-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
Advances in Life Sciences
p-ISSN: 2163-1387 e-ISSN: 2163-1395
2012; 2(2): 17-20
doi: 10.5923/j.als.20120202.04
Biological Properties of Thuja Orientalis Linn
Priya Srivastava 1, P. Kumar 2, D. K. Singh 2, V. K. Singh 2
1Department of Zoology, St. Xavier’s College, Ranchi, Jharkhand, 834001, India
2Malacology Laboratory, Department of Zoology, D.D.U. Gorakhpur University, Gorakhpur , 273009, India
Correspondence to: V. K. Singh , Malacology Laboratory, Department of Zoology, D.D.U. Gorakhpur University, Gorakhpur , 273009, India.
| Email: |  |
Copyright © 2012 Scientific & Academic Publishing. All Rights Reserved.
Thuja orientalis (Commonly- Morpankhi, Family- Cupressaceae) is an evergreen, monoecious trees or shrubs used in various forms of traditional medicines and homeopathy in various ways. In traditional practices Thuja is used for treatment of bronchial catarrh, enuresis, cystitis, psoriasis, uterine carcinomas, amenorrhea and rheumatism. Recent researches in different parts of the world have shown that T. orientalis and its active component thujone have the great potential against a various health problems. T. orientalis preparations can be efficiently used against microbial/worm infection. It can be used as antioxidant, anticancer and anti-inflammatory agent. Instead of these effects, it can be also used as insecticidal, molluscicidal and nematicidal activity against different pests. The present review highlights the some important biological properties of T. orientalis.
Keywords: Thuja Orientalis, Morpankhi, Thujone, Antimicrobial, Molluscicidal, Antimicrobial
Article Outline
1. Introduction
- Thuja orientalis (Commonly- Morpankhi, Family- Cupressaceae) is a genus of coniferous trees. T. orientalis is an evergreen, monoecious trees or shrubs growing to 10-60 feet tall. The shoot are flat, leaves are scale like. The leaves are arranged in flattened fan shaped growing with resine glands [1]. Their leaves contain essential oils used to treat fungus infections, cancer, moles and parasitic worms. The essential oil derived from the leaves is toxic. α-thujone is useful as an insecticide and an antihelminthic agent for the treatment of parasitic worms[2]. However, α -thujone is a toxic substance that disrupts neurological signals in the brain. Ingestion of the essential oils of Thuja leaves can cause death[2]. Seed with a pair of narrow lateral wings, seedlings produce 2 cotyledons. The wood is light, soft and aromatic. Thuja poles also often used to make fence posts and rails. The wood of Thuja is commonly used for guitar sound boards[3]. It is used as a medicinal plant in various forms of traditional medicines like folk medicine, homeopathy and treatment of bronchial catarrh, enuresis, cystitis, psoriasis, uterine carcinomas, amenorrhea and rheumatism[4-6]. Oil of Thuja contains thujone which has been studied for its GABA (gamma-aminobutyric acid) receptor antagonistic, with potentially lethal properties[2]. A yellow dye is obtained from the young branches[7]. Thuja is also occasionally used for treating diseases of skin, blood, gastrointestinal tract, kidney, brain, warty excrescences, spongy tumors[6]. Dubey and Batra[8,9] reported that the hepato-protective activities and antioxidant activity of Thuja occidentalis. Anti- proliferative and apoptosis- inducing properties of Thuja occidentalis has been evaluated by Biswas et al.,[6].
2. Chemical Constituents
- Thuja orientalis leaves contain rhodoxanthin, amentofl- avone, hinokiflavone, quercetin, myricetin, carotene, xan- thophylls and ascorbic acid. The fruit and roots are strongly aromatic. Distillation of the dried roots yields an essential oil having the following properties- Sp.gr.200, 0.971[α]D, -22.50 nD20, 1.5055: acid val, 2.1 ester. Val. 26.27; ester. Val. After acetylation, 89.39; Carbonyls (as C10H16O), 5.65% and 50% in 7-8 vols of 95% alcohol[10]. The composition of the oil is as follows: a new bicyclic sesquiterpene 51.10; l-borneol, 17.10; bornyl acetate, 9.1; α-thujone and camphor, 5.6; and a new sesquirterpenenic alcohol. The seed yields fatty oil having the fallowing composition palmitic 5.28, stearic, 7.3; C18 unsaturated acids, 1829 (linolenic, 44.6%); and C20 unsaturated acids, 6.10%. The heartwood contains aroma- dendrin, taxifolin, widdrene, cedrol, thujopsadiene, dehydro -α-curcumene, β-isobiotol and Curcumenether. It also con- tains an essential oil C is a complex blend of: Sesquiterpene hydrocarbons (cuparenes) 40; alcohols (Cedrol, widdrol, cuparenols) 50; monoterpenic acids[10]. Nickavar et al.,[11] 19 and 28 compounds have been identified in the volatile oils of the fruit and leaf, respectively, while the fruit oil contained α-pinene (52.4%), 3-carene (14.2%), α-cedrol (6.5%) and- phellandrene (5.1%), the leaf oil contained α-pinene (21.9%), α-cedrol (20.3%), 3-carene (10.5%) and limonene (7.2%) as the main components.Thujone is a ketone and a monoterpene that occurs naturally in two diastereomeric forms: α-thujone and β- thujone[12].
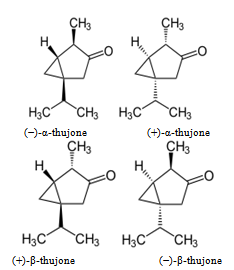 | Schema 1. Structure of diastereomeric isomer of thujone |
3. Biological Effects
- The main constituents of essential oils mono- and sesquiterpenes including carbohydrates, phenols, alcohols, ethers, aldehydes and ketones are responsible for the biological activity of aromatic and medicinal plants[12,13]. Thuja orientalis is used internally in the treatment of coughs, haemorrhages, excessive menstruation, bronchitis, asthma, skin infections, mumps, bacterial dysentery, arthritic pains and premature blandness[14]. The leaves are antipyretic, astringent, diuretic, emmenagogue, emollient, expectorant, refrigent, and stomachic[15,16]. Their use is said to improve the growth of hair[15]. The seed is aperients, lenitive and sedative. It is used internally in the treatment of palpitations, insomnia, nervous disorder and constipation in the elderly. The bark is used in the treatment of burn and scalds. The stems are used in the treatment of coughs, cold, dysentery, rheumatism and parasitic skin diseases[16]. Thujone was a weak inhibitor of acyl-CoA: lysophosphatidylcholine acyltransferase activity in mouse brain synaptosomes compared to psychoactive cannabinoids[17].
3.1. Antibacterial Activity
- Plants and their essential oils are potentially useful sources of antimicrobial compounds. Thuja orientalis contain large amounts of three substances (alpha, beta and gamma thujaplicin) that in low concentration would serve as chelators for Solmonella typhimurium[18]. T. orientalis was very effective in inhibiting the growth of serotypes c and d of Solmonella mutans (MIC less than or equal to 2.0-7.8 mg/ml)[19].
3.2. Antifungal Activity
- The essential oil showed antifungal activity in the inhibition zone against Alternaria alternata and Currularia lunata in a direct bioautography assay by lipophilic leaf extract of T. orientalis. Best bioactive component (Rf = 0.80) were observed and noted for antifungal activity. It produced an inhibition zone of 30 and 22 mm in diameter against A. alternata and C. Lunata, respectively[20,21]. Mishra et al.[22] reported antifungal activity of aqueous leaf extract of T. orientalis against Curvularia lunata. The essential oils from leaves, twigs and stems of large trees and shrub-like trees of Thuja sutchuenensis were extracted by hydrodistillation and supercritical fluid extraction and analysed by GC and GC-MS. The essential oils exhibited a certain degree of antifungal activity against six strains of human pathogenic fungi[23].
3.3. Antiviral Activity
- The chemical composition of the essential oil of T. orientalis was determined by GC/MS analysis. Essentials oils have been evaluated for their inhibitory activity against Sever Acute Respiratory Syndrome Coronavirus (SARS- Coronavirus) and Herpes Simplex Virus Type-1 (HSV-1) replication in vitro by visually scoring of the virus-induced cytopathogenic effect post-infection[24,25]. Several researches have demonstrated that allopathic extracts of T. orientalis could be used as strong antiviral agents against plant and animal viruses[26,27].
3.4. Inflammatory Activity
- Vascular inflammation is involved in the inhibition and progression of cardiovascular disease including atherosclerosis. Anti-vascular inflammatory activity of an aqueous extract of T. orientalis (ATO) and its possible mechanisms were investigated in human umbilical vein endothelial cells (HUVECs)[28]. Pre- incubation of ATO inhibited tumor necrosis factor and also inhibited U937 monocytes adhesion to HUVECs stimulated by tumor necrosis factor (TNF) suggesting that it may inhibit the binding of monocytes to endothelium. Furthermore, ATO significantly inhibited TNF-induced production of intracellular reactive oxygen species (ROS). Overall, ATO has an anti- inflammatory activity which is at least in part, is due to the decrease in the TNF-induced endothelial adhesion to monocytes by inhibiting intracellular ROS production, NF-kB activation and cell adhesion molecule in HUVECs[28,29].
3.5. Anticancer Activity
- Strong 5αa-reductase inhibitor are extracted and fractionated from T. orientalis and purified as diterpenes in isolated form[30]. The inhibitors are used either on their own or as active ingredients of therapeutics in the treatment of diseases caused by the over activity of 5αa-reductase or the hyper- secretion of androgens, such as male baldness, androgenetic alopecia, hirsutism, acne, prostatomegaly and cancer of the prostate[30,31]. Dubey and Batra[8,9] reported that the hepato-protective activities and antioxidant activity of Thuja occidentalis linn. Anti-proliferative and apoptosis- inducing properties of thujone-rich fraction (TRF) separated from Thuja occidentalis. Their possible anti-cancer potentials have been noted in the malignant melanoma cell line A375[6]. Sunila et al.,[32] concluded that a polysaccharide, or long-chain sugar molecule, derived from Thuja leaves extract decreased the inflammation caused by cancer. It also prevented the cancer from metastasizing, or spreading throughout the body.
3.6. Larvicidal Activity
- Larvicidal activities of T. orientalis oil against 4th-instar larvae of Aedes aegypti and Culex pipiens pallens has been observed by Ju-Hyun et al.[33]. Larvicidal activity of T. orientalis leaf oil was higher than those of stem, fruit, and seeds oils. Essential oils of leaves and fruits of T. orientalis at 400 ppm caused 100% and 71.6% mortalities against A. aegypti[33]. The lavricidal activity was observed from various age class (I-III) and found strong mortality in age class of II of T. orientalis against Aedes aegypti and Culex pipiens pallens larvae. Leaf part and age class II of T. orientalis has strong larvicidal activity against Aedes aegypti and Culex pipiens pallens. Leaf oil of T. orientalis shows natural larvicides against Aedes aegypti and Culex pipiens pallens[33].
3.7. Insecticidal Activity
- Leaf extracts of T. orientalis shows a repellent activity against Chilo partellus. T. orientalis ether extract (68.63%), acetone extracts (67.51%) have sufficient repellent action [34]. Foliar application of semi-solid crude extract of T. orientalis on maize was very effective against Chilo partellus[35].
3.8. Nematicidal Activity
- Ethanolic extract of Thuja orientalis leaf concentrations (20,40,60, and 80%) at 3 time intervals caused mortality in egg juvenile of Meloidogyne incognita[36]. It revealed a linear relationship between the concentration of the plant extract and the number of eggs hatched. Mortality of juveniles was directly proportional to the concentration and time of plant extracts[36].
3.9. Molluscicidal Activity
- Singh and Singh[37] reported that the ethanol extract of T. orientalis leaf (24 h LC50- 32.74 mg/l) and column purified fraction (24 h LC50- 29.25 mg/l) were potent molluscicide against Lymnaea acuminata. Thujone (24 h LC50- 08.09 mg/l) was identified as active molluscicidal component in T. orientalis. The molluscicidal activity of leaf/fruit of Thuja orientalis and their active components/column purified fraction with synergist Piperonyl butoxide (PB) and MGK- 264 (ENT 8184) was studied in binary combination (1:5) against L acuminata. Combination of T. orientalis leaf/ thujone or fruit powder/ column extract of T. orientalis fruit with PB or MGK-264 indicate synergised the toxicity up to 189.02 times. Toxicity of binary combination was increased hundreds folds than their individual components indicating synergistic action[38]. Sub-lethal (40% and 80% of 24h LC50) in vivo treatments of column purified fraction of Thuja orientalis and their active molluscicidal component thujone significantly inhibited the acetylcholinesterase (AChE), acid and alkaline phosphatase (ACP/ALP) activities in the nervous tissue of Lymnaea acuminata [39].It can be concluded from the ongoing literature that T. orientalis has the great potential against a number of health problem viz. bacterial, fungal and worm infection. It has antioxidant, antiviral, insecticidal nematicidal and molluscicidal activity. Recently, it has shown carcinogenic property. It needs greater attention by the researchers to explore its full potential and efficient use in the human welfare.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-Text HTML
Full-Text HTML