-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 2041-2046
doi:10.5923/j.ajmms.20261604.98
Received: Apr. 8, 2026; Accepted: Apr. 22, 2026; Published: Apr. 25, 2026

Tear Fluid Cytokine Profile as a Prognostic Marker of Keratoplasty Outcomes in Keratoconus
A. F. Yusupov1, M. Kh. Karimova1, S. S. Saidzhanov1, M. I. Inomzhonova1, 2
1Republican Specialized Scientific-Practical Medical Center of Eye Microsurgery, Tashkent, Uzbekistan
2Ophthalmology Clinic "Saif-Optima", Tashkent, Uzbekistan
Correspondence to: A. F. Yusupov, Republican Specialized Scientific-Practical Medical Center of Eye Microsurgery, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Purpose: To evaluate the dynamics of tear fluid cytokine profiles following deep anterior lamellar keratoplasty (DALK) and femtosecond laser-assisted DALK (femto-DALK) in patients with keratoconus, and to develop a cytokine-based prognostic model for predicting functional outcomes. Methods: This prospective, single-center study enrolled 50 patients with stage III–IV keratoconus who underwent DALK (n=28) or femto-DALK (n=22) between January 2023 and December 2025. Twenty healthy volunteers served as controls. Tear fluid concentrations of interleukin-6 (IL-6), interleukin-10 (IL-10), and transforming growth factor-β (TGF-β) were measured by enzyme-linked immunosorbent assay (ELISA) at baseline and at 1, 3, 6, and 12 months postoperatively. Correlations between cytokine levels and best spectacle-corrected visual acuity (BSCVA) were assessed by Pearson and Spearman coefficients. A receiver operating characteristic (ROC) model was constructed to predict favorable outcomes. Results: IL-6 peaked at 1 month in the DALK group (64,8±14,2 pg/mL) and declined to near-normal values by 12 months (18,2±5,4 pg/mL). Femto-DALK produced significantly lower IL-6 levels at all time points (p<0,05). IL-10 peaked at 3 months in both groups, with significantly higher concentrations in the femto-DALK group (p<0,05). No between-group differences were detected for TGF-β (p>0,05). IL-6 at 1 month correlated inversely with BSCVA at 6 months (r=−0,58, p<0,001). An IL-6/IL-10 ratio >5,0 at 1 month predicted unfavorable outcomes with 78% sensitivity and 82% specificity (AUC=0,89; 95% CI: 0,81–0,96). Conclusions: Tear fluid cytokine profiling represents a non-invasive and clinically meaningful tool for early prognostication after keratoplasty. Femtosecond laser-assisted DALK confers a more favorable immunological milieu characterized by attenuated pro-inflammatory and enhanced anti-inflammatory signaling.
Keywords: Keratoconus, Deep anterior lamellar keratoplasty, Cytokines, IL-6, IL-10, TGF-β, Tear fluid, Prognostic markers, Corneal transplantation
Cite this paper: A. F. Yusupov, M. Kh. Karimova, S. S. Saidzhanov, M. I. Inomzhonova, Tear Fluid Cytokine Profile as a Prognostic Marker of Keratoplasty Outcomes in Keratoconus, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 2041-2046. doi: 10.5923/j.ajmms.20261604.98.
Article Outline
1. Introduction
- Keratoconus is a progressive, non-inflammatory ectatic disorder of the cornea characterized by stromal thinning, cone-shaped protrusion, irregular astigmatism, and progressive deterioration of visual acuity. Its reported prevalence ranges from 50 to 230 per 100,000 individuals, with onset typically occurring in the second or third decade of life [1,2]. In advanced stages, when spectacle and contact-lens correction no longer achieves functional vision, surgical intervention becomes necessary.Deep anterior lamellar keratoplasty (DALK) has emerged as the preferred surgical technique for advanced keratoconus. By selectively replacing the anterior stroma while preserving the patient's own Descemet membrane (DM) and endothelium, DALK reduces the risk of endothelial immune rejection and improves long-term graft survival compared with full-thickness penetrating keratoplasty (PK) [3,4]. The introduction of femtosecond laser technology has further refined surgical precision, enabling reproducible lamellar dissections and potentially reducing intraoperative tissue trauma [5,6].Despite technical advances, functional outcomes following DALK remain variable and are influenced by complex immunological processes at the graft–host interface. Local inflammation plays a pivotal role in stromal healing, interface quality, and long-term graft transparency. An imbalance between pro- and anti-inflammatory signaling can culminate in interface haze, delayed visual rehabilitation, or, in rare cases, allograft rejection [7,8].Cytokines are central regulators of the ocular immune response. Interleukin-6 (IL-6), a pleiotropic pro-inflammatory mediator, orchestrates the acute-phase response and activates innate immune pathways following tissue injury. Interleukin-10 (IL-10) is the principal anti-inflammatory cytokine, suppressing pro-inflammatory cascades and promoting immune tolerance. Transforming growth factor-β (TGF-β) governs extracellular matrix remodeling and epithelial–stromal wound healing [9–11].Tear fluid represents an accessible, non-invasive biological matrix that reflects the local immunological environment of the ocular surface. Cytokine concentrations in tears have been correlated with several ocular surface conditions, yet systematic longitudinal data comparing tear cytokine dynamics after conventional versus femtosecond laser-assisted DALK, and their value for outcome prediction, remain scarce [12,13].The objectives of the present study were: (1) to characterize the temporal dynamics of IL-6, IL-10, and TGF-β in tear fluid over 12 months following DALK and femto-DALK; (2) to compare immunological profiles between the two surgical techniques; and (3) to develop a cytokine-based prognostic model for predicting functional outcomes after keratoplasty for keratoconus.
2. Materials and Methods
2.1. Study Design and Participants
- This prospective observational study was conducted between January 2023 and December 2025 at the Republican Specialized Scientific-Practical Medical Center of Eye Microsurgery and the ophthalmology clinic "Saif-Optima," Tashkent, Uzbekistan. The study protocol was approved by the Institutional Review Board, and all participants provided written informed consent in accordance with the principles of the Declaration of Helsinki. Fifty patients (50 eyes) with stage III–IV keratoconus (Amsler–Krumeich classification) were enrolled and allocated by surgical indication to one of two groups: conventional DALK (n=28) or femtosecond laser-assisted DALK (femto-DALK, n=22). A control cohort of 20 healthy volunteers, matched for age and sex, provided single baseline tear samples.Inclusion criteria: (1) clinically and topographically confirmed keratoconus stage III–IV; (2) age 18–45 years; (3) BSCVA ≤0.3 (decimal notation); (4) failure of contact-lens correction; (5) clear endothelium on confocal microscopy; (6) absence of stromal scarring. Exclusion criteria: active anterior-segment inflammation; systemic autoimmune disease; diabetes mellitus; prior corneal surgery; severe dry-eye disease (Schirmer I ≤5 mm/5 min); pregnancy or lactation; systemic immunosuppressive therapy within 6 months.
2.2. Surgical Techniques
- DALK. All procedures were performed under general anesthesia by a single experienced surgeon. Recipient and donor corneas were trephined mechanically to 80–90% stromal depth. The "big-bubble" technique (Anwar–Teichmann) was used to achieve pre-Descemet's plane dissection [14]. After successful DM baring, the residual stroma was excised, and the donor lenticule was secured with a 10-0 nylon running suture.Femto-DALK. A femtosecond laser (wavelength 1053 nm; repetition rate 500 kHz; pulse energy 1,2–1,5 μJ; spot separation 5 μm) was used to create a circular lamellar incision in the recipient cornea. Manual dissection was subsequently performed to complete the pre-Descemet's cleavage using the big-bubble technique, followed by donor lenticule fixation identical to the conventional approach.
2.3. Tear Fluid Collection and Cytokine Analysis
- Tear samples (5–10 μL) were collected from the lateral conjunctival fornix using sterile glass microcapillary tubes between 09:00 and 11:00 h, without topical anesthesia or artificial stimulation, at baseline (1–2 days preoperatively) and at 1, 3, 6, and 12 months postoperatively. Samples were immediately transferred to 1.5-mL polypropylene tubes and stored at −80°C until analysis.Concentrations of IL-6, IL-10, and TGF-β were quantified by sandwich ELISA using validated commercial kits (R&D Systems, Minneapolis, MN, USA) according to the manufacturer’s instructions. Optical absorbance was measured at 450 nm (reference wavelength 540 nm) on a microplate spectrophotometer. Lower limits of detection were 0,70, 3.90, and 31,2 pg/mL for IL-6, IL-10, and TGF-β, respectively. All samples were analyzed in duplicate; intra-assay coefficient of variation did not exceed 5%.
2.4. Ophthalmological Assessment
- Comprehensive ophthalmic evaluation included: BSCVA measurement using ETDRS charts; slit-lamp biomicroscopy with photodocumentation; anterior-segment optical coherence tomography (AS-OCT) for graft–host interface assessment; corneal topography and keratometry; and in vivo confocal microscopy. Examinations were performed at baseline and at 1, 3, 6, and 12 months postoperatively. Interface haze was graded on a 4-point scale: 0 = absent; 1 = trace (no visual impact); 2 = mild (minimal visual impact); 3 = significant (clinically relevant visual impairment).
2.5. Postoperative Management
- All patients received a standardized postoperative regimen: topical dexamethasone 0,1% four times daily for one month, tapering to once daily by six months; topical ofloxacin 0,3% four times daily for two weeks; and preservative-free hyaluronic acid lubricating drops four to six times daily throughout the follow-up period. Sutures were removed 12–18 months postoperatively according to individual clinical parameters.
2.6. Statistical Analysis
- Statistical analyses were performed using IBM SPSS version 26.0 (IBM Corporation, Armonk, NY, USA). Normality was assessed by the Shapiro–Wilk test. Continuous data are expressed as mean ± standard deviation (M±SD) or median (interquartile range) as appropriate. Between-group comparisons were made using the independent-samples t-test or Mann–Whitney U test; within-group temporal changes were assessed by the paired t-test or Wilcoxon signed-rank test. Categorical variables were compared by Pearson’s chi-squared test or Fisher’s exact test.Bivariate correlations between cytokine levels and clinical parameters were quantified by Pearson’s r or Spearman’s ρ depending on data distribution. Correlation strength was classified as weak (|ρ| <0,3), moderate (0,3–0,7), or strong (>0,7). A logistic regression model incorporating the cytokine composite score was developed, and its discriminative ability was evaluated by ROC analysis with area under the curve (AUC), sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV). The optimal cut-off was determined by the Youden index. A two-tailed p-value <0,05 was considered statistically significant.
3. Results
3.1. Baseline Characteristics
- The two surgical groups were well matched at baseline. Mean age was 28,4±6,2 years in the DALK group and 27,8±5,8 years in the femto-DALK group (p=0,72). The male-to-female ratio was 1,4:1 in both groups. Mean BSCVA was 0,18±0,06 and 0,19±0,05, respectively (p=0,54). No statistically significant differences were observed in keratoconus stage, keratometric indices, or central corneal thickness between groups (all p>0,05).
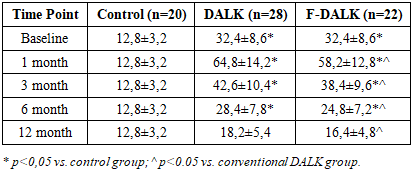
3.2. Dynamics of Pro-Inflammatory Cytokine IL-6
- Baseline tear IL-6 levels in keratoconus patients (32,4±8,6 pg/mL in both surgical groups) were significantly elevated compared with healthy controls (12,8±3,2 pg/mL; p<0,001), consistent with subclinical stromal inflammation intrinsic to keratoconus pathophysiology. Following conventional DALK, IL-6 peaked at 1 month (64,8±14,2 pg/mL), reflecting the acute-phase response to surgical trauma and allograft exposure, and declined progressively to 18,2±5,4 pg/mL by 12 months. In the femto-DALK group, IL-6 concentrations followed a parallel trajectory but were significantly lower at every assessment point (Table 1). The inter-group difference was most pronounced at 1 month (58,2±12,8 vs. 64,8±14,2 pg/mL; p=0,038) and persisted through 12 months (16,4±4,8 vs. 18,2±5,4 pg/mL; p=0,041.
|
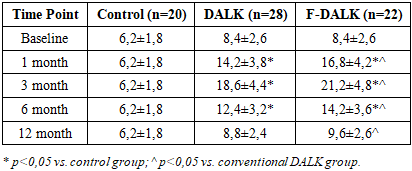
3.3. Dynamics of Anti-Inflammatory Cytokine IL-10
- Preoperative IL-10 levels were modestly elevated in keratoconus patients relative to controls (8,4±2,6 vs. 6,2±1,8 pg/mL; p=0,012). After DALK, IL-10 rose incrementally to peak at 3 months (18,6±4,4 pg/mL), representing the apex of regulatory immune counterbalancing, before declining toward baseline by 12 months (8,8±2,4 pg/mL). Femto-DALK was associated with significantly higher IL-10 concentrations at all time points (peak: 21,2±4,8 pg/mL at 3 months; p=0,018 vs. DALK), suggesting more vigorous engagement of anti-inflammatory pathways (Table 2).
|
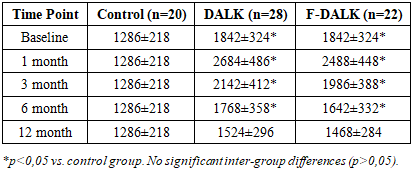
3.4. Dynamics of TGF-β
- Baseline TGF-β was elevated in keratoconus eyes versus controls (1842±324 vs. 1286±218 pg/mL; p<0.001), reflecting active stromal remodeling. Both groups exhibited a sharp postoperative rise at 1 month (DALK: 2684±486 pg/mL; femto-DALK: 2488±448 pg/mL), with gradual normalization over 12 months. No statistically significant difference in TGF-β was observed between the two surgical groups at any time point (all p>0,05), indicating that matrix remodeling proceeds similarly regardless of dissection modality (Table 3).
|
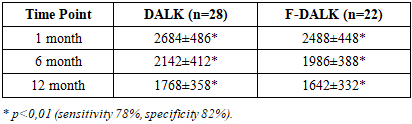
3.5. IL-6/IL-10 Ratio and Clinical Correlations
- The IL-6/IL-10 ratio was adopted as a composite index of the pro- to anti-inflammatory balance. At 1 month, this ratio was significantly higher in the DALK group (4,56±0,84) than in the femto-DALK group (3,46±0,62; p=0,008) and remained more favorable in the femto-DALK group throughout follow-up (Table 4). A ratio >5,0 at 1 month was associated with a relative risk of 3,8 (95% CI: 1,6–8,9; p=0,002) for clinically significant interface haze and delayed visual rehabilitation.
|
3.6. Prognostic Model
- Based on logistic regression and ROC analysis, a three-variable cytokine model (IL-6, IL-10, and IL-6/IL-10 ratio at 1 month) demonstrated excellent discriminative performance: AUC=0,89 (95% CI: 0,81–0,96), sensitivity 78%, specificity 82%, PPV 85%, NPV 92%. Patients classified as having a favorable immunological profile (IL-6 <60 pg/mL, IL-10 >15 pg/mL, ratio <4,0; n=43) achieved BSCVA ≥0,8 in 87,5% and 96,9% of cases at 6 and 12 months, respectively. Conversely, those with an unfavorable profile (IL-6 >70 pg/mL, IL-10 <12 pg/mL, ratio >5,5; n=7) reached this threshold in only 37,5% and 62,5% of cases (p<0,001) (Table 5).
|
4. Discussion
- The present study provides prospective longitudinal evidence that the tear fluid cytokine milieu following DALK is both surgically modifiable and clinically informative. Three principal findings emerged: (1) femtosecond laser-assisted dissection yields a significantly attenuated pro-inflammatory cytokine response compared with mechanical trephination; (2) early IL-6 concentration and the IL-6/IL-10 ratio are moderate-to-strong predictors of functional outcomes; and (3) a composite cytokine model achieves high prognostic accuracy (AUC=0.89) that may be applied clinically within one month of surgery.The markedly elevated baseline IL-6 levels in keratoconus patients relative to healthy controls (32,4 vs. 12,8 pg/mL) align with recent proteomic studies documenting subclinical inflammatory dysregulation intrinsic to the ectatic cornea, attributable to keratocyte apoptosis, altered epithelial turnover, and matrix metalloproteinase activation [15,16]. This pre-existing inflammatory state may prime the ocular surface for an amplified postoperative response.The significantly lower IL-6 levels observed in the femto-DALK group across all postoperative time points support the mechanistic hypothesis that femtosecond laser dissection minimizes cellular damage and inflammatory mediator release through its non-mechanical, photodisruptive tissue separation mechanism. Femtosecond laser-tissue interaction generates plasma-mediated photodisruption within a spatially confined optical breakdown zone, resulting in more predictable tissue planes and reduced collateral stromal trauma compared with manual mechanical dissection [5,6]. Consequently, fewer damage-associated molecular patterns (DAMPs) are liberated, attenuating Toll-like receptor signaling and downstream NF-κB-mediated IL-6 transcription.The higher IL-10 concentrations in the femto-DALK group reflect more robust activation of counter-regulatory immune circuits. IL-10 is produced principally by regulatory T cells (Tregs), alternatively activated macrophages, and corneal epithelial cells in response to limited antigen exposure [17]. By suppressing antigen-presenting cell function and inhibiting IL-1β, TNF-α, and IL-6 production, IL-10 creates an immunologically privileged environment conducive to immune tolerance and long-term graft acceptance. The observed positive correlation between early IL-10 levels and visual rehabilitation rate (r=+0,52, p<0,001) underscores the functional importance of this regulatory arm.The absence of between-group differences in TGF-β dynamics suggests that keratocyte-to-myofibroblast transdifferentiation and extracellular matrix remodeling proceed through a conserved pathway independent of dissection modality. TGF-β-driven stromal healing is essential for interface consolidation, but excessive or prolonged TGF-β signaling may paradoxically promote fibrosis and interface opacification. In our cohort, TGF-β normalized by 12 months in both groups, indicating resolution of the active repair phase without pathological fibrotic sequelae.The IL-6/IL-10 ratio emerged as a particularly powerful composite biomarker. Its superiority over individual cytokine levels as a predictive tool is conceptually consistent with the "immunological tipping point" model, wherein functional outcomes are determined not by the absolute magnitude of inflammation but by the balance between destructive and regulatory forces. A ratio >5,0 at 1 month predicted unfavorable outcomes with sensitivity and specificity comparable to established biomarkers in other transplant specialties (78% and 82%, respectively).The prognostic model developed here (AUC=0.89) compares favorably with previously reported predictors of keratoplasty outcomes, including topographic indices and endothelial cell density measures. Its principal clinical advantage lies in its early availability: meaningful stratification can be achieved one month postoperatively, a time window during which modification of topical corticosteroid dosing and immunosuppressive adjuncts is most likely to influence the immunological trajectory. Patients identified as high-risk (n=7; 14%) could be considered for intensified anti-inflammatory regimens, earlier follow-up intervals, or adjunctive therapies such as subconjunctival triamcinolone or low-dose systemic mycophenolate mofetil [18].Certain limitations of this study warrant acknowledgment. The sample size, while adequate for detecting moderate-to-large effect sizes, limits the power to detect interactions in subgroup analyses. Allocation to surgical technique was not randomized but was based on clinical and logistical considerations, introducing potential selection bias. Tear cytokine quantification is susceptible to variation in collection technique, storage conditions, and sample volume; however, standardized collection protocols and duplicate ELISA measurements were employed to minimize these sources of error. Finally, the 12-month follow-up may not capture late immunological events relevant to graft survival beyond the first year. Future multi-center, randomized studies with extended follow-up and expanded cytokine panels (including IL-1β, TNF-α, VEGF, and CXCL10) are warranted to validate and refine the prognostic model.
5. Conclusions
- Tear fluid cytokine profiling provides a non-invasive, early, and clinically meaningful window into the immunological processes governing graft integration after DALK. The principal conclusions are as follows:1. DALK for keratoconus induces a predictable postoperative cytokine trajectory characterized by a pro-inflammatory peak at 1 month, an anti-inflammatory apex at 3 months, and near-normalization by 12 months for both IL-6 and IL-10.2. Femtosecond laser-assisted DALK consistently produces a more favorable immunological profile than conventional mechanical DALK, manifested by lower IL-6 levels (p<0,05 at all time points) and higher IL-10 concentrations (p<0,05 at all time points), attributable to reduced intraoperative tissue trauma.3. Tear IL-6 at 1 month inversely predicts BSCVA at 6 months (r=−0,58, p<0,001), identifying patients with IL-6 >70 pg/mL as high-risk for delayed visual rehabilitation.4. The IL-6/IL-10 ratio is a robust composite prognostic index. A ratio >5,0 at 1 month predicts unfavorable functional outcomes (sensitivity 78%, specificity 82%, RR 3,8) and is significantly lower in femto-DALK patients at all time points (p<0,01).5. The three-variable cytokine prognostic model (IL-6, IL-10, IL-6/IL-10 ratio at 1 month) achieves an AUC of 0,89, enabling early identification of patients at risk for suboptimal outcomes and facilitating personalized postoperative management.
ACKNOWLEDGEMENTS
- The authors gratefully acknowledge the support of the clinical and laboratory staff of the Republican Specialized Scientific-Practical Medical Center of Eye Microsurgery and the ophthalmology clinic "Saif-Optima," Tashkent, Uzbekistan.
DISCLOSURE
- The authors declare no conflict of interest. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. All procedures adhered to the ethical standards of the institutional review board and to the 1964 Declaration of Helsinki and its later amendments.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML