patina layers (or they are normally removed during the conservation procedure), but here the surface analysis meets other pitfalls: alloys of precious and base metals are inhomogeneous and exhibit distinct surface enrichment [5]. The analysis may then indicate a much finer alloy than it is its mean bulk composition. Surface effects are less pronounced at high-grade alloys (Fig. 2).
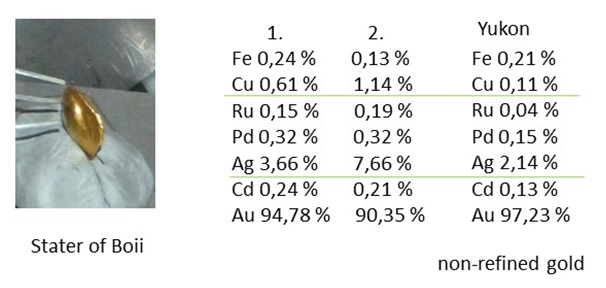 | Figure 2. PIXE analysis of two staters of the Celtic tribe Boii and of a gold nugget. The results indicate the use of non-refined alluvial gold for the coins |
2.2. Archaeological Glass
- Glass is attractive material for analysis as its composition is quite variable and reflects manufacturing techniques of the time. The range of elements extends from very light until the end of the periodic table. Glass is typically composed of siliceous matrix, whose melting point was reduced by alkalis, achieving final stabilization by adding earth-alkaline elements. X-ray based techniques can reach elements from Na until U, and NRA can be used for lighter elements (like B). For glass analysis it is normally assumed that all elements are in oxide form. However, alkaline elements are exposed to intensive leaching out of the surface, so it is recommendable to use a more deep-sensitive technique, such as PIGE for their analysis. The apparatus that apply measurements in vacuum or in He atmosphere, combine PIXE for the elements heavier than Mg and PIGE for Na [16]. Measurements in air render Mg and Al unobservable for PIXE, so PIGE needs to be applied instead, but for a price of a longer measuring time [17]. Measurements by IBA are almost non-destructive and can be performed on an unprepared artistic or archaeological object in selected spots. Irradiation may eventually leave dark spots, but which bleach out with time. Sampling is practiced on glass fragments; it usually exists of embedding a small portion of glass into resin and polishing. Such samples are efficiently analyzed in electron microscope exploiting electron-induced X-rays (energy-dispersive spectroscopy, EDS or electron microprobe analysis, EPMA), with the elemental range between sodium and approximately iron, X-rays of heavier elements being screened by the intense X-ray bremsstrahlung. Heavier elements are then analyzed in combination with XRF, based on X-ray tube or synchrotron radiation [18]. The two types of measurements provide comparable information which allows: (1) distinction between plant-ash or natron based alkalis, which is reflected in the concentrations of Mg and K oxides; (2) determination of the purity of silica according to the elements Al, Ti, Fe, Mn and Zr and rough characterization of silica source; (3) use of decolorants (MnO, Sb2O3, As2O3); identification of pigments and opacifiers. On the other hand, X-ray based methods cannot detect elements with concentrations below 1 µg/g. The only method that can reach below this limit is LA ICP MS. The concentrations of rare-earth elements are readily obtained, which are related to the geological past of the silica source. IBA methods can then provide basic analysis of glass objects, benefited by a non-destructive approach and simple object handling (Fig. 3), but it fails in detection of trace elements related to glass provenance.
2.3. (Semi) Precious Stones
- Stones popular in antiquity were emeralds in the imperial and garnets in Late Antique periods. Both are combination of different metal oxides with silicon oxides, which is characteristic for the region of origin. The range of interesting elements starts with boron, which can be analyses with NRA, yet the main light element is magnesium, which can be analyzed either by PIXE in vacuum or He atmosphere, or by PIGE in air. Of the trace elements, yttrium is important for garnets. It is a lucky circumstance that Y K-lines do not coincide with pile-uplines of other constituents. For the analysis of precious stones, it is tacitly assumed that the measuring procedure is completely non-destructive – a requirement that IBA fully meets. Another non-destructive method that provides complementary information is Raman spectroscopy. For garnets, both types of measurements were able to identify the countries of origin in India, Sri Lanka and Europe [19,20].
2.4. Pigment Analysis
- In historic paintings, the pigments were usually metal oxides or other compounds containing at least one metal ion. For characterization of the pigment it is then sufficient to identify this metal ion. Any X-ray technique, either XRF or PIXE, can this easily do. But the disadvantage of PIXE, especially for focused beams, is great deposition of energy, which results in observable radiation damage. For this reason, measurements with XRF are much less harmful. If PIXE is nevertheless applied care must be taken in using small currents and short measuring times, just to collect spectra with lowest acceptable statistics. The other advantage of PIXE is differential measurement with a set of different energies, which can distinguish individual paint layers. Thus, it is for example, possible to distinguish pigments in the painter’s signature [21]. The price is again high irradiation doses required at low impact energies, which may cause a harmful effect. Measurements with carefully controlled doses can be used for identification of pigments on paper or even for detection of characteristic materials in photographic techniques.
2.5. Other IBA Techniques
- Most of experimental facilities allow mapping measurements, which is achieved by precise mechanical movement of the sample holder. Resolution is of the size of the beam, which is about 30 µm in optimal cases, but practically rather around 100 µm. Mapping by PIXE competes with XRF mapping at advanced X-ray set-ups and synchrotrons. Further development of IBA methods now includes MeV SIMS, which is optimized for detection of molecular species of about 1000 atomic mass units, though lighter species can be detected as well [22].
3. Conclusions
- IBA methods can still compete with their less expensive competitors on account of their non-destructive approach, simple handling of the target, and possibility of simultaneous application of different technics that cover a wide range of elements. They are able to map areal distribution of detected elements and probe the concentration profiles.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML