-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Organic Chemistry
p-ISSN: 2163-1271 e-ISSN: 2163-1301
2014; 4(1): 11-13
doi:10.5923/j.ajoc.20140401.02
Reaction of an Organotin Halide with Salicylideneimine Ligands
Burl Yearwood
Department of Natural Sciences, LaGuardia Community College (City University of New York), Long Island City, NY, 11101, USA
Correspondence to: Burl Yearwood, Department of Natural Sciences, LaGuardia Community College (City University of New York), Long Island City, NY, 11101, USA.
| Email: |  |
Copyright © 2014 Scientific & Academic Publishing. All Rights Reserved.
Butyltin trichloride was reacted with Salhexen(tBu) [Salhen(tBu) = N,N'-(hexylene) bis (3,5-di-tert-butyl) salicylideneimine] and Salhepten(tBu) [Salpen(tBu) = N,N'-(heptylene) bis (3,5-di-tert-butyl) salicylideneimine]. The products of the reaction were analyzed by proton and carbon NMR, Sn NMR, FT-IR, melting point, and elemental analysis. The structure of the final products was determined from the analytical results. It is proposed that the metal binds to the oxygen and nitrogen of the ligands.
Keywords: Salen, Tin, Butyltin trichloride, Salicylideneimine, Salhexen, Salhepten
Cite this paper: Burl Yearwood, Reaction of an Organotin Halide with Salicylideneimine Ligands, American Journal of Organic Chemistry, Vol. 4 No. 1, 2014, pp. 11-13. doi: 10.5923/j.ajoc.20140401.02.
Article Outline
1. Introduction
- Schiff bases are very useful reagents in organic and organometallic chemistry. The use of Schiff bases to prepare coordination and organometallic compounds has increased rapidly during the past few years [1-4]. Their properties can be tuned by modifying the organic backbone between the nitrogens. The complexes between Schiff bases and metals have been shown to act as catalysts, as biocidal and anti-tumor agents, and as models for biological studies [5-7]. Our research looks at the complexes formed between Group 13, Group 14 metals and Salen type ligands [‘Salen’ is the name that has historically been used to describe the entire class of such ligands possessing various diamino backbones. However, it is also the specific name of the ethyl derivative, Salen = N,N’-ethylene-bis (salicylideneimine)]. In this communication, we look at the reaction between butyltin trichloride and Salhexen(tBu) [Salhen(tBu) = N,N'-(hexylene)bis (3,5-di-tert-butyl) salicylideneimine] and Salhepten(tBu) [Salpen(tBu) = N,N'-(heptylene)bis(3,5-di-tert-butyl)salicylideneimine].
2. Experimental
- Chemicals were of reagent grade. Butyltin trichloride (Aldrich) was used as received. Triethylamine (Aldrich) was dried over molecular sieves. The reagent 3,5-di-tert-butyl-2- hydroxybenzaldehyde was prepared according to the literature [8]. The ligands used in this study were prepared by the condensation reaction between the hydroxybenzaldehyde and the corresponding diamine. Infrared spectra were recorded on a Perkin-Elmer RX1 infrared spectrometer from 4000-200 cm-1 as KBr discs and were calibrated with respect to the 1601 cm-1 band of a polystyrene film. NMR data were obtained on a JEOL GX-400. Carbon, hydrogen, nitrogen, and tin analysis were carried out by Gailbraith Laboratories, Knoxville, TN. Melting points were determined in open capillaries and are uncorrected.
2.1. Preparation of Butyltin Trichloride Salhexen(tBu) and Salhepten(tBu) Complexes
- In a round bottom flask, fitted with a reflux condenser, triethylamine (0.98 ml, 7.04 mmol) was added to a solution of H2Salhexen(tBu) (1.65g, 3.00 mmol) in toluene (40 mL). A solution of butyltin trichloride (0.846g, 3.00 mmol) in 20mL of toluene was then added slowly to the round bottom flask. The solution turned red. After addition of butyltin trichloride, the mixture was refluxed. The suspension was allowed to cool to room temperature, and the precipitated triethylamine hydrochloride was filtered off. The final product was analyzed. The butyltin derivative of H2Salhepten(tBu) was synthesized in a similar procedure to that described above.
3. Results and Discussion
3.1. Results (Bu)Sn(Salhexen(tBu))Cl (1)
- 1 was prepared from 1.65 g (3.0 mmol) of H2Salhexen(tBu) and 0.846 g (3.00 mmol) of BuSnCl3. Yield 81%. M.p. 167-168°C. Red solid.Elemental Analysis: Anal % (Calc %) for C36H54N2O2SnCl2: C, 58.1 (58.7), H, 7.42 (7.39), N, 3.71 (3.80), Sn, 16.9 (16.1).IR Data: 2938(s), 2860(s), 1650(vs), 1610(vs), 1624(vs), 1540(s), 1455(m), 1405(m), 1380(s), 1345(m), 1335(m), 1280(s), 1260(vs), 1235(s), 1200(s), 1162(s), 1240(s), 1200(s), 1165 (m), 1100(s), 1030(s), 975(m), 928(w), 870(w), 795(s), 600(w), 547(m), 490(m), 300(m).Molecular weight (Rast method) = 730 g/mol1H (CDCl3): δ 1.30 (s, 18H, C(CH3)3), 1.50 (s, 18H, C(CH3)3), 2.35 (m, 6H, CH2), 4.10 (m, 4H, NCH2), 7.30 (d, 2H, C6H2), 7.70 (d, 2H, C6H2), 8.41 (s, 2H, N=CH). 13C NMR (CDCl3): δ 31.1 (C(CH3)3), 34.3 (NCH2), 35.1 (NCH2), 52.5 (C(CH3)3), 126.1 (Ph), 126.9 (Ph), 129.1 (Ph), 129.0 (Ph), 138.1 (Ph), 165.1 (N=CH).119Sn NMR (Me4Sn): δ -503.
3.2. Results (Bu)Sn(Salhepten(tBu))Cl (2)
- 2 was prepared from 1.69g (3.0 mmol) of H2Salhepten(tBu) and 0.846g (3.00 mmol) of BuSnCl3. Yield 81%. M.p. 167-168°C. Red solid.Elemental Analysis: Anal % (Calc %) for C37H56N2O2SnCl2: C, 59.6 (59.2), H, 7.49 (7.52), N, 3.69 (3.73), Sn, 15.0 (15.8).IR Data: 2940(s), 2860(s), 1651(vs), 1615(vs), 1650(vs), 1540(s), 1457(m), 1400(m), 1380(s), 1345(m), 1330(m), 1282(s), 1260(vs), 1231(s), 1200(s), 1164(s), 1240(s), 1200(s), 1166 (m), 1103(s), 1031(s), 973(m), 930(w), 871(w), 795(s), 602(w), 545(m), 495(m), 301(m)Molecular weight (Rast method) = 755 g/mol1H (CDCl3): δ 1.32 (s, 18H, C(CH3)3), 1.49 (s, 18H, C(CH3)3), 2.31 (m, 6H, CH2), 4.21 (m, 4H, NCH2), 7.28 (d, 2H, C6H2), 7.65 (d, 2H, C6H2), 8.39 (s, 2H, N=CH). 13C NMR (CDCl3): δ 32.3 (C(CH3)3), 34.8 (NCH2), 35.6 (NCH2), 51.5 (C(CH3)3), 127.0 (Ph), 126.9 (Ph), 130.0 (Ph), 129.5 (Ph), 133.4 (Ph), 165.2 (N=CH).119Sn NMR (Me4Sn): δ -505.
3.3. IR Results and Structure
- The reaction between the organotin trichloride and the Salen type ligand is an elimination reaction. Butane and triethylamine chloride are eliminated. In complexes 1 and 2, tin binds to the nitrogen and the oxygen of the Schiff base, as shown in Figure 1.
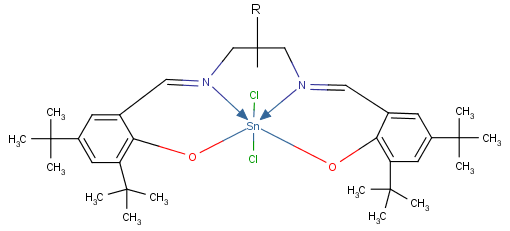 | Figure 1. General structure of Butyltin trichloride Salhexen(tBu) [R = CH2CH2CH2CH2] and Butyltin trichloride Salhepten(tBu) [R = CH2CH2CH2CH2CH2 |
3.4. NMR Results and Structure
- The NMR data suggest that compounds 1 and 2 are monomeric in solution. The methine proton for 1 and 2 were located at δ 8.41 and 8.39 respectively as a single peak. This suggests that the two N=CH protons are equivalent, which would be expected if 1 and 2 have a monomeric solution state structure. The chemical shifts due to the tBu of the ligands are observed as a pair of singlets from δ 1.3-1.5ppm. The presence of singlets also point to a monomeric solution state structure as a dimeric solution state would lead to multiplets.119Sn NMR was also carried out on 1 and 2. Signals were observed at δ -503 and -505 respectively. These chemical shifts are in the range for six-coordinate tin compounds [12]. Further evidence for the structure proposed in Figure 1 is provided by the elemental analysis results which agree with the calculated formula. Molecular weight studies also confirm the proposed structure. Molecular weight studies were carried out using the Rast method, using camphor as the solvent [11]. A value of 730 g/mol was obtained for 1, which compares favorable with the calculated value of 736.4 g/mol. A value of 755 g/mol was obtained for 2, which compares favorable with the calculated value of 750.5 g/mol.
4. Conclusions
- The NMR and IR data results suggest that the tin binds to the nitrogen and oxygen of the salicylideneimine ligands to form a six-coordinate tin complex.
ACKNOWLEDGEMENTS
- The author thanks the Department of Natural Sciences at LaGuardia Community College for use of its facilities for this research.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML