-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Advances in Life Sciences
p-ISSN: 2163-1387 e-ISSN: 2163-1395
2013; 3(1): 5-17
doi:10.5923/j.als.20130301.02
Algal Structural Complexity Effect on Diversity and Abundance of Some Ostracod Species from Red Sea Coast, Egypt
Ebtesam A. Yousef
Zoology Department, Faculty of Science, Sohag University, P.O. Box 82524 , Sohag, Egypt
Correspondence to: Ebtesam A. Yousef, Zoology Department, Faculty of Science, Sohag University, P.O. Box 82524 , Sohag, Egypt.
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
Four species of macroalgae (Sargassum obtusifolium, Sargassum polyphyllum, Chnoospora minima, Nemacystus decipiens) were collected monthly to study the changes in ostracod species abundance, richness and diversity between them during 12 months. Also, determine effect of algal structural complexity on ostracod abundance and diversity. Structural complexity was estimated by counting the number of branches cm-1 for each one of the four studied algae. There were significant differences in complexity between the 4 algal species. Sargassum obtusifolium had significantly more branches cm-1 than the remaining 3 algal species, and Sargassum polyphyllum had significantly more branches than Chnoospora minima, and Nemacystus decipiens. There were differences in abundance and diversity of ostracod assemblages between the fouralgal species. Where, Sargassum obtusifolium carried a higher abundance, species richness and diversity of ostracods during study period than that of the residual three algae. The ostracod samples of four studied algae nearly formed separate clusters, reflecting the difference between these algae throughout the year. Sixteen species of ostracod occurred on the algae, six only occurred at sufficient densities for determination of their life-cycles. Cylindroleberis sp., Ghardaglaia triebeli and Mosella striata occurred at high densities and had two generations per year. The other three abundant ostracod species; Loxoconcha ornatovalve, Paradoxostoma altecaudatum and Xestoleberis ghardaqe found at low densities and had one generation per year.
Keywords: Marine Ostracods, Structure Complexity, Population Dynamics, Reproduction and Ontogeny, Red Sea
Cite this paper: Ebtesam A. Yousef, Algal Structural Complexity Effect on Diversity and Abundance of Some Ostracod Species from Red Sea Coast, Egypt, Advances in Life Sciences, Vol. 3 No. 1, 2013, pp. 5-17. doi: 10.5923/j.als.20130301.02.
Article Outline
1. Introduction
- Ostracods are small bivalved Crustacea, their carapace envelope the whole body and limbs. They live in all aquatic environments and grow by molting their shells eight times to reach the adult stage during lifecycle. Ostracoda have a wide range of diets as carnivores, scavengers, filter feeders and herbivores, while they nearly form more than 98% of the phytal meifauna (Hull, 1997).Many of ecological studies have documented that habitats have an effect on species richness and diversity[1, 2]. On the other hand, habitat complexity is associated with texture, size and shape of algal species[1], so that the higher densities of animals correlate with more algal structural complexity[3].Other studies reported that an increasing of habitat complexity may be increase the diversity and abundance of organisms[4], as a result of increased protection from predation, wave action and dissection[5-7]. Above all, marine algae provide a suitable habitat for many of animal species especially invertebrates which use macroalgae as a shelter from physical stress, predators[8] and depend on them as a source of food[9, 10].The previous studies on phytal ostracods have been concentrated on the description of the assemblages in space and have not considered the seasonal dynamics of the assemblages over time or between different algal species [11-13], for the reason of collection and identification problems of small species. However, some authors studied the dynamics of ostracod assemblages[14-17].The current study aims to determine the source of the significance between mean branches of four species of Red Sea algae, to estimate monthly differences in ostracod densities on studied algae and examine the lifecycle of some ostracod species so as to determine if diversity and abundance of ostracod vary with the structure complexity of algae.
2. Material and Methods
- Samples of four species of macroalgae (Sargassum obtusifolium, Sargassum polyphyllum, Chnoospora minima, Nemacystus decipiens) were randomly collected each month from an exposed sand rocky shore rich with different types of macroalgae situated at 35km South of Safaga city of Egypt (Fig. 1), during the period from June 2010 and May 2011. The four studied macroalgal species were present in the studied area all over the year and they have difference in their frond structures. A sub-sample of the four species of algae was collected to measure their frond length and number of branches and to estimate the degree of branching per unit length for each one of them.
 | Figure 1. Location map of the study area |
3. Statistical Analysis for Data
- Statistical analyses were performed by using 1-way and 2-factor ANOVA tests. They were of fixed, balanced design; therefore Tukey’s tests were performed to determine the source of the significance between means[23]. One way ANOVA was used to determine significant differences in ostracod densities either between algal species or over the sampling period. Two-factor ANOVAs were used to examine the significance of the effect of algal species and month over the sampling period. Six ostracod species occurred in sufficiently high densities throughout the year to enable their lifecycles to be determined and changes in density to be statistically tested using ANOVA.Shannon Wiener diversity (H'), species richness (S) and Evenness (E) were calculated on ostracod samples for each species of algae within each month. ANOVA was used to test for significant differences in diversity between the algal species and over the sampling period within an algal species. A dendrogram was produced using SPSS version 16.0.
4. Results
4.1. Description of Algal Species
- By comparing the number of branches cm-1 for the four species of algae (Table 1) there was significant difference in number of branches between the four studied species of algae (ANOVA F= 582.33, P < 0.000; Tukey p = 0.05), where the highest degree of branches was found in Sargassum obtusifolium, which had higher number of branches than the three other species. Sargassum polyphyllum had higher number of branches than Chnoospora minima and Nemacystus decipiens and there was significant difference in number of branches between C. minima and N. decipiens.
4.2. Density of Ostracod Species
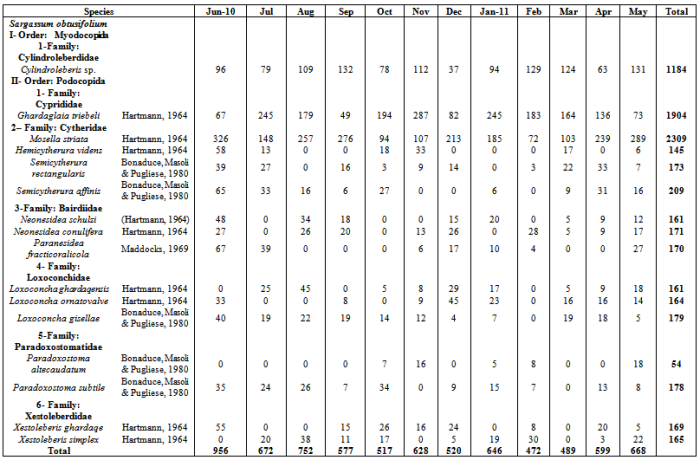
- Sixteen ostracod species from two orders and six families were identified from the samples of algae which collected during the study period (Appendix 1). Fig.2 shows the mean ostracod density on the 4 algal species during study period. There were significant differences in ostracod density both between the four species of algae each month (2-way ANOVA, p < 0.013 in all cases). In general, S. obtusifolium (mean ostracod density= (468.5 ± 134.086 SD) carried higher ostracod densities than C. minima (mean =174.312 ± 83.745), N. decipiens (mean =143.812 ± 62.778) and S. polyphyllum (mean = 250.75 ± 125.0166), whereas S. polyphyllum carried higher densities than C. minima and N. decipiens (ANOVA F= 41.11, p < 0.000; Tukey p = 0.05).
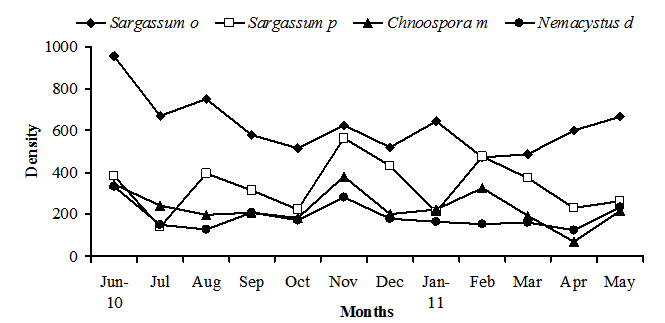 | Figure 2. Changes in ostracod density on the four studied algal species during 12 months |
4.3. Life Cycle and Population Density of Ostracod
- Sixteen ostracod species recovered from the studied algal species, six species of them occurred in abundant numbers to study changes in their life cycles and population density (Cylindroleberis sp., Ghardaglaia triebeli, Mosella striata, Loxoconcha ornatovalve, Paradoxostoma altecaudatum and Xestoleberis ghardaqe). The remaining species(Hemicytherura videns, Semicytherura rectangularis, Semicytherura affinis, Neonesidea schulzi, Neonesidea conulifera, Paranesidea fracticoralicola, Loxoconcha ghardaqensis, Loxoconcha gisella, Paradoxostoma subtile and Xestoleberis simplex) found in small number of densities so that they are excluded from this analysis. Six ostracod species showed significant changes in density between the algal species and month (2-way ANOVA p < 0.000 in all cases). In general, S. obtusifolium carried the highest population densities of Cylindroleberis sp., Ghardaglaia triebeli, Mosella striata, Semicytherura affinis Neonesidea conulifera, Paranesidea fracticoralicola, Loxoconcha ghardaqensis and Xestoleberis simplex comparing to the remaining three algae. Hemicytherura videns, Semicytherura rectangularis and Loxoconcha ornatovalve occurred in significantly higher densities on Sargassum polyphyllum, while Chnoospora minima carried the highest population densities of Paradoxostoma altecaudatum andParadoxostoma subtile.Fig. 3a demonstrates the life-cycle of Cylindroleberis sp. where reproduction occurred in last spring and last winter due to the appearance of A-5 and A-6 instar stages in July 2010 and January 2011. These instars matured during summer and spring months and the population appeared low of adult densities. There was significant difference in the population densities of Cylindroleberis sp. where higher densities occurred in September, May and February than that in all study year months (ANOVA F= 3.00, p < 0.011; Tukey p = 0.05). Fig. 3b illustrates the life-cycle of Ghardaglaia triebeli where adults appear in the population in summer and spring as soon as reproduction took place resulting appearance of A-4 and A-5 instar stages in September- October 2010 which moulting to adults in the next spring. Ghardaglaia triebeli had a significant difference in population density all over the year (ANOVA F= 4.85, p < 0.000; Tukey p = 0.05) where higher densities occurred in November, July and January, respectively. In the life-cycle of Mosella striata (Fig. 3c) adults occurred in the population from June to September and from November to January according to that A-6 and A-5 appeared in the population from the middle of summer to the end of winter. There was no significant difference in population density of Mosella striata (ANOVA F= 1.18, p < 0.326; Tukey p = 0.05) where the highest density found in June 2010 and the lowest one occurred in February 2011. Fig. 3d showing the life-cycle of Loxoconcha ornatovalve which include appearance of A and A-1 in summer where reproduction took place due to appearance of A-4, A-5 and A-6 in August- October. These juvenile moulting to adult forms in the following summer. L. ornatovalve had significant difference in population density (ANOVA F= 2.82, p < 0.015; Tukey p = 0.05) with higher density in September 2010 and May 2011 than the remaining months of year. A-1 stage of Paradoxostoma altecaudatum (Fig. 3e) moulted to adults in summer and A-4 and A-5 appeared in population from September to January which moulted to higher stages until became adults in the end of spring. There was no significant difference in P. altecaudatum population density (ANOVA F= 0.74, p < 0.617; Tukey p = 0.05) where higher population density occurred in November than that of July 2010 and February 2011. In life-cycle of Xestoleberis ghardaqe (Fig. 3f) A-6 and A-5 appeared in summer and then moulted to higher stages in the following months till became adults in spring. X. ghardaqe had no significant population density (ANOVA F= 1.63, p < 0.149; Tukey p = 0.05) where higher densities occurred in October 2010 and May 2011than those found in the residual months throughout year.
4.4. The Variation in Ostracod Diversity, Richness and Evenness
- Fig. 4 demonstrates the diversity (Fig. 4a), richness (Fig. 4b) and evenness (Fig. 4c) of the ostracod assemblages on the four species of algae throughout the year. There was a significant difference in Shannon Wiener H' both between algal species and months within each algal species (2-way ANOVA p < 0.038 in all cases). As soon as Nemacystus decipiens had lower diversity than Sargassum obtusifolium, Sargassum polyphyllum and Chnoospora minima (Fig. 4a: ANOVA F = 6.31, p = 0.001; Tukey p = 0.05), however there was no significant difference in ostracod diversity between the three other algal species (Fig. 4a: ANOVA F = 2.41, p = 0.134; Tukey p = 0.05). Sargassum obtusifolium showed no monthly significant differences in diversity (ANOVA F= 0.16, p = 0.999), where the lowest and highest values of diversity found in February 2011 and June 2010, respectively. As well as the residual three species of algae demonstrated no significant differences in ostracod diversity monthly. For Sargassum polyphyllum, the highest diversity value occurred in April 2011, while, November 2010 sample had a lower diversity than July and September 2010, and the March 2011 sample had a higher diversity than October 2010, January and February 2011 (ANOVA F = 0.19, p = 0.998; Tukey p = 0.05). On Chnoospora minima the April and June 2010 samples had the lowest and highest diversity values throughout the year samples (ANOVA F=0.17, p = 0.999; Tukey p =0.05). For Nemacystus decipiens, the highest and lowest of diversity values occurred in June and August 2010, respectively, In addition February and March 2011 samples had a higher diversity than September-November 2010 and April-May 2011 samples (ANOVA F=0.68, p = 0.753; Tukey p =0.05). Species richness on the four studied species of algae are distinguishing as follow (Fig. 3b), the median number of species on Sargapssum obtusifolium was 12 higher than that on Sargassum polyphyllum (median number= 11), on Chnoospora minima (median number= 9.5) and on Nemacystus decipiens (median number= 9).
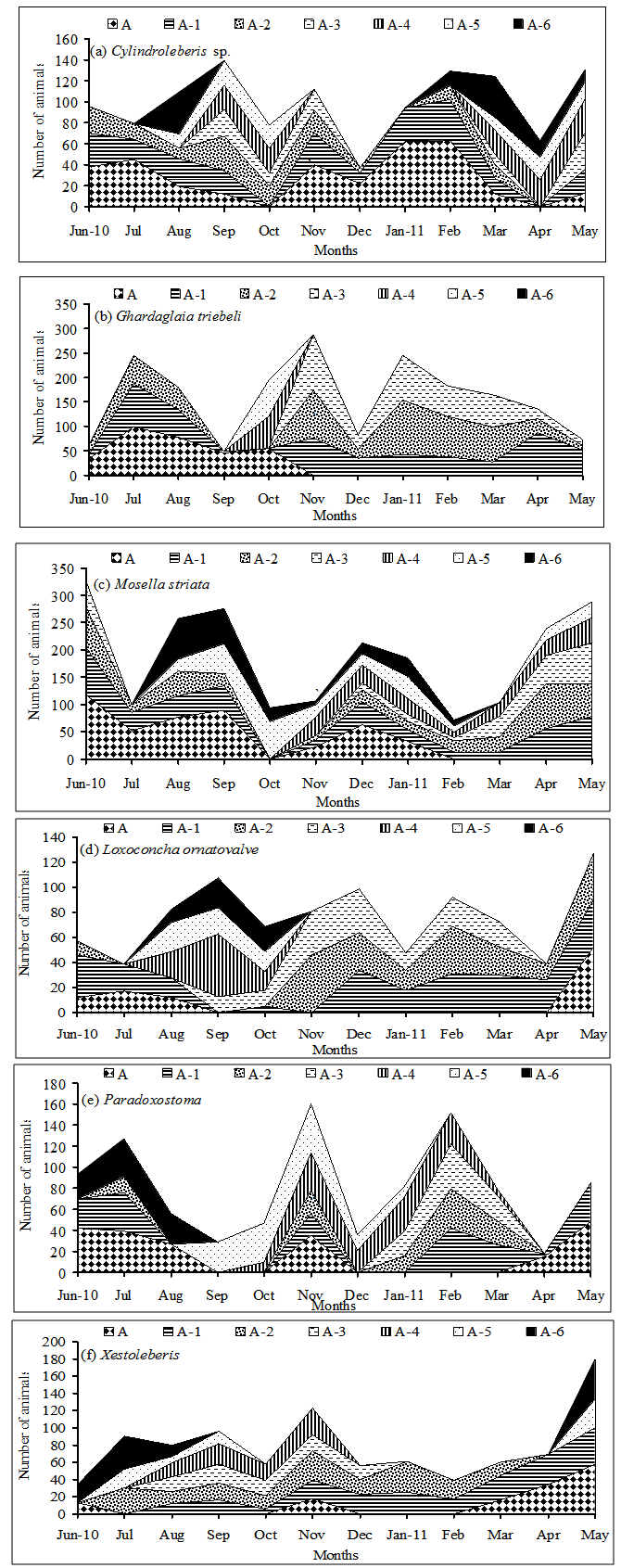 | Figure 3. Life-cycle of the six abundant ostracod species revealing changes in population density and appearance of different instars within each population. The instars are mareked as follow: A= adult, A-1= 8th instar, A-2= 7th instar, A-3= 6th instar, A-4= 5th instar, A-5= 4th instar, A-6 = 6th instar |
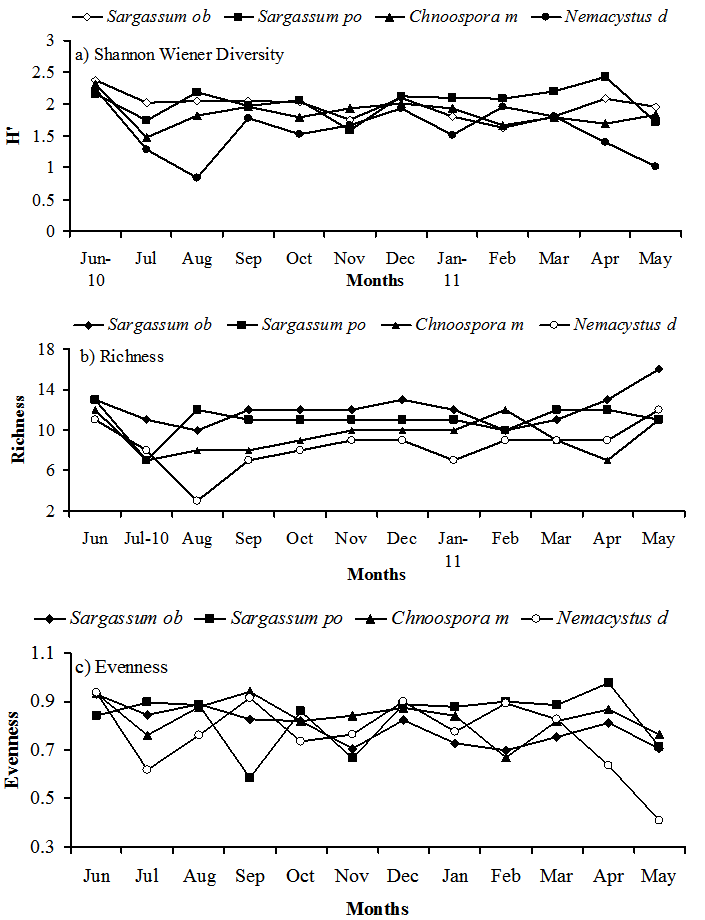 | Figure 4. Monthly changes in ostracod Shannon Wiener diversity, species Richness and Evenness on four studied species of algae during 12 months |
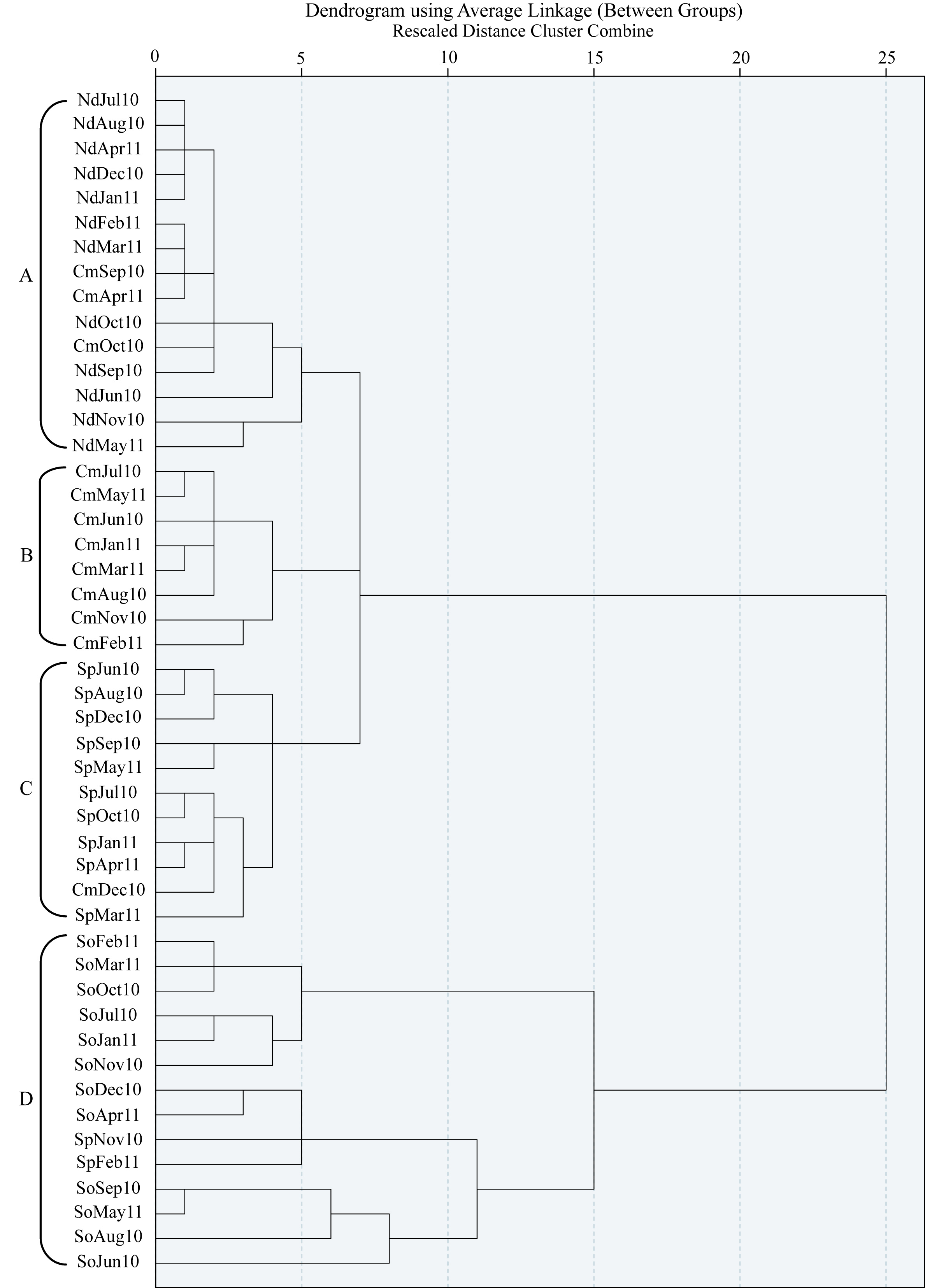 | Figure 5. Dendrogram cluster analysis for monthly samples of four species of algea during 12 months (Key: So=Sargassum obtusifolium, Sp=Sargassum polyphyllum, Cm=Chnoospora minima, Nd-Nemacystus decipiens) |
5. Discussion
- The density of phytal ostracods changes according to the degree of algal structural complexity. The results of the current study demonstrate that S. obtusifolium had the most complex frond structure and carried higher densities of ostracods than the residual 3 algal species. Similar data recorded by[17] on 18 ostracod species associated with four species of algae. Furthermore, high complexity habitats support a larger diversity, richness of species and a greater abundance of individuals than medium or low diversity habitats. Similar suggestion recorded by[3] and[11]. During the current study, there was no significant difference in ostracod density on the four studied species of algae. Also,[24] reported similar data, however[17] noted that there are significant difference in ostracod density on Ceramium, Chondrus and Corallina and there is no significant difference in ostracod density on Cladophora.It is likely to note that, life cycles of ostracod species in the Red Sea waters have been poorly investigated so the current study revealed that, Cylindroleberis sp., G. triebeli and M. striata have two generations per year. While L. ornatovalve, P. altecaudatum and X. ghardaqe have one generation per year.[25, 26] described one generation annually for Hirschmannia viridis and Cyprideis torosa, respectively. However,[27] found one to three generations per year of the seven species of ostracods he investigated.[16] found 4 to 5 generations per year of some ostracod species in brackish water.In Cylindroleberis sp. reproduction occurred in last spring and last winter, while Ghardaglaia triebeli matured in summer and spring and in Mosella sp. reproduction occurred during summer and last winter.The higher densities of Cylindroleberis sp. were found in autumn and summer, while in Ghardaglaia triebeli occurred in winter and summer. Additionally, higher density in Mosella sp. occurred in summer. [11, 17] reported that higher density of C. lutea occurred in winter.[11] noted that the low densities of H. viridis observed during the winter months due to seasonal migration to sublittoral areas.In our study, the other three studied species: Loxoconcha ornatovalve, which demonstrated positive difference in population density and its reproduction, took place in August-October due to appearance of A-4, A-5 and A-6. Paradoxostoma altecaudatum and Xestoleberis ghardaqe they demonstrated no significant difference in their population density. P. altecaudatum moulted to adults in summer and A-4 and A-5 appeared in population in autumn, while A-6 and A-5 of X. ghardaqe appeared in summer.[17] noted that X. aurantia demonstrated positive covariation in population density with high densities duringAugust-September and reported that the genus Paradoxostoma occurred at low densities during spring and early summer. A total of sixteen ostracod species were recovered from the studied algal species, six species of them occurred in abundant numbers to study changes in their life cycles compared to ten species at low densities.[17] studied 18 species of ostracod were found on the algae, but only 8 occurred at densities sufficient for determination of their life-cycles compared with ten species at low densities,[11] found 7 species at low densities on Chondrus, compared with 14 species in high densities during spring and summer on Cladophora
ACKNOWLEDGEMENTS
- The author is deeply grateful to the editor and the anonymous reviewers for their helpful comments on the manuscript.
Appendix : Monthly Ostracod Numbers on Four Studied Species of Algae during One Year
 Continued:
Continued: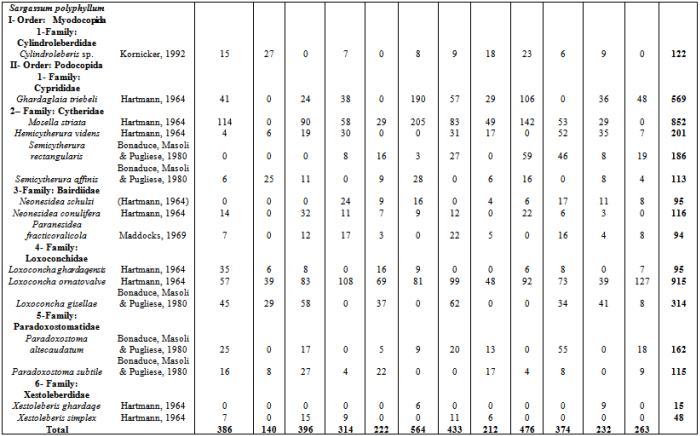 Continued:
Continued: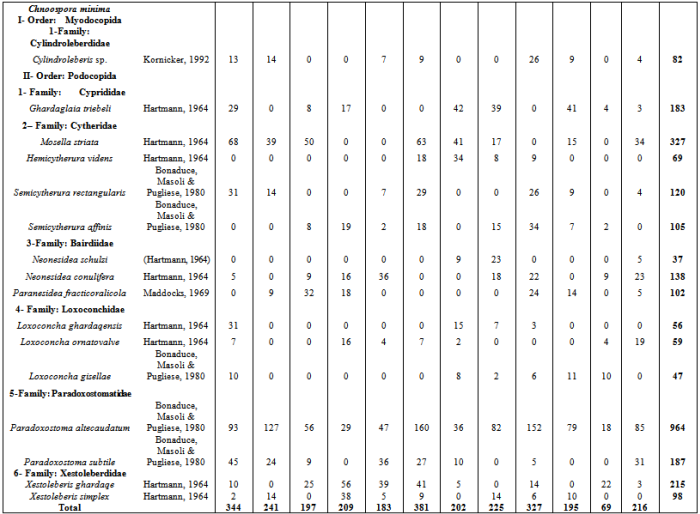 Continued:
Continued: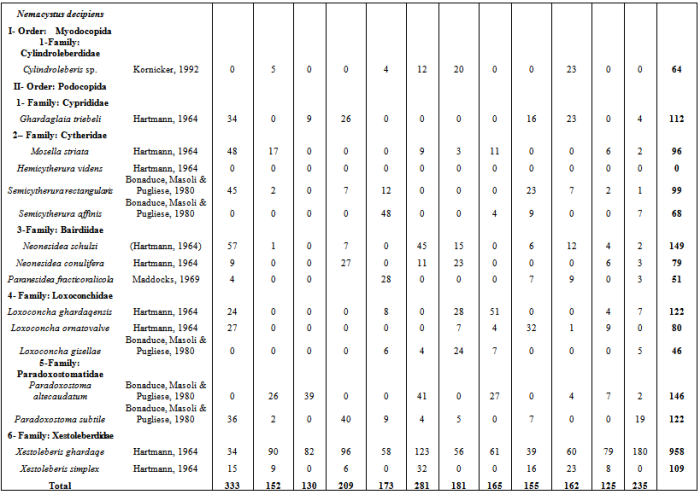
References
| [1] | Gee, J. M., Warwick, R. M. (1994): Metazoan community structure in relation to the fractal dimensions of marine macroalgae. Mar Ecol Prog Ser 103:141-150 |
| [2] | Jarvis, S. C., Seed, R., (1996): The meiofauna of Ascophyllum nodosum (L.) Le Jolis: characterization of the assemblages associated with two common epiphytes. Journal of Exp Mar Bio and Ecol 199: 249–267 |
| [3] | Hicks, G. R., (1985): Meiofauna associated with rocky shore algae. In: Moore PG, Seed R (eds) Ecology of rocky coasts. Hodder & Stoughton, London |
| [4] | Hixon, M. A., Menge, B. A., (1991): Species diversity: prey refuges modify the interaction effects of predation and competition. Theor. Popul. Bio 39:178–200 |
| [5] | Coull, B.C., Wells, J. B., (1983). Refuges from fish predation: experiments with phytal meiofaunal from the New Zealand rocky intertidal. Ecol 64:1599–1609 |
| [6] | Gibbons, M. J., (1988a): The impact of sediment accumulation, relative habitat complexlty and elevation on rocky shore meiofauna. J. Exp. Bar. Biol. Ecol. 122: 225-241 |
| [7] | Gibbons, M. J., (1988b): Impact of predation by juvenile Chnus superciliosus on phytal meiofauna are fish important as predators? Mar. Ecol. Prog. Ser. 45: 13-22 |
| [8] | Viejo, R. M., (1999): Mobile epifauna inhabiting the invasive Sargassum muticum and two local seaweeds in northern Spain. Aquat Bio 64:131–149 |
| [9] | Duffy J. E., (1990): Amphipods on seaweeds: partners or pests? Oecologia, 83:267–276 |
| [10] | Bell, S. S., (1991): Amphipods are Insects equivalents? an alternative view. Ecol 72:350–354 |
| [11] | Whatley, R. C., Wall, D. R., (1975): The relationship between Ostracoda and algae in littoral and sublittoral marine environments. In: Swain FM (ed) Biology and palaeobiology of Ostracoda. Bull. Am. Palaeontol. Soc 65:173-203 |
| [12] | Trier, K., (1993): A preliminary study of the brackish and marine Ostracoda of the Pembrokeshire Coast, S.W. Wales. In: McKenzie KG, Jones PJ (eds) Ostracoda in the earth and life sciences. A.A Balkema, Rotterdam, p 571-581 |
| [13] | Neale, J. W., (1970): The marine fauna and flora of the Isles of Scilly. Crustacea: Ostracoda. J. Nat. Hist.4: 399- 411 |
| [14] | Hagerman, L., (1966): The macro and microfauna associated with Fucus serratus L. with some ecological remarks. Ophelia 3:1–43 |
| [15] | Hagerman, L., (1968): The ostracod fauna of Corallina officinalis L. InWestern Norway. Sarsia 36:49-54 |
| [16] | Hagerman, L., (1978): The life-cycle of three species of algal living ostracods from brackish water. Ophelia 17:231-237 |
| [17] | Hull, S. L., (1997): Seasonal changes in diversity and abundance of ostracods on four species of intertidal algae with differing structural complexity. Mar. Ecol. Prog. Ser. 161:71–82 |
| [18] | Hartmann, G., (1964): Zur Kenntnis der Ostracoden des Roten Meeres. Kieler Meeres for schungen, 20: 35-127 |
| [19] | Maddocks, R. F. (1969): Revision of Recent Bairdiidae (Ostracoda): Smithsonian Institution U. S. National Museum Bull. No .295, pp.1 –126 |
| [20] | Bonaduce, G., Masoli, M., Pugliese, N., (1976): Benthic ostracoda from the Gulf of Aqaba. Puppl Staz Zool. Napoli, vol. 41, 2 |
| [21] | Bonaduce, G., Masoli, M., Pugliese, N., (1980): Some new benthic ostracod species from the Gulf of Aqaba (Red Sea). Bollettino della Societa Paleontologica Italiano 19: 143-178 |
| [22] | Kornicker, L. S. (1992): Myodocopid Ostracoda of the Benthedi Expedition, 1977, to the NE Mozambique channel, Indian Ocean. Smithsonian contribution to zoology, 531, 1-243 |
| [23] | Fowler J., Cohen, L., (1990): Practical statistics for field biology. John Wiley & Sons, Chichester |
| [24] | Johnson, S. C., Scheibhng, R. E., (1987): Structure and dynamics of epifaunal assemblages on intertidal macroalgae Ascophyllum nodosum and Fucus vesiculosus in Nova Scotla, Canada Mar Ecol. Prog Ser. 37:209-227 |
| [25] | Hagerman, L., (1969): Environmental factors affecting Hirshmannia viridis (O.F. Muller) (Ostracoda) in shallow brackish water. Ophelia. 7: 79 99 |
| [26] | Heib, C., (1976): The life-Cycle of Cyprideis torosa (Crustacea, Ostracoda). Oecologia (Berl.) 24. 229- 245 |
| [27] | Theisen, B. F., (1966): The life history of seven species of ostracods from a Danish brackish water locality. Medd. Danm. Fisk. Og Havunders. 4,215- 270 |
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML