-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 2047-2051
doi:10.5923/j.ajmms.20261604.99
Received: Apr. 2, 2026; Accepted: Apr. 22, 2026; Published: Apr. 25, 2026

Development and Validation of a Prognostic Model for Risk Assessment of Unfavorable Outcomes in Patients with Diabetic Foot Syndrome and Concomitant Ischemic Heart Disease
Zufarov M. M.1, Kamalov S. T.2
1D.Sc., Professor, Department of Vascular Surgery, Republican Specialized Center of Surgery named after Academician V.V. Vakhidov, Tashkent, Uzbekistan
2PhD Candidate, Department of Vascular Surgery, Republican Specialized Center of Surgery named after Academician V.V. Vakhidov, Tashkent, Uzbekistan
Correspondence to: Kamalov S. T., PhD Candidate, Department of Vascular Surgery, Republican Specialized Center of Surgery named after Academician V.V. Vakhidov, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

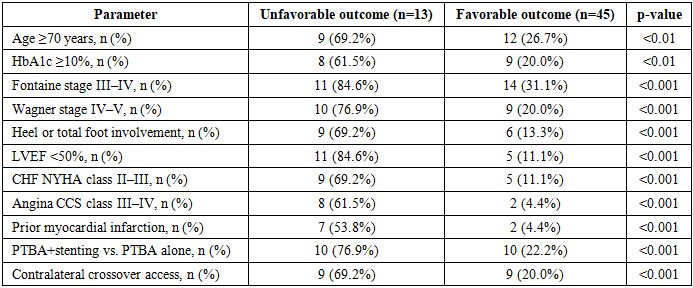
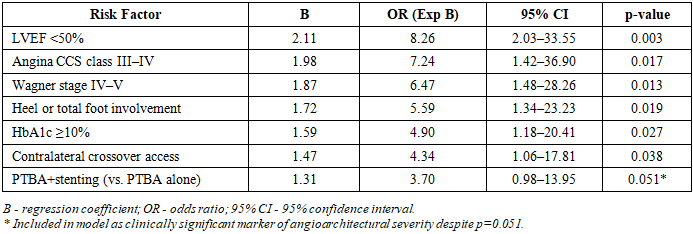
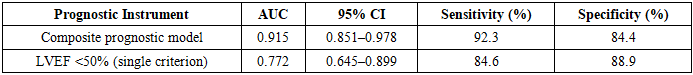
Background: Diabetic foot syndrome (DFS) combined with ischemic heart disease (IHD) represents a high-risk comorbid condition with frequent limb loss and mortality. No validated prognostic instrument integrating cardiac, metabolic, and angioarchitectural parameters exists for pre-interventional risk stratification in this population. Methods: A retrospective study included 58 patients with DFS and IHD who underwent X-ray endovascular interventions (XEI) at the Republican Specialized Center of Surgery named after V.V. Vakhidov (2015–2019). Binary logistic regression identified independent predictors of major amputation or 30-day mortality. Prognostic accuracy was evaluated by ROC analysis. Results: Unfavorable outcomes occurred in 13 patients (22.4%). Independent predictors were: LVEF <50% (OR=8.26; 95% CI: 2.03–33.55; p=0.003), angina CCS class III–IV (OR=7.24; p=0.017), Wagner stage IV–V (OR=6.47; p=0.013), heel or total foot involvement (OR=5.59; p=0.019), HbA1c ≥10% (OR=4.90; p=0.027), and crossover vascular access (OR=4.34; p=0.038). The composite model achieved AUC=0.915 (95% CI: 0.851–0.978), sensitivity 92.3%, specificity 84.4% - significantly superior to LVEF alone (AUC=0.772). Conclusions: The developed multifactorial prognostic model demonstrates high accuracy for pre-interventional risk stratification in patients with DFS and IHD and can guide individualized therapeutic decision-making.
Keywords: Diabetic foot syndrome, Ischemic heart disease, Prognostic model, Logistic regression, ROC analysis, Amputation, Risk stratification, Endovascular intervention
Cite this paper: Zufarov M. M., Kamalov S. T., Development and Validation of a Prognostic Model for Risk Assessment of Unfavorable Outcomes in Patients with Diabetic Foot Syndrome and Concomitant Ischemic Heart Disease, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 2047-2051. doi: 10.5923/j.ajmms.20261604.99.
1. Introduction
- Diabetic foot syndrome (DFS) is one of the most severe complications of diabetes mellitus, determining a high incidence of amputations, disability, and mortality. According to the International Diabetes Federation, up to 25% of patients with diabetes mellitus develop ulcerative-necrotic lesions of the lower extremities during their lifetime, and five-year mortality following amputation exceeds 50%, comparable to oncological diseases [1]. DFS is not only a complication of micro- and macroangiopathy but also a marker of systemic vascular dysfunction, frequently coexisting with ischemic heart disease (IHD), which substantially worsens the clinical course and prognosis.More than 70% of patients with DFS have concomitant IHD, with verified systolic dysfunction (LVEF <50%) detected in one-third of patients, and prior myocardial infarction in every fifth patient [2,3]. Despite the introduction of high-technology X-ray endovascular interventions (XEI), a significant proportion of patients with DFS and IHD continue to experience unfavorable outcomes - major amputation, mortality, repeat interventions, and cardiac decompensation [4]. This is largely attributable to the traditional approach, in which revascularization of the lower extremity is planned without systematic assessment of myocardial reserve or cardiac functional status.Current guidelines emphasize the necessity of integrating systemic vascular assessment and cardiac function into revascularization planning for patients with severe comorbid pathology [5,6]. However, formalized, validated prognostic tools that quantitatively integrate angioarchitectural, cardiological, and metabolic parameters for risk-adapted therapeutic decision-making remain absent. This gap necessitates the development of an evidence-based predictive model capable of identifying patients at high risk of treatment failure prior to XEI in the setting of combined DFS and IHD.
2. Materials and Methods
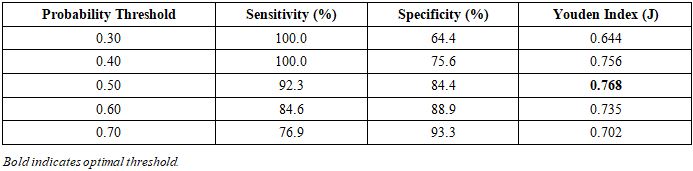
- Study design and patient population. This retrospective observational study was conducted at the Republican Specialized Center of Surgery named after Academician V.V. Vakhidov (Tashkent, Republic of Uzbekistan) between January 2015 and December 2019. The study cohort comprised 58 consecutive patients with confirmed DFS complicated by lower-limb ischemia and documented IHD who underwent XEI on lower extremity arteries under the standard institutional protocol operative at that time, without preliminary cardiac risk stratification.Inclusion criteria: confirmed type 2 diabetes mellitus; clinically and instrumentally verified chronic lower-limb ischemia (Fontaine stage IIb–IV); ulcerative or necrotic foot lesions (Wagner stage I–V); documented IHD based on clinical history, ECG findings, echocardiographic wall motion abnormalities, or prior coronary angiography; complete clinical, laboratory, and angiographic records. Patients with decompensated NYHA class IV heart failure, acute coronary syndrome within 30 days prior to XEI, advanced renal failure requiring dialysis, or incomplete follow-up data were excluded.For the purposes of prognostic model construction, patients were dichotomized by outcome: unfavorable outcome - major amputation (above ankle level) or all-cause mortality within 30 days of XEI (n=13); favorable outcome - all remaining patients with limb preservation, including those requiring minor amputation with retained weight-bearing function (n=45).Clinical and instrumental assessment. Severity of peripheral ischemia was graded using the Fontaine classification. Tissue destruction extent was assessed by the Wagner classification. Cardiac functional status was evaluated by the NYHA functional classification for heart failure and the Canadian Cardiovascular Society (CCS) angina grading. LVEF was measured by transthoracic echocardiography (Simpson biplane method, Philips Affiniti 50, Netherlands). HbA1c was determined by high-performance liquid chromatography (HPLC) on a Bio-Rad D-10 analyzer (USA), calibrated per IFCC/NGSP standards. Digital subtraction angiography (DSA) was performed on an Azurion fluoroscopic system (Philips, Germany). Angiographic parameters analyzed included number of affected arterial segments, anatomical level of involvement, and distal runoff status. Vascular access was categorized as ipsilateral antegrade femoral or contralateral crossover. Procedural strategy was classified as balloon angioplasty alone (PTBA) or combined with stenting (PTBA+stenting).Statistical analysis. Data were analyzed using IBM SPSS Statistics v.26.0 (USA) and Microsoft Excel 2019. Continuous variables are presented as M±SD. Between-group comparisons used the Student's t-test or Mann-Whitney U test; categorical variables were compared by χ² or Fisher's exact test. Statistical significance was set at p<0.05.Binary logistic regression was performed to identify independent predictors of the unfavorable primary outcome. Variables with p<0.10 in univariable analysis were entered into the multivariable model using stepwise forward selection. For each retained predictor, regression coefficient (B), odds ratio (OR) with 95% CI, and p-value are reported. Prognostic model accuracy was assessed by ROC curve analysis with calculation of AUC, sensitivity, specificity, and Youden index (J = sensitivity + specificity − 1). The optimal probability threshold was determined at the maximum Youden index value.
3. Results and Discussion
- Baseline characteristics by outcome. Among 58 patients, unfavorable outcomes (major amputation or 30-day mortality) occurred in 13 (22.4%), while 45 (77.6%) had favorable outcomes. Significant between-group differences were identified across clinical, metabolic, cardiac, and angioarchitectural parameters (Table 1).
|
|
|
|
4. Conclusions
- A multifactorial prognostic model was developed and validated for quantitative assessment of unfavorable outcome risk (major amputation or 30-day mortality) in patients with diabetic foot syndrome and concomitant ischemic heart disease undergoing X-ray endovascular interventions. Six independent predictors were identified: LVEF <50% (OR=8.26), angina CCS class III–IV (OR=7.24), Wagner stage IV–V (OR=6.47), heel or total foot localization (OR=5.59), HbA1c ≥10% (OR=4.90), and contralateral crossover vascular access (OR=4.34).The composite model demonstrated high discriminatory accuracy (AUC=0.915), substantially superior to isolated LVEF assessment (AUC=0.772). At the optimal probability threshold of 0.50, sensitivity reached 92.3% and specificity 84.4%. Defined risk strata (low: P<0.40; intermediate: P=0.40–0.60; high: P>0.60) provide a practical framework for prioritizing either lower-extremity or coronary revascularization in individualized treatment planning.Routine application of this model in pre-procedural assessment may reduce the frequency of unnecessary interventions in high-risk patients, guide sequencing of revascularization strategies, and ultimately contribute to a reduction in amputation rates and perioperative mortality in patients with severe vascular comorbidity.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML