-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 2037-2040
doi:10.5923/j.ajmms.20261604.97
Received: Apr. 5, 2026; Accepted: Apr. 20, 2026; Published: Apr. 25, 2026

Functional State of the Kidneys in Paracetamol-Induced Hepatitis Under Conditions of Water Deficit
Safarova Sayyora Rustam qizi1, Yariyev Alisher Alijonovich2
1Researcher, Republican Scientific Center for Emergency Medical Care, Syrdarya Branch, Uzbekistan
2DSc., Head of the Surgical Department, Termez Branch of Tashkent State Medical University, Uzbekistan
Correspondence to: Safarova Sayyora Rustam qizi, Researcher, Republican Scientific Center for Emergency Medical Care, Syrdarya Branch, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

In an experimental study, the effects of dehydration and paracetamol on the functional state of the kidneys in laboratory rats were investigated. It was established that water restriction was accompanied by a marked decrease in diuresis, an increase in urine specific gravity, and the development of proteinuria. Administration of paracetamol under conditions of preserved hydration did not cause significant changes in the parameters of the general urinalysis. However, when paracetamol administration was combined with dehydration, signs of renal dysfunction were observed, including proteinuria and hematuria. The obtained results indicate the leading role of water deficit in the development of renal functional disorders.
Keywords: Paracetamol, Dehydration, Diuresis, Proteinuria, Hematuria, Renal functional status, Toxic hepatitis, Experimental study
Cite this paper: Safarova Sayyora Rustam qizi, Yariyev Alisher Alijonovich, Functional State of the Kidneys in Paracetamol-Induced Hepatitis Under Conditions of Water Deficit, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 2037-2040. doi: 10.5923/j.ajmms.20261604.97.
1. Introduction
- Drug-induced liver injury remains one of the most significant problems in modern hepatology and toxicology. One of the most commonly used drugs capable of causing toxic liver damage when therapeutic doses are exceeded is paracetamol. It is well known that its metabolism is accompanied by the formation of a highly reactive metabolite, N-acetyl-p-benzoquinone imine (NAPQI), which, under conditions of glutathione depletion, initiates oxidative stress, mitochondrial dysfunction, and necrotic damage to hepatocytes [6].Along with the hepatotoxic effects of paracetamol, increasing attention has recently been paid to its influence on renal functional status. Experimental and clinical studies indicate that paracetamol intoxication may lead to the development of acute kidney injury accompanied by increased serum creatinine and urea levels, impaired glomerular filtration, and damage to the proximal tubular epithelium [7]. It has been established that the nephrotoxic effect of the drug is associated both with the formation of reactive metabolites and with the activation of lipid peroxidation processes and inflammatory reactions in renal tissue [5].An additional factor capable of enhancing the toxic effects of paracetamol on detoxification organs is dehydration. Water deficit leads to hemoconcentration, decreased renal blood flow, and impaired microcirculation, which in turn contributes to deterioration of the renal filtration function and increases tissue sensitivity to toxic agents. Experimental models have demonstrated that toxic exposure to paracetamol is accompanied by pronounced morphofunctional changes in the kidneys, including damage to the tubular apparatus and impairment of antioxidant defense mechanisms [9].Despite the considerable number of studies devoted to toxic liver injury associated with paracetamol use, the influence of dehydration on renal functional parameters under conditions of paracetamol-induced toxic hepatitis remains insufficiently investigated. In this regard, studying changes in renal function under the combined effects of dehydration and paracetamol is of important scientific and practical interest, as it will deepen the understanding of the mechanisms of systemic toxic injury and contribute to improving strategies for the prevention and correction of these disorders [3].Purpose of the researchAim of the study is to investigate changes in diuresis and urinalysis parameters in toxic liver injury under conditions of dehydration.
2. Materials and Methods
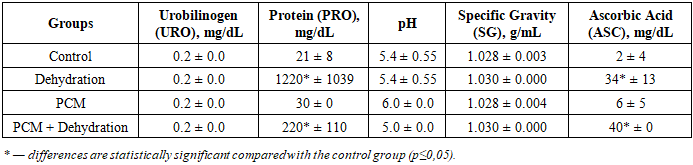
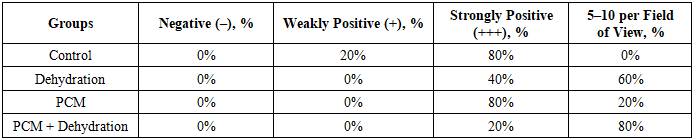
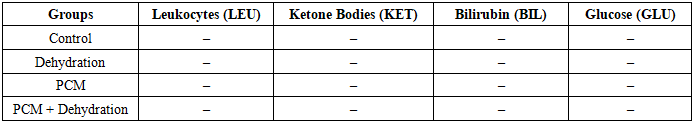
- The study was conducted on laboratory white rats weighing 250–290 g, maintained under standard vivarium conditions: in metal cages at a temperature of 23 ± 1 °C, relative humidity of 30–70%, and a 12/12 h light–dark cycle, with free access to standard laboratory feed in accordance with GLP requirements [1,2]. The animals were randomized into experimental groups consisting of six rats each.A model of paracetamol-induced toxic hepatitis was reproduced by intraperitoneal administration of paracetamol in increasing doses of 200, 300, and 600 mg/kg body weight for three consecutive days [4]. The control group received an equivalent volume of solvent according to the same protocol. Dehydration in the corresponding groups was initiated on the first day of the experiment and continued for three days, allowing the evaluation of the effect of water deficit on the severity of toxic liver injury. The experimental animals were divided into the following groups:1. Intact group — animals were maintained under standard conditions without paracetamol administration or dehydration modeling.2. Dehydration group (control-1) — animals were subjected to dehydration without paracetamol administration.3. Paracetamol group (PCM, control-2) — animals received paracetamol at the indicated doses without restriction of the water regimen.4. Paracetamol + dehydration group (experimental group) — animals received paracetamol in combination with experimentally induced dehydration.Immediately after the final administration of paracetamol, animals from all experimental groups were placed in metabolic cages for 24 hours to collect daily urine samples. At the end of this period, on the fourth day of the experiment, urinalysis was performed, after which blood samples were collected for subsequent laboratory analysis.The functional state of the kidneys in experimental animals was assessed based on daily diuresis and urinalysis parameters. For this purpose, animals from all groups were placed in metabolic cages for 24 hours after the final administration of the drug, which ensured separate and quantitative urine collection. The total volume of urine excreted within 24 hours was measured using graduated cylinders and expressed in milliliters, after which the mean values were calculated for each experimental group.The collected daily urine samples were used to perform general urinalysis. The analysis was carried out using multiparameter diagnostic test strips U-11 Urinalysis Reagent Strips (Mindray). The method is based on the colorimetric assessment of changes in the color of the reaction zones of the test strip after immersion in the urine sample. The results were interpreted in accordance with the manufacturer’s instructions by visually comparing the color changes with the provided reference color scale.Using the test strips, the following parameters were determined: leukocytes, nitrites, urobilinogen, protein, pH, blood (erythrocytes/hemoglobin), specific gravity, ketone bodies, bilirubin, glucose, and ascorbic acid. All parameters were recorded semi-quantitatively with subsequent comparative analysis between the experimental groups.A comprehensive evaluation of daily diuresis and urinalysis parameters made it possible to characterize renal functional status, the degree of impairment of the concentrating function and water–electrolyte balance, as well as to identify possible signs of paracetamol-induced nephrotoxic effects under dehydration conditions. Statistical analysis of the obtained results was performed using analysis of variance (ANOVA) with a significance level of p = 0.05 using GraphPad Prism software version 8.0.0 for Windows (GraphPad Software, San Diego, CA, USA; www.graphpad.com) [8].
3. Results and Discussion
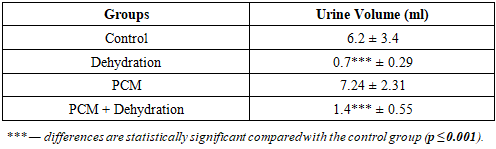
- The results of the analysis of the total urine volume (Table 1) demonstrated a pronounced dependence of diuresis on the water intake conditions of the animals. In the control group, the daily urine volume was 6.2 ± 3.4 ml. In the dehydration group, a sharp and statistically significant decrease in diuresis was observed, reaching 0.7 ± 0.29 ml (p ≤ 0.001 compared with the control).
|
|
|
|
4. Conclusions
- 1. Dehydration leads to a pronounced decrease in daily diuresis and the development of proteinuria, indicating impairment of the renal concentrating function.2. Administration of paracetamol under preserved hydration conditions does not cause significant changes in diuresis or in the main parameters of urinalysis.3. The combination of paracetamol with dehydration is accompanied by reduced diuresis and the development of proteinuria and hematuria, indicating an enhancement of nephrotoxic manifestations under conditions of water deficit.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML