-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Polymer Science
p-ISSN: 2163-1344 e-ISSN: 2163-1352
2014; 4(3): 73-87
doi:10.5923/j.ajps.20140403.02
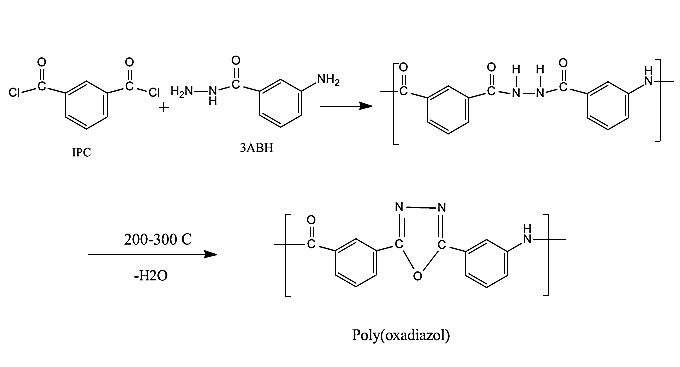
Preparation of Soluble Polyamides and Characterization of Their Composites with Carbon Nanotubes
Mohamed Nassar1, Maher Z. Elsabee2
1Science and technology center of excellence, Ministry of military production
2Department of Chemistry, Cairo University, Egypt
Correspondence to: Maher Z. Elsabee, Department of Chemistry, Cairo University, Egypt.
| Email: |  |
Copyright © 2014 Scientific & Academic Publishing. All Rights Reserved.
Hydrized derivatives of phenylene diamine were reacted with aromatic diacid chloride to give wholly aromatic polyamide/hydrazide compounds with much better solubility in organic solvents thus improving their presses- ability. Carbon nano-tubes were used as fillers to enhance their thermal and mechanical properties. The polymers as well as the composite were characterized by spectral and mechanical tools as well as by X-ray analysis.
Keywords: Polyamide, Poly (amide–hydrazides), Viscosity, Solubility, Thermal analysis, Dynamic Mechanical Analysis (DMA), XRD, DSC
Cite this paper: Mohamed Nassar, Maher Z. Elsabee, Preparation of Soluble Polyamides and Characterization of Their Composites with Carbon Nanotubes, American Journal of Polymer Science, Vol. 4 No. 3, 2014, pp. 73-87. doi: 10.5923/j.ajps.20140403.02.
Article Outline
1. Introduction
- Aromatic polyamides are thermally stable polymers with attractive combination of excellent chemical and mechanical properties, they are used in advanced technologies and have been transformed into high-strength and flame resistant fibers and coatings, with applications in the aerospace and armament industry, bullet-proof body armor, protective clothing, sport fabrics, electrical insulation, asbestos substitutes, and industrial filters, among others. Owing to their chemical structure, they exhibit extremely high transition temperatures that lie above their decomposition temperatures. However, their applications are usually hampered by their poor solubility which makes their processing very hard and difficult. Tremendous synthetic efforts, in the area of high-performance polymers have been focused on improving their process ability and solubility through design and synthesis of new monomers. A variety of structural modifications to the polyamide backbone have been employed in order to increase the solubility and consequently improving the processing process. Such trials included insertion of flexible side group on the main chain [1, 2] insertion of flexible linkage on the main chain [3, 4, 15] and utilization of noncoplanar or asymmetric monomers. In recent years, wholly aromatic polyamide-hydrazides have attracted considerable attention as one of the most important classes of specialty polymers. They exhibit some interesting and potentially useful characteristics that have enormous technical and economic importance. They have favorable rheological behavior which allowed for successful preparation of fibers and films; they possess high thermal and thermo-oxidative stability Aromatic poly (amid-hydrazides) has been synthesized by solution polycondensation at low temperature of aromatic hydrazides with aroyl dichlorides. By heating these polymers at tem peratures near 300, dehydrocyclization occurs to give polymers containing the oxadiazole ring in the main chain. Their excellent thermal stability has been tested by thermogravimetric analysis. A series of new poly (amide-hydrazide) s were obtained by the direct polycondensation with triphenyl phosphite and pyridine in N-methyl-2-pyrrolidone (NMP) solutions containing dissolved LiCl. All copolymers were soluble in polar solvents such as NMP and dimethyl sulfoxide (DMSO). Most of the amorphous hydrazide copolymers formed flexible and tough films by solvent casting. In this work a trial has been made to increase the solubility of polyamide by using hydrazide derivative of phenylene diamine to react with aromatic diacid chloride. In this way a wholly aromatic polyamide/hydrazide polymer could be obtained bearing the characteristics of polyamide and having better solubility in several solvents in this way the process ability of polymer will be improved. Multi walled nano-carbon tubes were used as a filer in order to increase the thermal and mechanical properties of the final product. The aim of work is mainly directed to the production of thermally stable composites which could have further application in armament applications.
2. Experimental
2.1. Materials
- Isophthaloyl chloride (C8H4Cl2O2), with minimum assay 99.0%, Terphthaloyl chloride (C8H4Cl2O2), with minimum assay 99.0%, were purchased from (Fluka), 3-Aminobenzhydrazide (C7H9N3O) with minimum assay 97%, 4-Aminobenzhydrazide (C7H9N3O) with minimum assay 97% were purchased from (Alfa Aesar) , Isophthalic Dihydrazide>95%(T) (C6H4(CONHNH2)2 was purchased from (TCI Europe), Multi-wall carbon nanotube (Nanoamor) Purity: > 95% outside diameter: 40-60 nm, Core diameter: 5-10 nm Length: 5-15 µm. Color: black, pore volume: 0.8 cm3/g. Bulk density: ~0.1 g/cm3. True density: ~2.1 g/cm3, SSA: 40-600 m2/g.1-Methyl-2-Pyrrolidone (C5H9NO) with minimum assay 98.0% was purchased from (Fluka), Ethanol absolute ≥99.8% was purchased from (Sigma Aldrich). Acetone and N, N-dimethylformamide (DMF) (BDH, England), Dimethylsulphoxide (DMSO) (Aldrich, Germany), were extra pure reagents and were used as received without further purifications. N, N-dimethylacetamide (DMAc) (Aldrich, Germany) was guaranteed reagents, dried over calcium hydride for 24 h, and followed by distillation under reduced pressure. The fractions, which boiled at 40–42°C/ 2 mm Hg were collected and stored over molecular sieves before use.
2.2. Polymerization Procedures
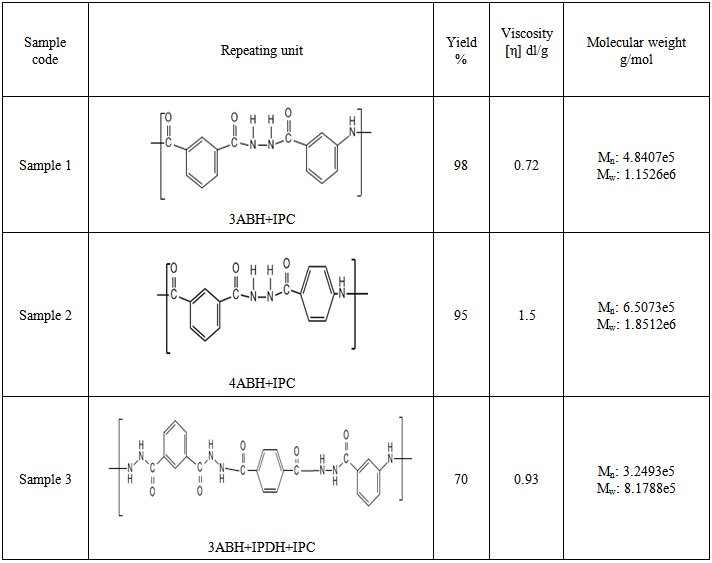
- All polymers were prepared by essentially the same experimental procedure, which is given here for:Preparation of polymer 1: 3ABH, 1.963 gm (0.013 mol), was charged into the reaction flask followed by 40 ml of dry NMP. Stirring was started until complete dissolution. The reaction flask was placed in a crushed ice-salt water bath and cooled at 10°C for 15 min. When this was completed, 2.639 g (0.013mol) solid isophthaloyl dichloride [ICl] was added slowly under constant stirring over a period of 1 h after this period, stirring was continued for another 2 h at the afore-mentioned temperature. The 1:1 ratio of ABH to ICl is chosen in order to obtain maximum molecular weight polymers.Then the cooling bath was removed and the temperature of the polymerization reaction was allowed to rise gradually to room temperature and maintained for 24 h with stirring. Afterwards, a clear, slightly yellow, highly viscous solution was obtained. Finally, the polymer solution was slowly poured into 300 ml of rapidly stirred methanol + distilled water upon which a fibrous white precipitate of polymer immediately formed. The polymer was isolated by filtration, washed successively with methanol, acetone and dried in a vacuum oven at 75°C to a constant weight. Polymer samples were purified by repeated precipitation from their solutions in NMP using methanol as a non-solvent. The precipitated polymers were isolated, washed and dried to constant weights was achieved (yield 98%).The reaction scheme is similar for the three copolymers and can be represented by schemes (1).
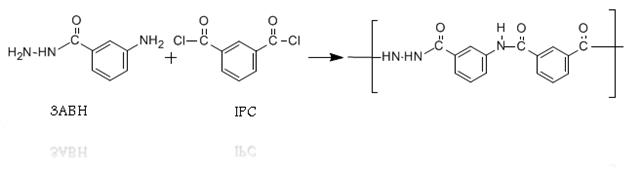 | Scheme 1. Polymerization reaction of 3BHA and IPC |
 In order to obtain uniform dispersion and strong interactions between the carbon nanotubes and the polymer matrix, carboxylic acid functional groups were introduced onto the nanotube surface. This synthetic functionalization procedure has been reported and briefly described as follows [5]:Oxidation of MWCNTs by H2O2 was carried out by stirring a MWCNT of 0.5 g in 30% H2O2 of 10 mL for 1-6 days at 65 °C. To keep sufficient concentration of H2O2, 5 mL of H2O2 was added to the reaction mixture every day. The oxidized MWCNTs were washed with deionized water, filtered through 0.45 in Milli-pore PVDF membrane. The filtrate was dried overnight at 110°C, and then was vacuum-dried for 6 h at 150°C to remove possible adsorbed H2O2 and attached peroxide groups on the surface of the MWCNTs.
In order to obtain uniform dispersion and strong interactions between the carbon nanotubes and the polymer matrix, carboxylic acid functional groups were introduced onto the nanotube surface. This synthetic functionalization procedure has been reported and briefly described as follows [5]:Oxidation of MWCNTs by H2O2 was carried out by stirring a MWCNT of 0.5 g in 30% H2O2 of 10 mL for 1-6 days at 65 °C. To keep sufficient concentration of H2O2, 5 mL of H2O2 was added to the reaction mixture every day. The oxidized MWCNTs were washed with deionized water, filtered through 0.45 in Milli-pore PVDF membrane. The filtrate was dried overnight at 110°C, and then was vacuum-dried for 6 h at 150°C to remove possible adsorbed H2O2 and attached peroxide groups on the surface of the MWCNTs.2.3. MWCNT/polymer Nanocomposites
- A desired amount of MWCNTs was added into a solvent the solution was sonicated in an ultransonic bath for 1 h at temperature of 50°C to separate the aggregation of the MWCNTs and achieve good dispersion. Then, the soluble polymer was mixed with the MWCNT/ solution, and softly stirred it for about 1h. After that, the nanocomposite solution was slowly poured into 300 ml of rapidly stirred in aqueous methanol upon which a fibrous grey precipitate of polymer immediately formed.The nanocomposite was isolated by filtration, washed successively with methanol, acetone and dried in a vacuum oven at 75°C to constant weight.
2.4. Film Preparation
- Films were prepared by casting 7% (w/v) polymer solutions (in DMAc) onto dry clean Pyrex glass plates to a uniform thickness. Solvent evaporation was performed at a constant temperature of 100 °C in an electrically heated oven with forced air circulation. The resulting films were kept in the oven until no change in weight could be observed. The films were then immersed in deionized water overnight to remove any residual impurities; finally the films were dried in a vacuum oven at 75°C to constant weight.
2.5. Instruments
- Infrared spectra of the prepared polymers were measured with Nicolet Avatar 370Cs2 FTIR spectro-photometer between 400 and 4000 cm -1 at 25°C. Intrinsic viscosity measurements were carried out on 0.5% solutions of the polymers in DMAc at 30oC using a suspended-level Ubbelohde viscometer with negligible kinetic energy correction. Flow times were measured at five different concentrations of the polymer sample. All the plots obtained were linear. Intrinsic viscosity was determined by usual extrapolation of ηsp/C to zero concentration and expressed in deciliter per gram (dl g-1). All the investigated polymers were found to be highly soluble in several organic polar solvents such as DMAc, DMSO, NMP, DMF, and at room temperature. Molecular weight of the polymers were measured using a GPC Instrument (Waters) at the National Research Centre in Cairo, DMF was the eluent solvent. Thermogravimetric analysis (TG) curves were recorded on a TA instrument TGA Q500 V20.10 Build 36 in nitrogen at a heating rate of 10°C min-1 and a heating range from room temperature to 800°C. Differential scanning calorimetry (DSC) curves were recorded on a DSC Q2000 V24.4 Build 116 in nitrogen at a heating rate of 10°C min-1 and a heating range from room temperature to 300 °C. X-Ray Diffraction was measured using Scintag/USA XGEN- 4000 at 45 kV and 40 mA using nickel-filtered CuKα radiation. Storage Modulus, Loss Modulus and Complex Viscosity of the polymers films were measured on TA instrument DMA Q800 V20.24 Build 43 in nitrogen from room temperature to 250°C.
3. Results and Discussion
- In this work a trial has been made to increase the solubility of polyamide by using hydrazide derivative of phenylene diamine to react with aromatic diacid chloride. In this way a wholly aromatic polyamide/hydrazide could be obtained bearing the characteristics of polyamide and having better solubility in several solvents in this way the process ability of polymer will be improved. Table (1) depicts physical data of the different polyamide-hydrazides, the content of para- and meta moieties was varied, it observed that the viscosity increase with increasing of Para content.
|
3.1. Characterization
3.1.1. FTIR
- The FTIR spectra of the polymers, Figs (1-3) show stretching vibration bands at the following wave numbers: (1) 3400–3200 cm-1 (intensive band) assigned to NH group; (2) 2360–2340 cm-1 (weak) is attributed to possible enol-type configuration of hydrazide and amide groups; (3) 1670–1650 cm-1 (strong) corresponded to the amide carbonyl group; (4) 1600 cm-1 indicated the aromatic carbon–carbon double bonds; (5) 1540–1520 cm-1 is due to -NH of amide II; (6) 1500 cm-1 indicated carbon– carbon single bond in ring; (7) 1420 cm-1 is attributed to carbon–oxygen (phenolic); and finally (8) 1480,1330–1320, 1290 and 1280 cm-1 corresponded to carbon– hydrogen in the aromatic ring.
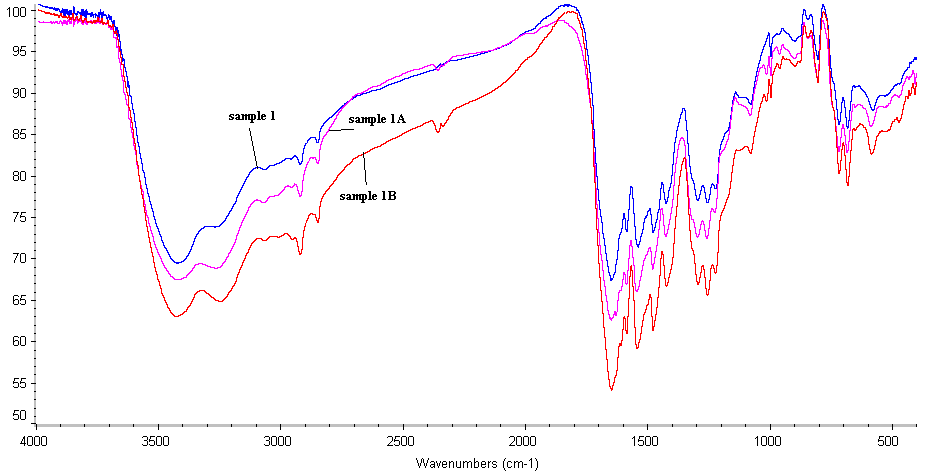 | Figure 1. FTIR of Sample 1, 1A, 1B |
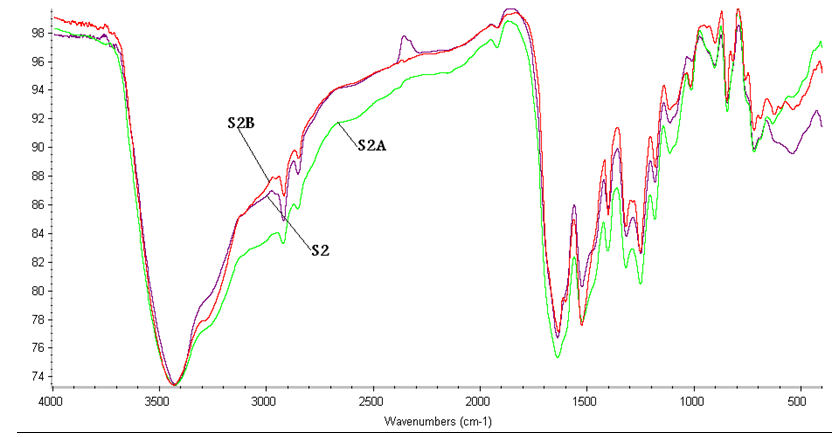 | Figure 2. FTIR of Sample 2, 2A, 2B |
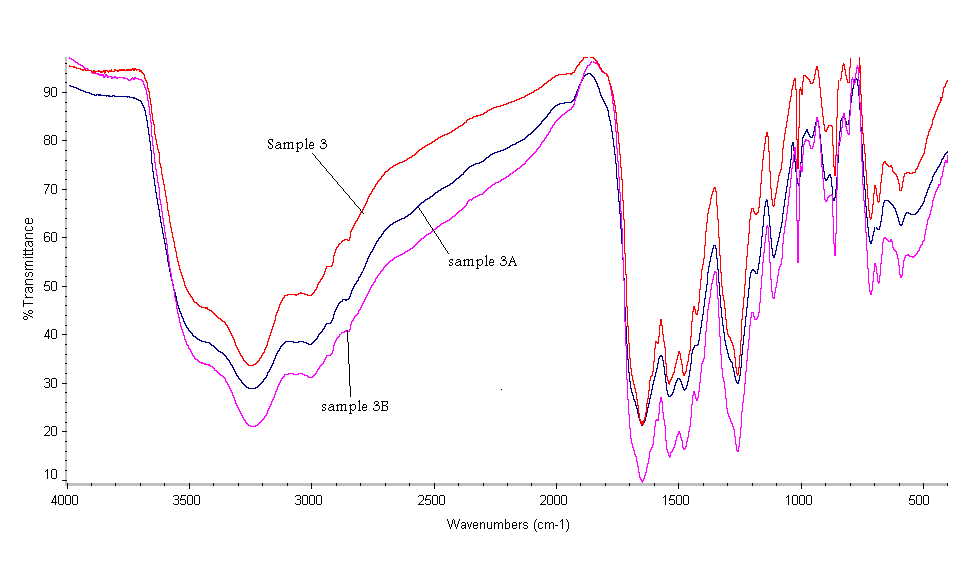 | Figure 3. FTIR of Sample 3, 3A, 3B |
3.1.2. X-Ray Spectral Analysis
- Figs (4-6) illustrate the X-ray diffraction pattern of powdered samples of poly (amide–hydrazide) s over the 2θ range of 5–60°.
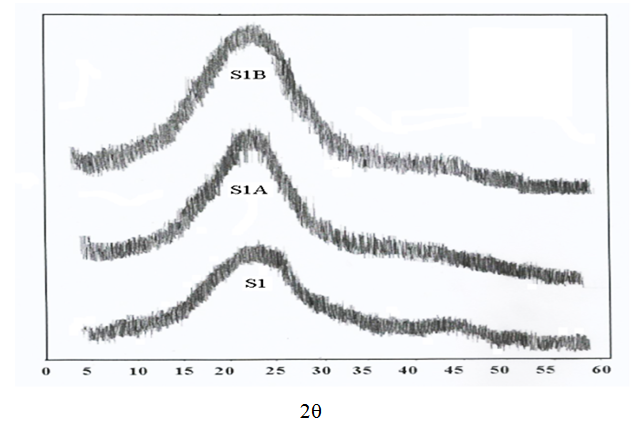 | Figure 4. X-Ray patterns for samples 1, 1A, 1B |
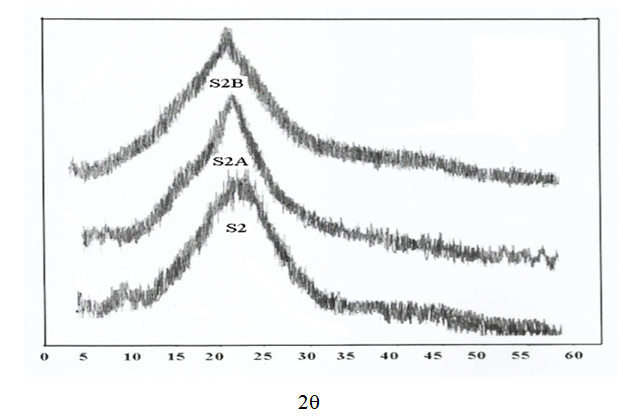 | Figure 5. X-Ray patterns for samples 2, 2A, 2B |
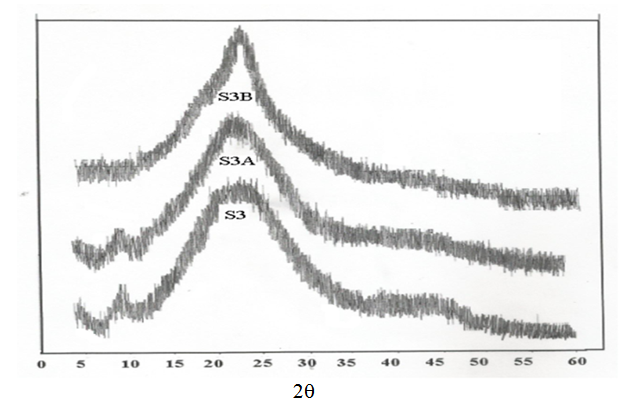 | Figure 6. X-Ray patterns for samples 3, 3A, 3B |
3.1.3. Thermo Gravimetric Analysis (TG)
- Thermal stability and degradation behavior of several wholly aromatic polyamide-hydrazides containing various proportions of FMWCNT were also investigated by TG measurements. All these measurements were performed at 10 min-1 heating rate, under the constant streams of purified nitrogen and the results obtained are shown in Figs (7, 8, 9).
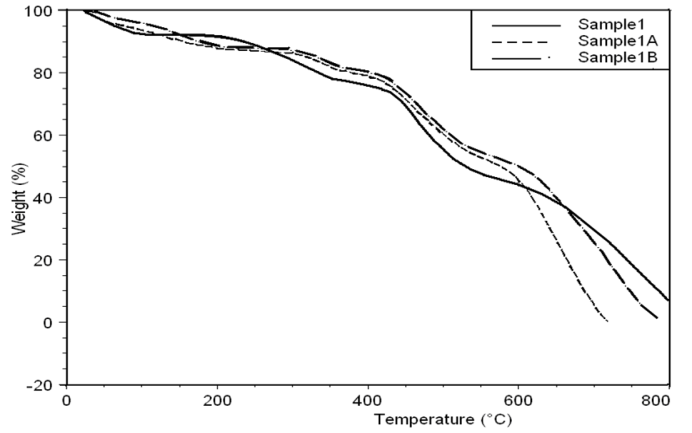 | Figure 7. Thermo gravimetric curves of sample 1, 1A, 1B in nitrogen at a heating rate of 10°C min-1 |
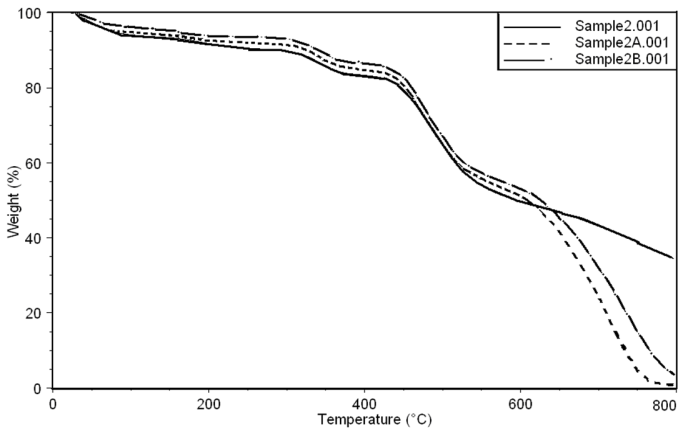 | Figure 8. Thermo gravimetric curves of sample 2, 2A, 2B in nitrogen at a heating rate of 10 °C min-1 |
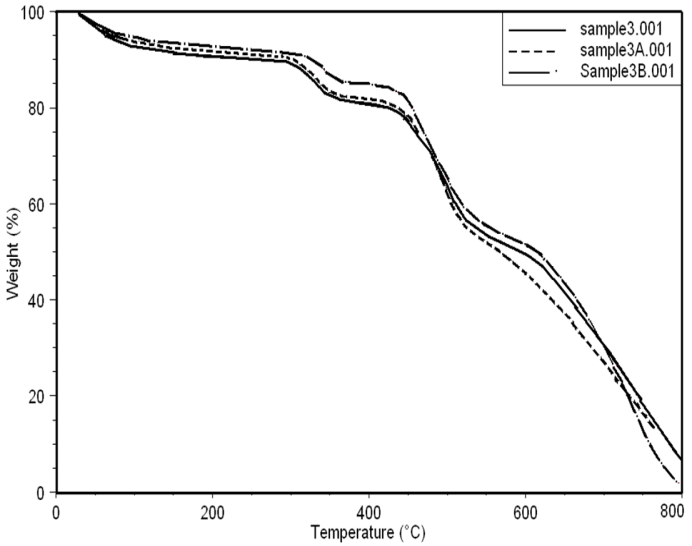 | Figure 9. Thermo gravimetric curves of sample 3, 3A, 3B in nitrogen at a heating rate of 10 °C min-1 |
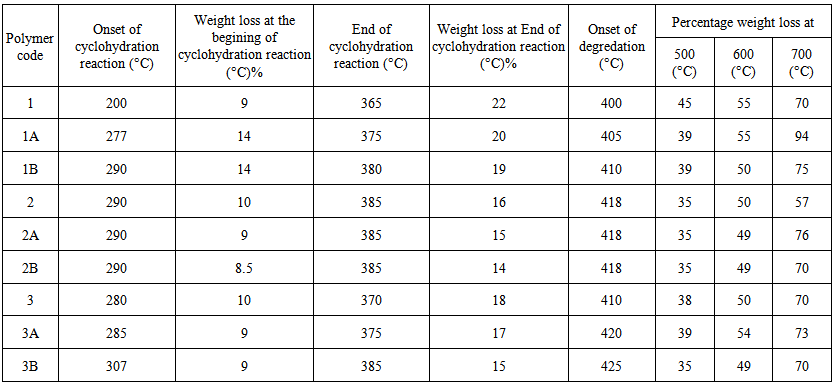 | Table (2). TGA analyses of aromatic polyamide–hydrazides samples |
3.1.4. Differential Scanning Calorimetry (DSC)
- Differential scanning calorimetry (DSC) curves are shown in Figs (10, 11, 12), it can be seen from these figures that Glass Transition temperature (Tg) values for poly (amide-hydrazide) s; could not be determined. On providing heat energy in DSC experiment, poly (amide-hydrazide) s start losing molecule of water at about 80– 100°C, due to water loss or residual solvents in these polymers.
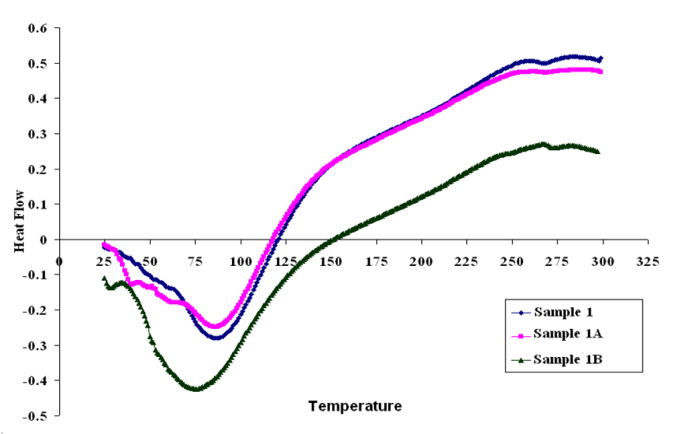 | Figure 10. DSC curves of sample 1, 1A, 1B in nitrogen at a heating rate of 10°C min-1 |
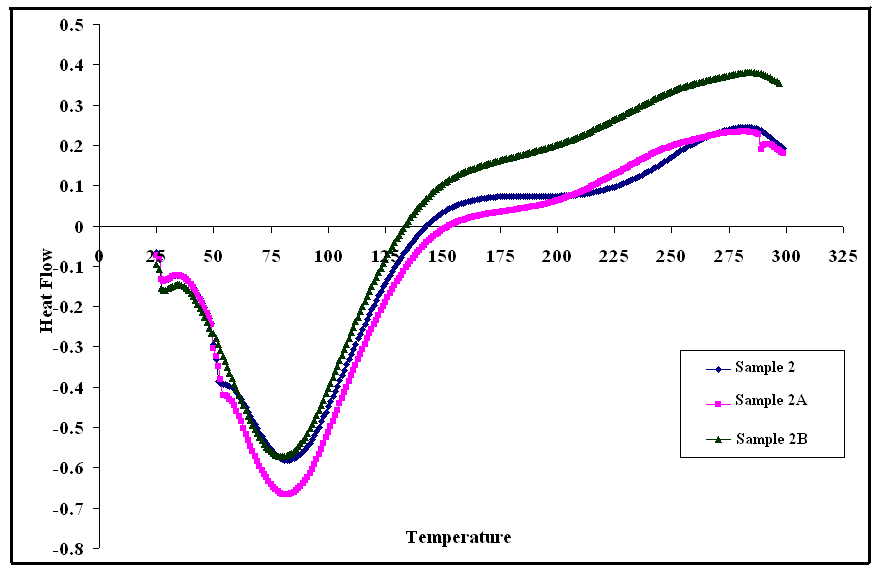 | Figure 11. DSC curves of sample 2, 2A, 2B in nitrogen at a heating rate of 10°C min-1 |
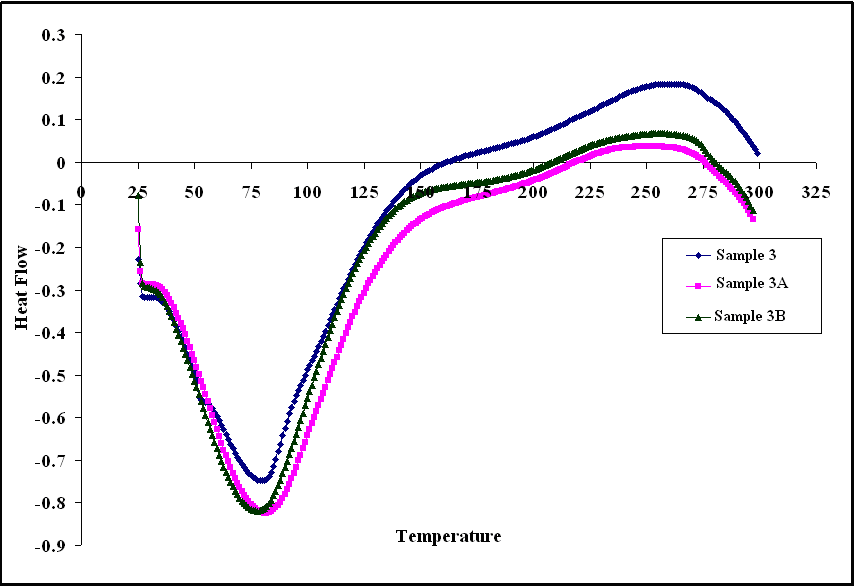 | Figure 12. DSC curves of sample 3, 3A, 3B in nitrogen at a heating rate of 10°/min |
3.1.5. Dynamic Mechanical Analysis(DMA)
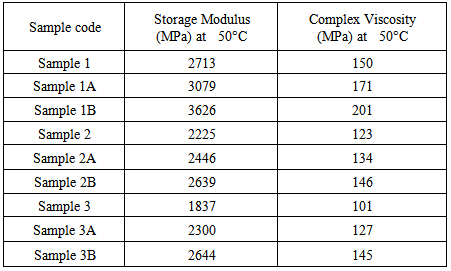
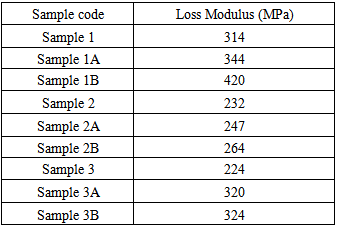
- Dynamic mechanical analysis (DMA) of the FMWCNTs nano-composites was carried out to monitor the effect of the FMWCNTs on the thermo-mechanical properties of the poly(amide-hydrazide) s films [11]. Values of the Storage modulus, Loss Modulus and Complex Viscosity at various FMWCNTs contents are plotted against temperature in Figs (13-18). The values of storage modulus and Complex Viscosity, for the neat poly (amide-hydrazide) s and the FMWCNT composites containing 0.1, 0.5 wt. % of FMWCNT at 50℃ are summarized in Table (3). The increase in Storage modulus and Complex Viscosity reflects the interaction between the FMWCNT and the poly (amide-hydrazide) s, which enhances the chain packing.
|
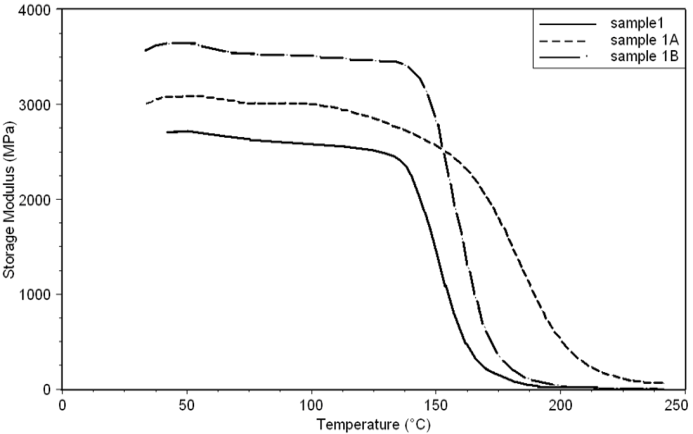 | Figure 13. Storage Modulus curves of sample 1, 1A, 1B at a heating rate of 10°C min-1 |
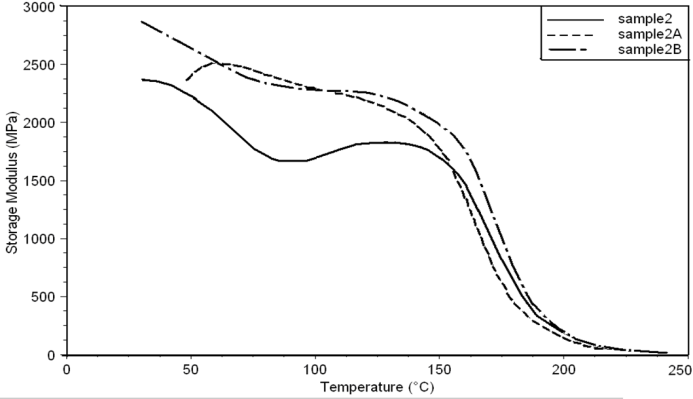 | Figure 14. Storage Modulus curves of sample 2, 2A, 2B at a heating rate of 10°C min-1 |
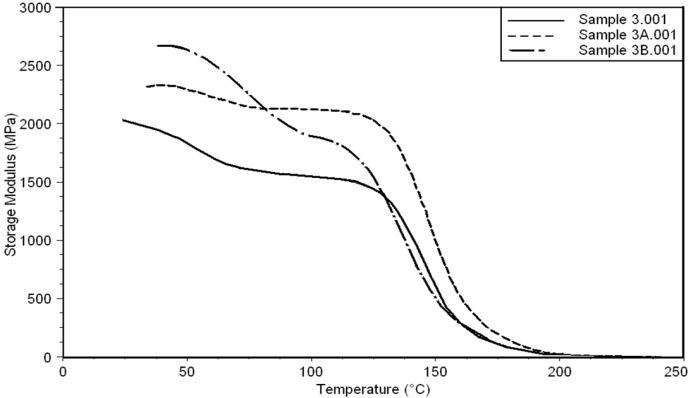 | Figure 15. Storage Modulus curves of sample 3, 3A, 3B at a heating rate of 10°C min-1 |
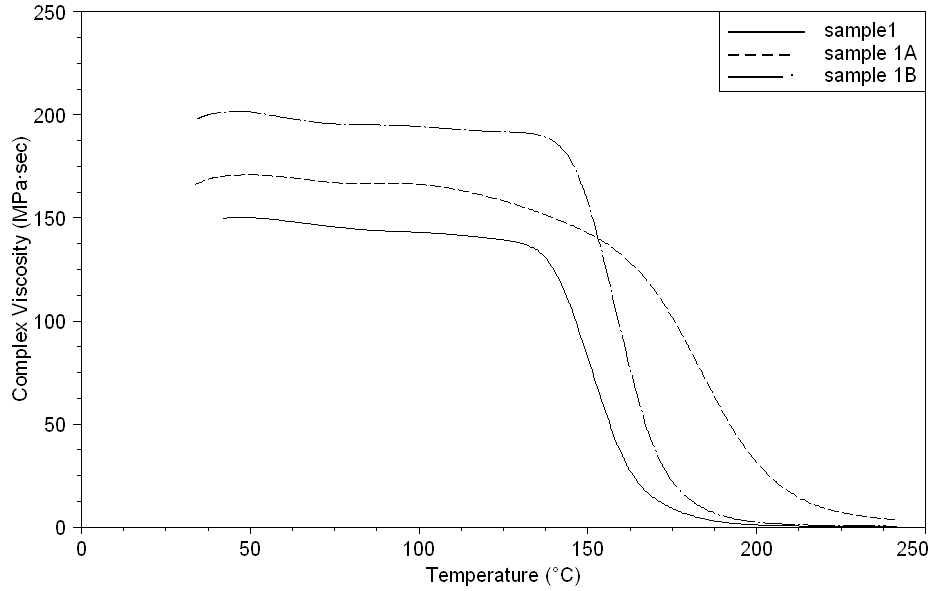 | Figure 16. Complex Viscosity curves of sample 1, 1A, 1B at a heating rate of 10°C min-1 |
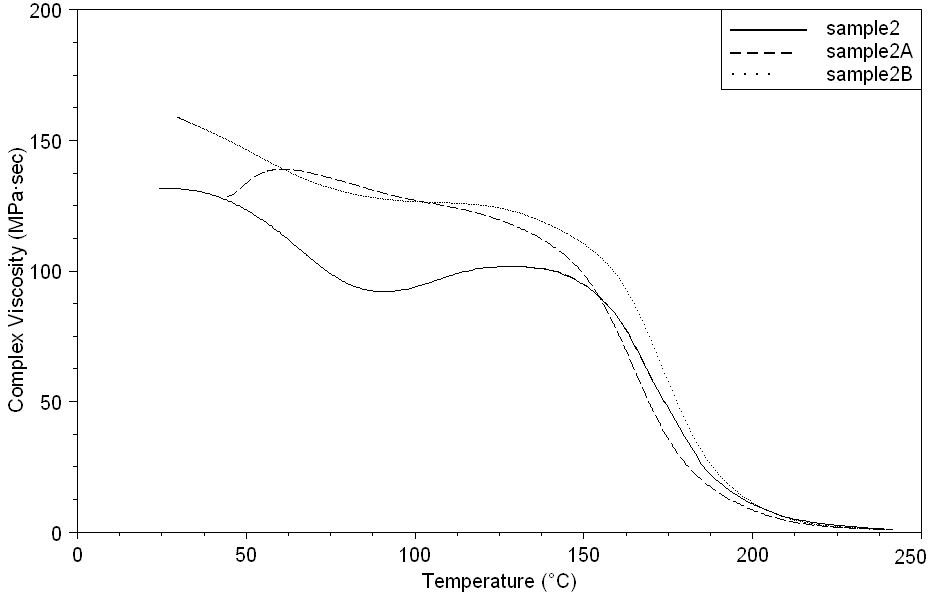 | Figure 17. Complex Viscosity curves of sample 2, 2A, 2B at a heating rate of 10°C min-1 |
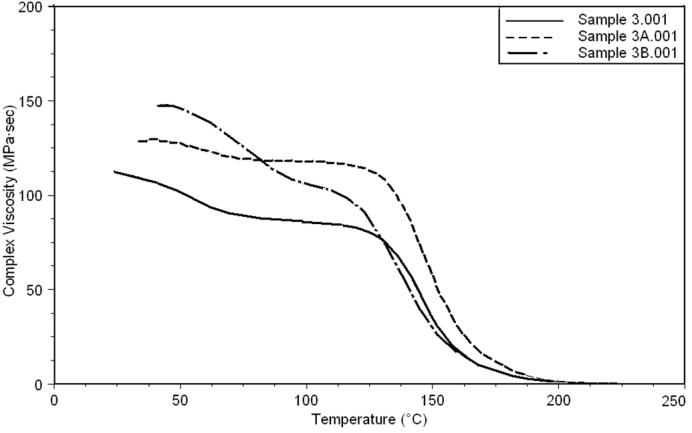 | Figure 18. Complex Viscosity curves of sample 3, 3A, 3B at a heating rate of 10°C min-1 |
|
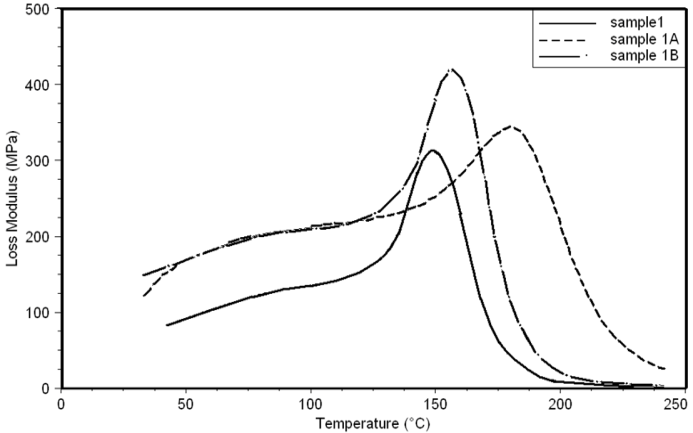 | Figure 19. Loss Modulus curves of sample 1, 1A, 1B at a heating rate of 10°C min-1 |
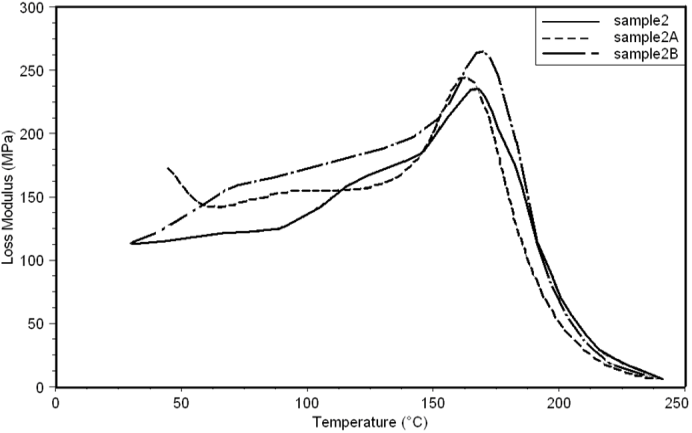 | Figure 20. Loss Modulus curves of sample 2, 2A, 2B at a heating rate of 10°C min-1 |
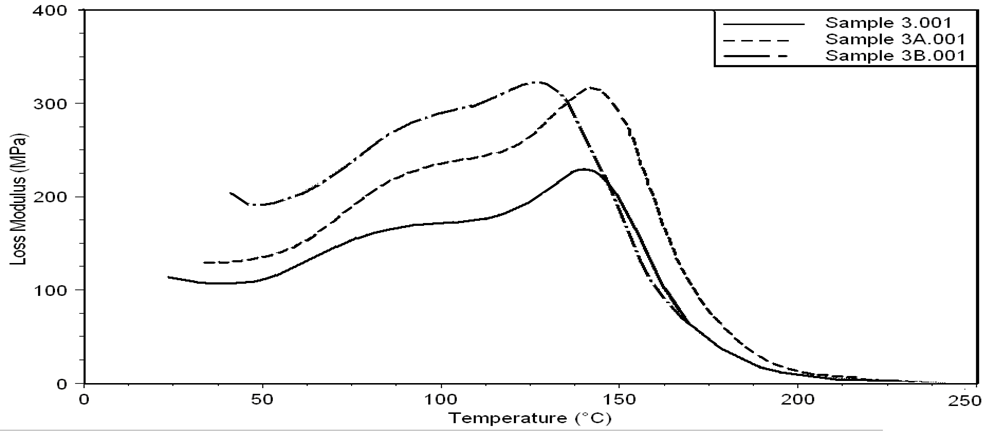 | Figure 21. Loss Modulus curves of sample 3, 3A, 3B at a heating rate of 10°C min-1 |
4. Conclusions
- Aromatic polyamides were found to be insoluble in most common solvents which render their fabrication a difficult task. By introducing a hydrazide group inside the polymer chain it showed dramatic effect on the solubility of the resulting polymer without affecting greatly its thermal or mechanical properties. Three different hydrazide/amide copolymers were prepared and showed good thermal characteristics. The copolymers also exhibited a degree of crystallinity which could enhance their mechanical properties. Applying carbon nano-tubes to the copolymers did not improve their characteristics; however when the carbon nano tubes were functionalized by treatment with hydrogen peroxide their attachment with polymers increased and an improvement in the thermal and mechanical characteristics was observed. The obtained composites are therefore good potential candidates for the production of high thermal and mechanically strong fibres which could be used for several industrial and military applications.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML