-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Polymer Science
p-ISSN: 2163-1344 e-ISSN: 2163-1352
2016; 6(3): 75-85
doi:10.5923/j.ajps.20160603.03

Highly Durable and Novel Anticorrosive Coating Based on Epoxy Reinforced with Poly(Aniline-co-Pentafluoroaniline)/SiO2 Composite
Brij Mohan Singh Bisht1, 2, Hema Bhandari3, Pradeep Sambyal4, S. P. Gairola2, Sundeep K. Dhawan4
1Chemical Division, National Test House, Kamla Nehru Nagar, Ghaziabad (U.P.), India
2Uttaranchal University, Prem Nagar, Dehradun (Uttarakhand), India
3Department of Chemistry, Maitreyi College, University of Delhi, India
4Polymeric & Soft Materials Section, CSIR-National Physical Laboratory, New Delhi, India
Correspondence to: Sundeep K. Dhawan, Polymeric & Soft Materials Section, CSIR-National Physical Laboratory, New Delhi, India.
| Email: |  |
Copyright © 2016 Scientific & Academic Publishing. All Rights Reserved.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The paper presents the development of high strength anticorrosive coating based on epoxy formulated with poly(aniline-co-2,3,4,5,6 pentafluoro aniline)/SiO2 composite. The composites display outstanding barrier properties against corrosive ions. SiO2 particles were found to significantly improve the microstructure of the coating matrix and thus enhanced both the anticorrosive performance and mechanical property of the epoxy coating. Electrochemical characterization was carried out in 3.5 wt% NaCl which show a remarkable improvement in the corrosion resistance of epoxy coating with addition of copolymer composites. The high pore resistance indicates the slow migration of ions into the metal and coating interface. Remarkable reduction of corrosion current density from 84.0 µA/cm2 in epoxy coating to 2.0 µA/cm2 in epoxy with copolymer/SiO2 composite coating was observed. Salt spray test of coated mild steel was carried out in 5.0 wt.% NaCl solution for 120 days as per ASTM B117 method. Electrochemical studies and salt spray test indicate the superior anticorrosive performance offered by the co-polymer composite under accelerated test conditions. Mechanical testing such as bend test, Taber abrasion resistance and cross cut adhesion test of epoxy formulated copolymer composite were carried out to evaluate the durability and adhesion performance of coating on the metal surface. The results show that the coating based on copolymer/SiO2 composites has improved corrosion resistance performance as well as mechanical properties more than several times as compared to epoxy coating. Better mechanical and barrier properties of the epoxy based copolymer composite were able to make the coating highly durable and anticorrosive in aggressive environment. Moreover, the significantly improved corrosion protection and mechanical strength is attributed to the synergistic effect of SiO2 particles and pentafluoro aniline in copolymer chain. We expect that the present concept opens new ways to prepare conducting polymer composite based epoxy coating for anticorrosion and other functional purposes.
Keywords: Anticorrosive Coating, Copolymers Composites, Tafel Plot
Cite this paper: Brij Mohan Singh Bisht, Hema Bhandari, Pradeep Sambyal, S. P. Gairola, Sundeep K. Dhawan, Highly Durable and Novel Anticorrosive Coating Based on Epoxy Reinforced with Poly(Aniline-co-Pentafluoroaniline)/SiO2 Composite, American Journal of Polymer Science, Vol. 6 No. 3, 2016, pp. 75-85. doi: 10.5923/j.ajps.20160603.03.
Article Outline
1. Introduction
- Corrosion is a vast technological problem, which causes a serious waste of money. However, now a days, people are not troubled only with the waste of money but also with pollution problems aggravated by some hazardous chemicals used in protection of metals from corrosion. Use of protective coating has been one of the most efficient method for protection of metals from corrosion. Uses of chromates, which are well known and very effective anticorrosive agent, are very hazardous to people health due to their carcinogenic nature. Moreover, metal rich primers such as zinc require high pigment volume concentration and have negative environmental effect. Taking into consideration, the limitations of existing coating system along with rapidly growing heavy industrializations and increasing pollutants, there is need to discover environmentally gentle solution to protect the metals from corrosion having superior property. Conducting polymers (CPs) such as polypyrrole, polyaniline and their copolymers have attracted attention for their potential in the protection of metals against corrosion due to their low cost, ease of synthesis, non toxic property, thermal and chemical stability [1-5]. The use of conducting polymers and their copolymers [6-10] as corrosion inhibitors have widely been used for protection of metals in aggressive environment. Anticorrosive coatings of conducting polymers on the metal surface are also found to be an effective method to control the rate of corrosion. These coatings present dual protection to underlying metals against corrosion. They act as a barrier layer and show high resistance towards the diffusion of corrosive ions [11]. Moreover, they protect the underlying metals by forming a very protective iron oxide layer that generally consists of Fe2O3 and Fe3O4 and have a tendency to replenish the oxide layer [12], if the coating is damaged. Conducting polymers as either film forming corrosion inhibitors or as protective coating have attracted great attention due to excellent anticorrosion ability and their eco friendly nature [13]. In the past decade, the use of polyaniline as anticorrosive coatings had been highly explored as the potential candidates to replace the materials having chromium [14, 15]. However, there are a few drawbacks of polyaniline such as insolubility, poor mechanical properties, poor adherence on metal surface, difficult processability and porosity [16-18]. Consequently, protective properties of the polymer coating may be lost with time. Also, porosity and anion exchange properties of conducting polymers could be unfavourable, particularly when it comes to pitting corrosion due to small aggressive anions (e.g. chlorides). In order to overcome these drawbacks one of most important solution has been employed by the several researchers such as use of conducting polymer composites with inorganic materials. Conducting polymer/inorganic composites have also attracted more and more attention, different metals and metal oxide particles have so far been encapsulated into the shell of conducting polymer to produce a host of composite materials. These composite materials have exposed better mechanical, physical and chemical properties due to combining the properties of conducting polymers and inorganic particles [19-21]. Recently; many researchers improved anticorrosion properties of polyaniline using its composites with active pigments such as glass flake [22], metal oxide [23], zeolite minerals [24] etc. which is used with epoxy coatings in the corrosive environment. Polyaniline-Fe2O3 composite is reported to offer excellent corrosion protection [25]. Shi et al. have reported the anticorrosion performance of mild steel using organic coating containing TiO2 and SiO2 [26]. Recently Jadhav and Gelling investigated anticorrosive performance of conducting polymer/TiO2 composites for protection of cold rolled steel [27].Among these inorganic fillers, SiO2 is an excellent reinforcing material for conducting polymers; it has tremendous potential in anti corrosive applications. As filler in polymer coatings, it improves the resistance of the coatings towards penetration of chloride ions and significantly reduces corrosion process. It was observed from the literature that the coatings prepared from conducting polymer-SiO2 composites synthesized by in situ chemical polymerization revealed an excellent corrosion resistance and found to be much superior to conducting polymers in aggressive environments [3]. The novelty of these coatings lies in the generation of corrosion protection by enhancing the barrier properties and preventing easy charge transport of corrosive ions when the coating is damaged by a scratch or scribes. Corrosion protective coatings on the mild steel surface by electrochemical deposition of conducting polymers composites have been widely studied [28, 30]. These types of coatings lack long lasting ability in a corrosive medium. The development of conducting polymer composites to be used as corrosion protection with hydrophobic properties, greater durability and high wear resistance is expected to encourage a major revolution in the world of corrosion. Hydrophobic substrates have ability to remove the water droplets, dust particles and other contaminants from the surface. Recently Dhawan et al. developed highly hydrophobic anticorrosive coating based on polyaniline-SiO2 composites using perfluoro-octanoic acid as a dopant [31]. The purpose of using perfluoro-octanoic acid as a dopant was to introduce the hydrophobic character in the coating.Hence, the present study was aimed at the development of highly durable, cost effective hydrophobic and eco-friendly coating based on conducting copolymer composite; poly(aniline-co-pentafluoro aniline)/SiO2 with epoxy resin on mild steel panels and to study the physico-mechanical properties as well as anti-corrosion performance of these coatings by cross cut adhesion test, bend test and abrasion resistance test. An attempt is done to show the synergistic effect of individual materials (aniline, pentafluoroaniline, and SiO2) on coating system. In order to improve the adherence capacity and efficiency of conducting polymer coating on the metal surface, the use of powder coating technique have been performed in the present paper. The key benefits of powder coating technique is to achieve durable, environmentally friendly, cost effective, excellence finish coating in single coat.
2. Experimental
2.1. Materials
- Aniline (AN), 2,3,4,5,6 pentafluoro aniline (PFA) and Tetraethylorthosilicate (TEOS) (Merck Chemicals) were distilled and kept under nitrogen at 4 °C prior to synthesis. Ortho phosphoric acid and ammonium persulphate (Merck Chemicals) were purchased from Across Organics. The dimension of the mild steel specimens for corrosion tests and salt spray test are 10mm × 40mm × 2mm and 150mm × 100mm × 2mm, respectively. These specimens were subjected to metallographic polishing by grinding them with emery papers of 120, 600 and 800 grit size to get a smooth finish. Thereafter, the steel specimens were degreased properly with acetone and kept in desiccators.
2.2. Synthesis of the SiO2
- SiO2 was synthesized in presence of ammonia as catalyst and ethanol as solvent by hydrolysis method of tetra-ethylorthosilicates (TEOS) [32]. Aqueous ammonia (0.1M) was added to a solution containing ethanol (1.0 M) and 20 ml of distill water which was stirred for 1-2 h then 0.05M TEOS was added and again stirred for 1 hour at room temperature. Formation of SiO2 particles was indicated by appearance of white turbid suspension this suspension was recovered by centrifugation and further calcination at about 820 K for 6-7 h. The formation of SiO2 particles was confirmed by FTIR spectroscopy and SEM analysis.
2.3. Preparation of Poly(AN-co-PFA)/SiO2 Composites
- The copolymerization of freshly distilled aniline (AN) and 2,3,4,5,6 pentafluoro aniline (PFA) was carried out by chemical oxidative polymerization using ammonium peroxydisulfate (APS) as an oxidant and ortho phosphoric acid as a dopant. For preparation of poly(AN-co-PFA)/SiO2 composite. The aqueous mixture of aniline (0.1M), PFA (0.01M), ortho phosphoric acid (0.2M) and SiO2 (20g) was homogenized using high speed blender for about 30-40 minutes to form an emulsion. The emulsion solution has been transferred to double walled glass reactor under constant stirring. The polymerization was initiated by the drop wise addition of aqueous solution of APS (0.1 M). The polymerization was carried out at a temperature of 0-5 °C for a period of 5-6 h. Synthesized copolymer composite was isolated from reaction mixture by filtration and washed with distilled water to remove oxidant and oligomers and followed by drying in the vacuum oven at about 60 oC. The schematic diagram for the preparation of poly(AN-co-PFA) is shown in scheme 1.
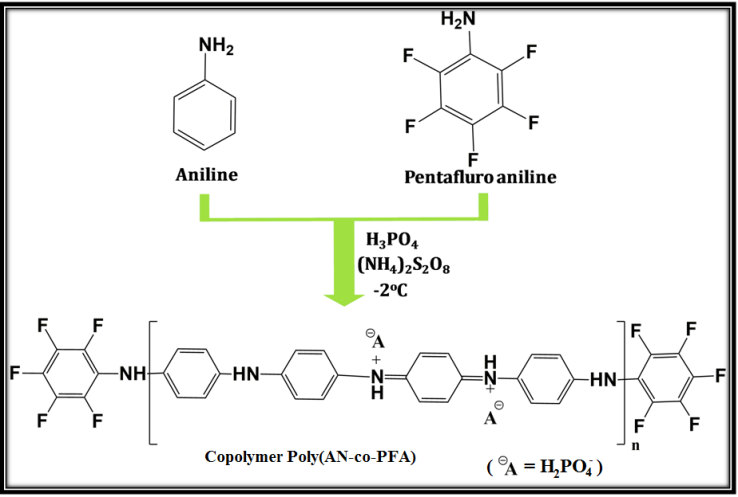 | Scheme 1. Schematic diagram of formation of Poly(AN-co-PFA) |
2.4. Development of Epoxy Formulated poly(AN-co-PFA)/SiO2 Composites Coating on Mild Steel
- Mild steel panels were coated with different wt. ratio of poly(AN-co-PFA)/SiO2 formulated with epoxy resin used for corrosion studies. Surface treatments were applied on the mild steel panels before coating. Cleaning of the panels was carried out by 1/0, 2/0, 3/0 and 4/0 grade emery papers. The panels were then thoroughly cleaned using trichloroethylene and acetone to remove any contamination on the surface. The powder polymer was mixed with epoxy formulation in various proportions ranging from 1.0 % to 4.0 wt. %. The epoxy powder coating formulation of composition: resin {epoxy (bisphenol A + polyester) (70 wt.%), flow agent (D-88) (2.3 wt.%), degassing agent (benzoin) (0.7 wt.%), and fillers (TiO2 and BaSO4) (27 wt.%) was used for the coating purpose. Different weight percentage (1.0, 2.0, 3.0 and 4.0) of the synthesized copolymer composite was blended with epoxy resin using a laboratory ball mill. The blended powder coating formulation was applied on mild steel panels using an electrostatic spray gun held at 67.4 kV potential with respect to the substrate (grounded). The powder coated mild steel panels were cured at 130-140°C for 25 min. Mild steel panels coated with epoxy resin is designated as EC, epoxy coatings with different wt,% loadings of poly(AN-co-PFA) are designated as PF1 (1.0 wt.%), PF4 (4.0 wt.%) and epoxy coatings with different wt,% loadings of poly(AN-co-PFA)/SiO2 composites are designated as PFS1 (1.0 wt.%), PFS2 (2.0 wt.%), PFS3 (3.0 wt.%) and PFS4 (4.0 wt.%).
3. Characterization
3.1. Characterization of Copolymer Composite
- The structure of the copolymer composite was characterized by Fourier Transform Infrared (FTIR) Spectrometer (Nicolet 5700 FTIR, USA) in KBr pellets in the range of 400-4000 cm-1. TGA (Mettler Toledo TGA/SDTA 851e, Switzerland) was used to study the thermal stability of copolymer composite in nitrogen atmosphere at the heating rate of 10oC/min. Scanning Electron Microscope (JEOL- JSM-6360A) was used to the morphology of copolymer-SiO2 composite. The energy dispersive X-Ray (EDX) was used to determine the elemental composition of the copolymer composite. The particle size and morphology of the copolymer composites were studied using Transmission Electron Microscopy (Tecnai G2 F30 S-Twin) operated at an accelerating voltage of 300 kV, with a point resolution of 0.2 nm and a lattice resolution of 0.14 nm. Contact angle measurements was used to check the hydrophobic nature of the copolymer composite coated mild steel by the Sessile drop method using a drop shape analyzer (DSA 100, DSA/V 1.9).
3.2. Characterization of Epoxy Formulated Copolymer Composite Coated Substrate
3.2.1. Mechanical Testing of Coating
- Adhesion of coating to mild steel surface was evaluated by cross cut adhesion test. This test was carried out at room temperature as per ASTM D3359-09 (Test method B). In order to carry out this test, a lattice pattern with either six or eleven cuts in each direction was made in the film to the substrate, pressure-sensitive tape was applied over the lattice and then removed, and adhesion of coating on metal surface was evaluated.Coating performance on the metal surface was evaluated using mandrel bend test as per ASTM D522M/D522-93a. This test was carried out by placing the coated test samples over a mandrel (size 6.25 mm) with the uncoated side in contact and with at least 50 mm overhang on either side using a steady pressure of the fingers bend the panel approx. 180 degree around the mandrel at a uniform velocity in a time 1.0 s remove and examine the panel for cracking visible to the unaided eye. Abrasion Resistance test of coated panels were carried out using Taber Abrasion apparatus as per ASTM D 4060-95. This test was carried out on the rigid panels (dimension 10 cm2) having uniform coating. The specimens were a disk of 100 mm in diameter or a plate (100mm) square with rounded corners and with a 6.3-mm hole centrally located on each panel. The test was performed at 25°C and 50 % relative humidity for 24 h. The organic coating was applied at uniform thickness to a plane. After curing, the surface was abraded by rotating the panel under 1.0 Kg weighted abrasive CS10 wheels. A test of 1000 cycles was done to observe the weight loss in coatings. Abrasion resistance was calculated as loss in weight at a specified number of abrasion cycles, as loss in weight per cycle, or as number of cycles required to remove a unit amount of coating thickness.
3.2.2. Corrosion studies of Coating
- Electrochemical studies of epoxy formulated copolymer composites coated mild steel panels were carried out using a three electrode cell system having coated mild steel specimens (1 cm2 exposed area) as working electrodes, Platinum as counter electrode and Ag/AgCl as reference electrode. Autolab Potentiostat/ Galvanostat, PGSTAT100 (NOVA Software) was employed for the electrochemical characterization. Open Circuit Potential (OCP) vs time, potentiodynamic polarization (Tafel plots) and Electrochemical Impedance Spectroscopy (EIS) were performed in 3.5 wt.% NaCl solution at room temperature (25 ± 2 °C). The test specimens were kept under OCP conditions in NaCl solution for 24 h, prior to the electrochemical tests. Tafel polarization was performed at a constant scan rate of 1.0 mV/s by sweeping the potential between±250mV vs Ag/AgCl from OCP. The values of corrosion potential (Ecorr) and corrosion current density (icorr) are extracted from the Tafel plots. The linear Tafel segments to the anodic and cathodic curves (-0.2 to + 0.2 V versus corrosion potential) were extrapolated to corrosion potential to find the corrosion current densities. The corrosion current density [icorr (A/cm2)] was calculated with the Stern-Geary equation [33]. The corrosion protection efficiency (% P.E.) of coated mild steel samples was determined from the measured icorr (corrosion current density of epoxy coated mild steel electrode (iEcorr) and corrosion current density of epoxy formulated polymer coated mild steel electrode (ipcorr) using the following relationship;
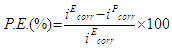 [Eq 1.0]EIS was performed to find the values of pore resistance (Rp) and coating capacitance (Cc). The measurements were carried out in a frequency range of 100 kHz to 0.1 Hz at OCP by applying a potential signal of 10 mV. Each experiment was repeated thrice to check the reproducibility of the test results.
[Eq 1.0]EIS was performed to find the values of pore resistance (Rp) and coating capacitance (Cc). The measurements were carried out in a frequency range of 100 kHz to 0.1 Hz at OCP by applying a potential signal of 10 mV. Each experiment was repeated thrice to check the reproducibility of the test results. 3.2.3. Salt Spray Tests
- Coated mild steel panels were prepared by coating on fully finished mild steel specimen dimension 15.0 cm × 10.0 cm × 0.12 cm. The coated panels were exposed to salt spray of 5.0% NaCl solution for 120 days as per ASTM B117 method.
4. Result and Discussion
4.1. FTIR Spectra
- The Fig.1 shows the FTIR spectra of SiO2, poly(AN-co-PFA) and poly(AN-co-PFA)/SiO2. The FTIR spectra of SiO2 showed the characteristic peaks at 1099 cm-1 and 802 cm-1 are assigned to the stretching and bending vibration of Si–O–Si respectively [3].
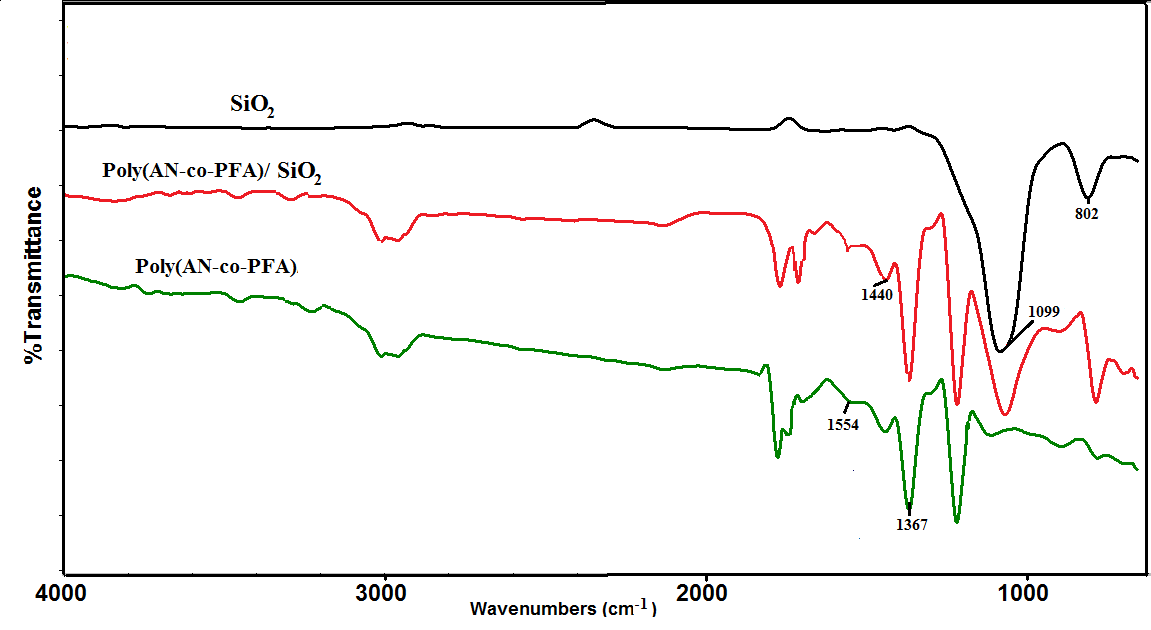 | Figure 1. FTIR Spectra of SiO2, Poly(AN-co-PFA) and Poly(AN-co-PFA)/ SiO2 composites |
4.2. Thermo Gravimetric Analysis (TGA)
- Fig. 2 shows the thermo-gravimetric curves (TGA) of SiO2, poly(AN-co-PFA) and poly(AN-co-PFA)/SiO2 composites. These samples were heated from 25 to 700°C under a constant heating rate of 10°C/min and inert atmosphere of nitrogen gas.
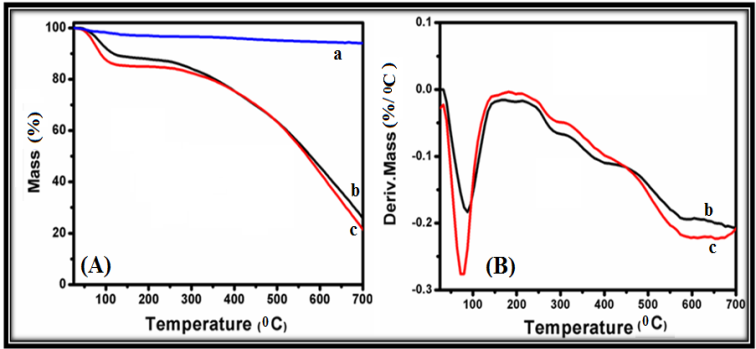 | Figure 2. (A) TGA and (B) DTG curves of (a) SiO2, (b) Poly(AN-co-PFA) and (c) Poly(AN-co-PFA)/ SiO2 composite |
4.3. Microstructural Analysis
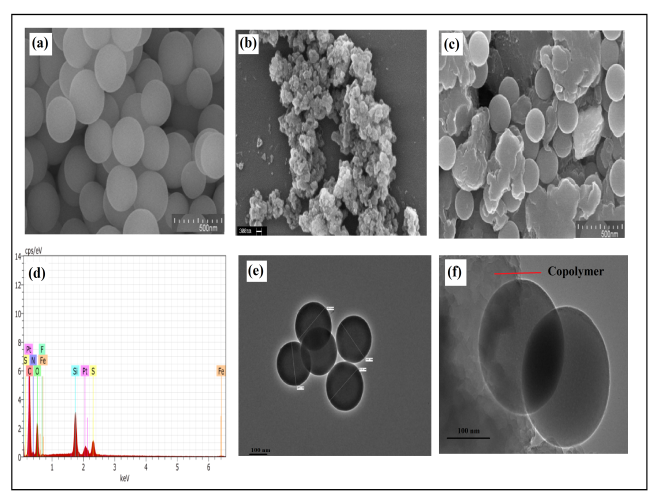
- Fig.3 shows the SEM micrograph of SiO2, poly(AN-co-PFA) and poly(AN-co-PFA)/SiO2 respectively. The SEM micrograph revealed the spherical morphology of SiO2 particles. The SEM micrograph of poly(AN-co-PFA) composite showed cauliflower like morphology. The dimension of SiO2 particles was found to be in range of 200-300 nm as shown in Fig. 3 e. SiO2 particles were found to be well dispersed in the copolymer matrix which suggested good compatibility between SiO2 and copolymer matrix as shown in SEM (Fig. 3c) and TEM image of poly(AN-co-PFA)/SiO2 composite (Fig.3f). Fig.3d shows the EDS analysis of poly(AN-co-PFA)/SiO2. The EDS analysis of poly(AN-co-PFA)/SiO2 composite shows the peaks of Carbon, Oxygen, Nitrogen, Fluorine, Phosphorous and Silicon which indicates the presence of PFA and SiO2 molecules in copolymer chain.
4.4. Contact Angle Measurement
- This test is useful for characterizing the wettability of coated surfaces. A low contact angle value (<90°) is indicative of wetting ability of the coating. Water has been used as a test liquid to establish whether a surface is hydrophilic (angle <90°) or hydrophobic (angle >90°) in nature. It was observed that surface contact angle of epoxy coating with poly(AN-co-PFA) and poly(AN-co-PFA)/SiO2 composite was found to be 95° resulting in hydrophobic coating as shown in Fig.4. While the contact angle of epoxy coating with water droplets was 73°.
 | Figure 4. Contact angle images of (a) Epoxy coating and (b) epoxy with Poly(AN-co-PFA)/SiO2 coating |
4.5. Physico-mechanical Testing of Coating
4.5.1. Cross Cut Tape Test
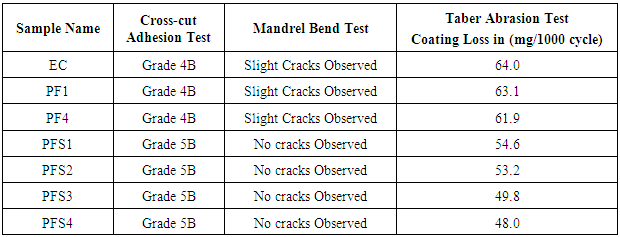
- Fig 5A shows the results of cross cut tape test carried out on mild steel panels with epoxy and epoxy with different loading of poly(AN-co-PFA) and Poly(AN-co-PFA)/SiO2 composites. It was observed that the small flakes of coating were detached along edges and at intersection of cuts in epoxy and epoxy containg poly(AN-co-PFA) coated mild steel. The area affected in epoxy coated panel was found to be about 5% (Grade 4B), while epoxy coating with different loading of poly(AN-co-PFA)/SiO2 showed completely smooth edges of cuts and none of the square of the lattice was detached (Grade 5B). Results indicate that incorporation of SiO2 particles in copolymer matrix reinforced the coating on the metal surface.
4.5.2. Mandrel Bend Test
- Fig. 5B shows the Mandrel Bend Test result of the epoxy coating and epoxy with copolymer composite coating. It was observed from the Fig. 5B(a) that the panels coated with epoxy resin showed slight cracks on metal surface this indicates the detachment of coating from metal surface. On the other hand, panel coated with epoxy formulated poly(AN-co-PFA) and poly(AN-co-PFA)/SiO2 composites did not show any cracks on the coating surface as shown in Fig. 5B (b) and 5B (c). This indicates that the copolymers and copolymer composites in epoxy resin were able to enhance the binding properties of coating.
4.5.3. Taber Abrasion and Test
- Taber abrasion test is used to evaluate the abrasion resistance of attached coatings. This test showed the wear resistance of the coatings. Abrasion resistance was calculated as loss in weight at a specified number of abrasion cycles or as number of cycles required to remove a unit amount of coating thickness.Fig.5C and Table 1 shows the Taber Abrasion test results of epoxy coating and epoxy with different loading of copolymer composite coating, which indicate that epoxy coating showed the weight loss up to 64.0 mg/1000 cycle. There was no remarkable reduction of weight loss in PF1 and PF4 coated samples. These samples showed the weight loss near to epoxy coating while epoxy coating containing copolymer composites (PFS3 and PFS4) showed remarkable reduction in weight loss per 1000 cycle. It was observed to be reduced from 64.0 mg/1000 cycle (EC coating) to 48.0 mg/1000 cycles (PFS4 coating). Hence, epoxy formulated poly(AN-co-PFA)/SiO2 coatings behave as good wear resistance comparable to epoxy coating and epoxy with poly(AN-co-PFA) coating.
|
4.6. Salt Spray Test
- Salt spray test was performed on epoxy coated, epoxy with poly(AN-co-PFA) and poly(AN-co-PFA)/SiO2 composites coated mild steel panels in 5.0 wt.% NaCl (pH of 6.5–7.2) for 120 days as per ASTM B117 method. All the coated mild steel panels were provided with a scribe mark across the panel and placed in salt spray chamber. Fig.6 shows the photographs of epoxy coated (EC) and epoxy with PF4 and copolymer composite (PFS1, PFS2, PFS3 and PFS4) coated mild steel panels after exposure to salt spray fog of 5.0 wt.% NaCl solution. Results show that the presence of spread of corrosion along the scribe mark for epoxy coated steel panel only on 60 days of exposure to salt spray chamber. The presence of rust on coating shows the loss of adherence of the epoxy coating during prolong exposure to salt fog. Severe blistering and pin holes were also noticed. The addition of copolymers (PF1 and PF4) and co-polymer composite (PFS1, PFS2, PFS3 and PFS4) in epoxy coatings improve the corrosion resistance properties of the epoxy resin this can be clearly observed from the photographs (Fig.6).The epoxy coated specimen with 4.0 wt.% loading of copolymer (PF1and PF4) showed less extended corrosion along the scribe. Same behavior was observed in samples coated with PFS1 and PFS2. Further, almost no extension of corrosion along the scribe mark was observed for the samples coated with PFS3 and PFS4 (Fig.6). Specimen PFS3 and PFS4 also showed better corrosion resistance with less extended corrosion along the scribe mark as shown in Fig.6. The salt spray results clearly indicate that the poly(AN-co-PFA)/SiO2 composite effectively inhibit the spread of corrosion near scribe mark that exposes the steel surface. It is assumed that the copolymer; poly(AN-co-PFA) and its composite with SiO2 might have promoted the adhesion of the epoxy coating to the metal substrate and improving the corrosion resistance under accelerated test conditions. Furthermore, the results revealed that the inorganic filler such SiO2 particles also play an important role in improving the corrosion protection performance of the coating. As compared to epoxy and epoxy with poly(AN-co-PFA) coating, poly(AN-co-PFA)/SiO2 based epoxy coating provided efficient protective function against delamination around scribed areas, ensured intense galvanic function of the primer.
 | Figure 6. Photographs of (a) epoxy coated (EC) and epoxy with PF4 and different loading of Poly(AN-co-PFA)/SiO2 composite coated mild steel specimens exposed to salt spray fog after 120 days |
4.7. Electrochemical Studies of the Coating
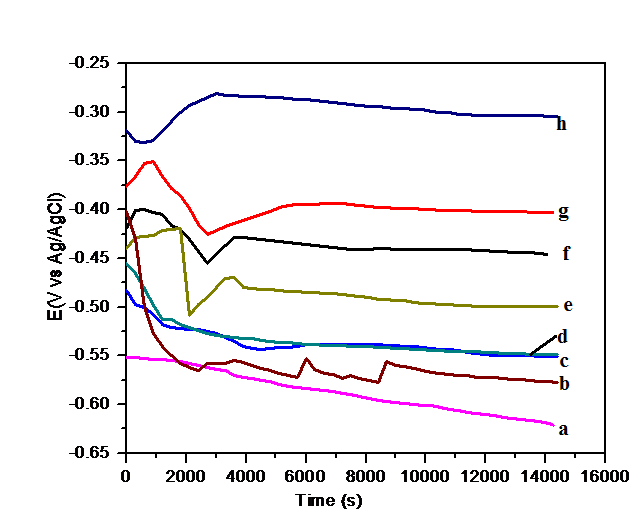
4.7.1. Open Circuit Potential (OCP) vs Time
- The corrosion behavior of mild steel coated with epoxy, epoxy containing different weight loading of copolymers and copolymer composites was investigated by OCP vs. time measurements. The samples were immersed in 3.5 wt.% NaCl solution and the OCP was recorded versus time at room temperature (25±2°C). The protection time was characterized by the time during which the OCP of the covered electrode remains in the passive state of mild steel before it drops down to the corrosion potential of unprotected mild steel. The OCP-time curves of uncoated and polymer coated mild steel in NaCl solution is shown in Fig.7. The trend of OCP variation for bare mild steel shows a continuous shifting of OCP towards negative potential. A steady state OCP is not noticed for the metal. This is owing to the occurrence of general corrosion on metal surface in presence of corrosive medium as shown in Fig.7a.The OCP curves of epoxy, PF1 and PF4 coated mild steel were initially high at about -0.40 V,-0.48 V and -0.44 V respectively, which indicates that these coated mild steel electrodes maintains in its passive state at initial time of immersion. The trend of OCP variation for epoxy coating showed a sharp decrease OCP towards negative potential within few minutes of immersion. This is due to the rapid diffusion of chloride ions through the coating. However, the potential decreased sharply within few minutes and became almost stable till the end of the test. This is due to the penetration of the large concentration of chloride ions in the epoxy/mild steel interface and thus epoxy cannot protect mild steel any longer. The OCP curve of epoxy coating with copolymers (PF1 and PF4) also exhibits similar trend of the variation of potential. The potential reduced sharply and then shifted towards the slightly positive direction followed by the attainment of steady state value till the end of immersion time (Fig.7c and 7e), this behavior indicates that the copolymers coating was more able to protect the metal from corrosion as compared to epoxy coating.
4.7.2. Tafel Extrapolation Measurement
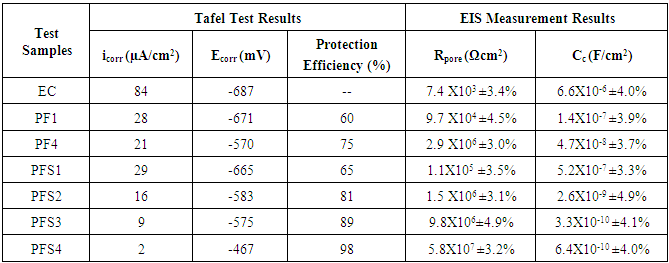
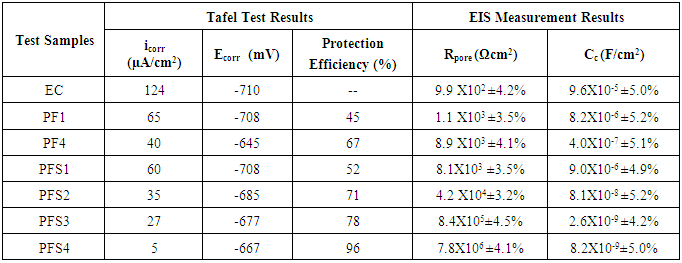
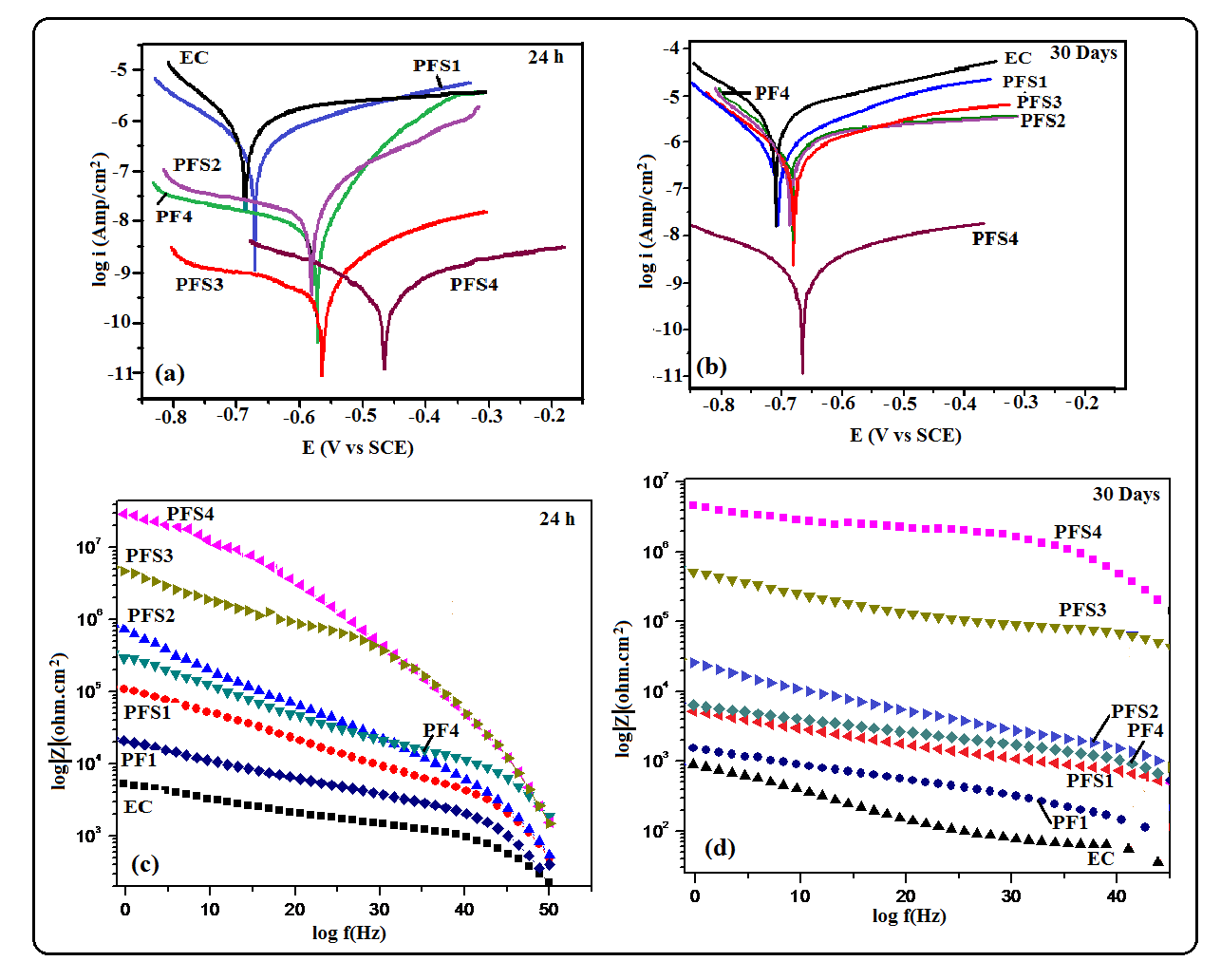
- Corrosion protection performance of various polymeric coatings was investigated by electrochemical studies using Tafel Extrapoaltion method. Tafel polarization study of epoxy coated (EC) and epoxy with different wt.% loadings of copolymer composite coated mild steel sample (PFS1, PFS2, PFS3 and PFS4) was carried out after immersing the mild steel samples in 3.5 wt.% NaCl solution for different interval such as 24 h to 30 days at room temperature (25 ± 2°C) as shown in Fig 8a. The electrochemical parameters related to Tafel plots like corrosion potential (Ecorr) and corrosion current density (icorr) are shown in Table 2 and Table 3. Results show that the coated mild steel specimens exhibited significantly lower icorr values and more positive corrosion potential (Ecorr) with respect to uncoated mild steel. This suggests that the coatings promote the generation of a more stable passive layer and protect the underlying metal against corrosion. The icorr value of epoxy coated sample was 84.0 µA/cm2 which was reduced to 16.0 µA/cm2 in case of PFS2, 9.0 µA/cm2 in PFS3 and 2.0 µA/cm2 in PFS4 coated samples. The icorr value was found to decrease on increasing the loading of poly(AN-co-PFA)/SiO2 composite in epoxy coating. As a result, the corrosion protection efficiency (% P.E.) as calculated from equation 1.0 was found to be 89% and 98.0% for specimens PFS3 and PFS4 respectively as shown in Table 2. Epoxy coating was found to be known for their brilliant barrier properties and good adhesion to their metal substrates [28, 29] under corrosive conditions.
|
|
4.7.3. Electrochemical Impedance Spectroscopy (EIS)
- Electrochemical impedance spectroscopy (EIS) is the method in which the impedance of an electrochemical system is studied as a function of the frequency of an applied A.C. wave. EIS is a nondestructive electrochemical technique for the evaluation of quality and corrosion protection performance of organic coatings. Impedance measurements were carried out by immersing the test specimens in 3.5 wt.% NaCl solution at open circuit potential conditions for 24 h to 30 days at room temperature. The impedance graphs obtained for epoxy coated mild steel electrode (EC) and epoxy with different wt.% loadings of copolymer composite (PFS1, PFS2, PFS3 and PFS4) coated electrode are exhibited in Bode plots. In general Bode plots exhibit the different stages of degradation of coatings on metal surface. The unexposed coating systems act as a pure dielectric, separating the mild steel substrate from the corrosive medium such as NaCl solution. This nature of the coating system results in pure capacitive behavior [5]. Fig 8B shows the Bode plots of epoxy coated and epoxy containing different loading of copolymer composites coated mild steel electrode at 24 h of immersion in 3.5 wt. % NaCl solution. The electrical resistance of a coating system is measured in terms of pore resistance (Rp) and it indicates the performance of the surface coating. Whereas, coating capacitance (Cc) is an important parameter to measure the integrity of the coating and is related to water uptake tendency of the coating. It was observed that epoxy coated mild steel sample showed that pore resistance value up to about 7.4x103 Ωcm2 which was found to increased to 5.8x107 Ωcm2 on addition poly(AN-co-PFA)/SiO2 composite in epoxy resin. The pore resistance for the epoxy and epoxy with different loading of poly(AN-co-PFA) and poly(AN-co-PFA)/SiO2 composite coated samples as measured using EIS data fitting and listed in Table 2. Results indicate that the Rp value increased on increasing the loading level of poly(AN-co-PFA)/SiO2 composite in epoxy resin Moreover, Rp value of epoxy coating containing poly(AN-co-PFA)/SiO2 composite was found to be higher than epoxy containing poly(AN-co-PFA) coating. The high pore resistance value is characteristic of well protective coatings on mild steel surface [35]. It was observed that these coatings had a good resistance to penetration of corrosive ions through the films structure at the beginning of immersion in corrosive solution and had very low probable paths for water molecules and other ions to diffuse from coating-water interface to the metal-coating interface. However, evaluation of corrosion resistance of coatings for relatively longer period of immersion is equally important. Therefore, impedance parameters of the coatings were also measured at 30 days of immersion in 3.5% NaCl solution and results are shown in Table 3. Among the test samples, the epoxy coated mild steel electrode showed low pore resistance and high coating capacitance (Cc) on 30 days of immersion as shown in Table 3. This is owing to the high water uptake by the epoxy coating due to its poor barrier property. The Rp value increased with increasing the loading level of copolymer composites (from PFS1 to PSF4) in epoxy resin as compared to epoxy coated mild steel sample. The mild steel sample coated with PFS4 showed the highest value of pore resistance (7.8x106 Ωcm2) and lowest Cc (8.2X10-9 F/cm2) exhibiting its superior corrosion protection performance even during prolong immersion time. The coating capacitance (Cc), occur in the decreasing order as EC, PFS1, PFS2, PFS3 and PFS4 as shown in Table 3. The low coating capacitance observed for epoxy coating containing 1.0 to 4.0 wt% loading of poly(AN-co-PFA)/SiO2 composite is due to the low penetration of electrolyte into the surface coating. Moreover, it was also observed that the Rp value of PF1 and PF4 coated mild steel was lower than PFS1 and PFS4, it indicates that the poor barrier property shown by epoxy coating based on poly(AN-co-PFA) as compared to poly(AN-co-PFA)/SiO2 composite containing coating. Epoxy coatings are well known for high adhesive strength due to the presence of polar groups in the structure which is chemically bonded to the metal surface. In the presence of corrosive medium, the interfacial bonding between epoxy/metal is destroyed because of the entry of electrolyte into the interface. This results in the failure of the coatings. Therefore in this copolymer composite system, SiO2 particles act as a reinforcing material which enhance the mechanical integrity of the coating. SiO2 particles in coating provide additional barrier protection to the substrate and inhibit the penetration of electrolyte into the interface.
5. Conclusions
- Conductive copolymer composites based on poly(AN-co-PFA)/SiO2 were prepared by insitu chemical oxidative polymerization. The study of FTIR, TGA, SEM and TEM confirmed the formation of copolymer composites. Synthesized copolymer composites were successfully formulated with epoxy resin in different loading and their coating was carried out on mild steel substrate by powder coating technique. The corrosion test studies in 3.5% NaCl medium have shown that the epoxy coating containing poly(AN-co-PFA)/SiO2 had higher corrosion protection property as compared to epoxy and epoxy formulated poly(AN-co-PFA) coating. The Tafel parameters indicate low corrosion current for coatings with 3.0 and 4.0 wt.% loading of copolymer composite in 3.5 wt.% NaCl solution. These coatings have shown superior corrosion protection even for prolong exposure to 3.5 wt.% NaCl solution. The results of mechanical testing such as abrasion resistance, scratch resistance, bend test and cross cut tape test for epoxy formulated poly(AN-co-PFA)/SiO2 coating were found to be superior than epoxy coating and epoxy formulated poly(AN-co-PFA) coating. Apart from the prevention of corrosion, it was observed that the epoxy formulated with (AN-co-PFA)/SiO2 coating showed better mechanical properties as compared to epoxy and epoxy with (AN-co-PFA) coating. It was observed that the mechanical properties of the epoxy based poly(AN-co-PFA)/SiO2 coatings were found to be significantly improved with the loading of poly(AN-co-PFA)/SiO2 composites in coating due to the presence of SiO2 particles in polymer matrix which reinforced the polymer by providing addition mechanical strength. The synergistic interaction between PFA and SiO2 in the copolymer composite resulted in the enhanced thermal, mechanical, hydrophobic and anticorrosive properties of coating. Hence, such system could be used as an effective anticorrosive coating on mild steel where the probability of corrosion is greater.
ACKNOWLEDGEMENTS
- Authors are highly obliged to Shri. Sher Singh Director National Test House Ghaziabad for his keen interest in the work. The authors are also thankful Dr. B.P. Singh, Punjab University, Chandigarh for recording the SEM and TEM characterization of the samples.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML