-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 2026-2030
doi:10.5923/j.ajmms.20261604.95
Received: Mar. 25, 2026; Accepted: Apr. 16, 2026; Published: Apr. 25, 2026

Thyroid Dysfunction in Patients Receiving Combined Therapy for Breast Cancer: A Prospective Clinical Study
Nasirova K. Khurshida1, Yusupbekov A. Abrorjon1, 2, Azimova A. Makhbuba1
1Tashkent State Medical University, Uzbekistan
2Republic Specialized Scientific Practical Medical Center of Oncology and Radiology, Uzbekistan
Correspondence to: Nasirova K. Khurshida, Tashkent State Medical University, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

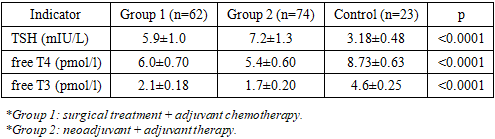
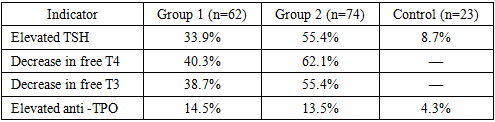
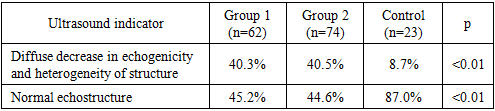
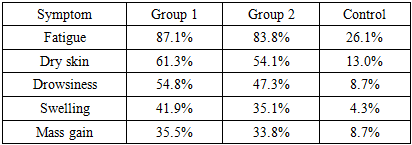
Thyroid dysfunction is an increasingly recognized manifestation of endocrine toxicity associated with systemic antitumor therapy in breast cancer; however, its clinical significance during combined treatment remains insufficiently characterized. This prospective study aimed to evaluate functional and structural changes in the thyroid gland in patients with breast cancer undergoing multimodal therapy. A total of 136 patients with stage II–III disease were enrolled and stratified into two groups: surgery followed by adjuvant chemotherapy (n=62) and neoadjuvant chemotherapy followed by surgery and adjuvant treatment (n=74). A control group included 23 patients with stage I disease. Thyroid function was assessed by serum TSH, free T4, free T3, and anti-thyroid peroxidase antibodies, while structural alterations were evaluated using ultrasound imaging. Combined therapy was associated with a significant increase in TSH levels and a concomitant decrease in free T4 and free T3 concentrations compared with controls (p<0.0001), indicating a predominance of hypothyroid states. The prevalence of laboratory-confirmed hypothyroidism exceeded 60%, with more pronounced hormonal disturbances observed in patients receiving neoadjuvant regimens, suggesting a cumulative treatment effect. Ultrasound examination revealed structural abnormalities, including reduced echogenicity and parenchymal heterogeneity, in approximately 40% of treated patients, significantly exceeding control values (p<0.01). Clinical manifestations consistent with hypothyroidism were frequently observed but demonstrated limited correlation with biochemical parameters. These findings indicate that thyroid dysfunction represents a common and clinically relevant consequence of combined breast cancer therapy, characterized by both functional impairment and structural alterations. Routine thyroid monitoring should be incorporated into standard oncological care to enable early detection and management of endocrine toxicity.
Keywords: Breast cancer, Thyroid dysfunction, Hypothyroidism, Chemotherapy, Endocrine toxicity, Neoadjuvant therapy, Thyroid ultrasound, TSH, Free T4, Free T3, Autoimmune thyroiditis
Cite this paper: Nasirova K. Khurshida, Yusupbekov A. Abrorjon, Azimova A. Makhbuba, Thyroid Dysfunction in Patients Receiving Combined Therapy for Breast Cancer: A Prospective Clinical Study, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 2026-2030. doi: 10.5923/j.ajmms.20261604.95.
1. Introduction
- Breast cancer is the leading cause of cancer incidence among women worldwide and remains the leading cause of cancer-related mortality [1,2]. According to international databases, more than 2.3 million new cases are registered annually, with the upward trend continuing [1]. In Central Asian countries, including the Republic of Uzbekistan, a steady increase in incidence rates and decrease of patient age are being observed [3].Modern advances in molecular diagnostics, targeted therapy, and hormonal therapy have significantly improved overall and disease-free survival rates [4,5]. Combined treatment regimens, including surgery, neoadjuvant and adjuvant chemotherapy, radiation therapy, and endocrine therapy, are the standard for managing patients with stages II–III of the disease [6,7]. However, intensification of therapy is accompanied by an increase in the incidence of systemic complications, which requires expanding the range of monitoring for concomitant disorders [8].In recent years, increasing attention has been paid to the problem of endocrine toxicity of antitumor treatment [9,10]. The thyroid gland is a target organ sensitive to the effects of cytostatic drugs, targeted agents, and immunomodulatory therapy [11]. The mechanisms of damage include direct cytotoxic damage to thyrocytes, impaired peripheral conversion of thyroid hormones, vascular changes, and immune reactions [12].Thyroid hormones play a fundamental role in the regulation of energy metabolism, cell proliferation, differentiation, and apoptosis [13]. Impaired secretion of these hormones can lead to decreased tolerance to therapy, deterioration in quality of life, and the development of asthenic syndrome, which is often mistakenly interpreted as a manifestation of chemotherapeutic toxicity [14].According to various authors, the incidence of subclinical and overt hypothyroidism in cancer patients varies from 30 to 60% depending on the treatment regimens used [9,15,16]. A particularly high risk of developing thyroid dysfunction has been described with the use of targeted drugs and combination therapy regimens [12,17]. However, data on the state of the thyroid gland in patients with breast cancer under neoadjuvant and adjuvant chemotherapy remain limited and contradictory [18,19].In addition to functional changes, structural abnormalities of the thyroid gland detected by ultrasound examination are of great importance, including decreased echogenicity, heterogeneity of the parenchyma, and signs of autoimmune inflammation [19,20,21]. A comprehensive assessment of hormonal and ultrasound parameters allows for a more accurate determination of the nature of the lesion and the degree of its severity [22,23].Thus, the study of the functional and structural state of the thyroid gland in patients with breast cancer receiving combination treatment is an urgent scientific and clinical task.Purpose of the studyTo evaluate functional and structural changes in the thyroid gland in patients with breast cancer during combination treatment.
2. Materials and Methods
- The study was conducted in a prospective format at a specialized oncology and radiology center from November 2023 to January 2025.The study included 136 patients aged 38 to 68 years (mean age 52.4±6.8 years) with morphologically confirmed stage II–III breast cancer.The control group consisted of 23 patients (mean age 50.9±5.7 years) with stage I disease who did not receive aggressive combined therapy.The patients in the main cohort were divided into two groups:The first group (n=62) consisted of surgical treatment followed by adjuvant chemotherapy. The second group (n=74) consisted of neoadjuvant chemotherapy followed by surgery and adjuvant therapy.Inclusion criteria: 1. Age over 18 years; 2. Histologically confirmed diagnosis; 3. No previously diagnosed thyroid diseases; 4. Written informed consent.Exclusion criteria: 1. Previous thyroid surgery; 2. Taking thyroid medications before treatment; 3. The presence of severe concomitant endocrine pathology.Thyroid function was assessed by measuring levels of thyroid-stimulating hormone (TSH), free thyroxine (fT4), free triiodothyronine (fT3), and thyroid peroxidase antibodies (anti-TPO). The studies were performed using enzyme-linked immunosorbent assay.The structural condition was assessed using ultrasound examination to determine the volume of the gland, echogenicity, homogeneity of the parenchyma, the presence of nodular formations and signs of an autoimmune process.Statistical analysis was performed using SPSS 16.0. Quantitative indicators are presented as mean ±SD. Student's t-test was used to compare groups. Differences were considered statistically significant at p < 0.05.
3. Results
|
|
|
|
4. Discussion
- The results obtained indicate the development of persistent thyroid dysfunction in patients undergoing combined treatment for breast cancer. The identified changes are systemic and are confirmed by a combination of laboratory, ultrasound, and clinical data.Elevated TSH levels combined with decreased free thyroxine and triiodothyronine levels suggest the prevalence of primary hypothyroidism. However, more pronounced hormonal changes in the second group suggest the possible role of cumulative effects of cytostatic drugs. Sequential use of neoadjuvant and adjuvant therapy likely increases the burden on the pituitary-thyroid system, contributing to the exhaustion of compensatory regulatory mechanisms.The absence of significant differences in the frequency of ultrasound changes between the first and second groups suggests that structural abnormalities develop early in the course of systemic therapy and persist regardless of the stage of treatment. Decreased echogenicity and parenchymal heterogeneity may reflect both degenerative processes and subclinical inflammation of the glandular tissue.The immunological component, represented by a moderate increase in anti -TPO, is not dominant, but its presence in some patients allows us to consider the immunomodulatory effect of antitumor therapy as one of the possible pathogenetic mechanisms.Clinical symptoms largely correspond to laboratory findings, but the severity of subjective manifestations does not always directly correlate with hormonal levels. This highlights the complexity of interpreting asthenic and metabolic syndromes in cancer patients, where symptoms can be multifactorial.Key points1. In patients with breast cancer receiving combination therapy, according to reliable data, thyroid dysfunction develops, characterized by an increase in TSH and a decrease in free T4 and free T3.2. The frequency of laboratory signs of hypothyroidism exceeds 60%, indicating a high prevalence of endocrine toxicity during systemic antitumor therapy.3. The use of neoadjuvant regimens is associated with more pronounced hormonal changes, which indicates a possible cumulative and dose-dependent effect of the cytostatic effect.4. Ultrasound signs of structural changes in the thyroid gland are detected in a significant proportion of patients and confirm the morphological basis of functional disorders.5. Clinical manifestations of hypothyroidism are recorded significantly more frequently in the combination treatment groups, but their severity does not always correspond to the degree of laboratory abnormalities.6. It is advisable to include mandatory monitoring of thyroid status and ultrasound assessment of the thyroid gland in the algorithm for dynamic observation of patients with breast cancer.7. Further research should be aimed at studying the long-term consequences of thyroid dysfunction and its impact on therapy tolerability and oncological prognosis.
5. Conclusions
- The study results confirm that thyroid dysfunction is a common manifestation of endocrine toxicity associated with combination therapy for breast cancer. The findings expand our understanding of the nature and prevalence of these disorders and highlight the need for their timely diagnosis.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML