-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 2010-2014
doi:10.5923/j.ajmms.20261604.91
Received: Mar. 11, 2026; Accepted: Apr. 6, 2026; Published: Apr. 25, 2026

Radiation-Induced Microbial Translocation and Colonization in Internal Organs: An Experimental Study
Murotov Nurshod Farxodovich
Bukhara State Medical Institute, Uzbekistan
Correspondence to: Murotov Nurshod Farxodovich, Bukhara State Medical Institute, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Bacterial translocation (BT), defined as the movement of viable microorganisms from the intestinal lumen to normally sterile internal organs, plays a significant role in the pathogenesis of systemic inflammatory disorders, including sepsis and multiple organ dysfunction. The present study was designed to experimentally investigate the capacity of microorganisms to proliferate after migrating from the large intestine to internal organs in white outbred rats subjected to acute radiation exposure. In addition, the effectiveness of preventive biocorrection interventions was evaluated. The results revealed a substantial increase in microbial proliferation (PMG) in the mesenteric lymph nodes, liver, lungs, and peripheral blood of irradiated animals. The highest level of PMG (100%) was detected in the mesenteric lymph nodes on the ninth day after irradiation. Conversely, irradiated rats receiving a biopreparation demonstrated a progressive reduction in PMG during the observation period. In non-irradiated animals, PMG was not detected in the spleen or lungs; however, irradiated rats showed significant microbial proliferation in these organs, which markedly decreased following biocorrective treatment. Overall, the findings indicate that acute radiation exposure intensifies bacterial translocation, whereas the application of biocorrection strategies significantly limits microbial germination and reduces the extent of translocation across multiple internal organs.
Keywords: Bacterial translocation, Acute irradiation, Microbial germination, Biocorrection, Mesenteric lymph nodes, Systemic inflammation
Cite this paper: Murotov Nurshod Farxodovich, Radiation-Induced Microbial Translocation and Colonization in Internal Organs: An Experimental Study, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 2010-2014. doi: 10.5923/j.ajmms.20261604.91.
1. Introduction
- When the balance between indigenous and facultative microorganisms, which are representatives of the normal microbiota in the human large intestine, is disrupted and the permeability of the intestinal mucosa increases, the transfer of viable microorganisms to various internal organs through blood and lymph intensifies. This phenomenon was termed "bacterial translocation" by Berg R. D. [1].There are also scientific sources that associate the phenomenon of bacterial translocation (BT) with the development of systemic inflammatory response syndrome, sepsis, and multiple organ failure [2]. Some authors have proved that the majority of hospital infections are caused by translocated intestinal microorganisms [1-6].When evaluating the methods currently employed by various researchers to study BT, it is possible to categorize them into several main groups [4,8].The aim of the study was to experimentally investigate and evaluate the germination state of microorganisms translocated from the large intestine to internal organs in white outbred rats subjected to acute irradiation.
2. Materials and Methods
- The experimental study included 162 male white outbred rats, each weighing approximately 150–180 grams and aged 3 months. The animals were randomly assigned to three groups: the main group (n=54) – rats subjected to a single exposure to acute total body irradiation at a dose of 5 Gray without prophylactic biocorrection; the comparison group (n=54) – rats that received the same irradiation dose but were pre-treated with prophylactic biocorrection; and the control group (n=54) – intact rats not subjected to irradiation. Each of the three groups was further divided into three subgroups depending on the time of microbiological analysis after irradiation: 120h (n=18), 168h (n=18), and 216h (n=18).The housing, feeding, and handling of animals were carried out in accordance with the methodological guidelines developed by Nuraliev N.A. et al. [2-3]. All experimental procedures complied with ethical standards for laboratory animal care and biosafety regulations [1]. Irradiation was conducted using the AGAT-R1 γ-therapy device (Estonia), which utilizes cobalt-60 (Co-60) as the radiation source. The irradiation procedures took place at the Bukhara branch of the Republican Specialized Scientific and Practical Center for Oncology and Radiology, Republic of Uzbekistan.Prophylactic biocorrection in the comparison group was carried out using the biologically active supplement “Lactopropolis-AWL”, developed by the Institute of Microbiology of the Academy of Sciences of the Republic of Uzbekistan in collaboration with “AllWellLab” LLC. This supplement was administered orally once daily in the morning, with dosage adjusted according to animal body weight. It was given for 20 days prior to irradiation. The formulation of "Lactopropolis-AWL" includes probiotic strains Lactobacillus rhamnosus 925 and Enterococcus durans, along with an extract of biologically active compounds from propolis. The supplement exhibits anti-inflammatory, antimicrobial, and immunomodulatory properties.Microbiological identification of isolates cultured from biological samples was conducted following the protocols described in Bergey’s Manual of Systematic Bacteriology, using various nutrient media, including blood agar, Endo agar, egg yolk-salt agar, Blaurock medium, and SRM-4, all supplied by HiMedia (India). The representatives of normal colonic microbiota (Escherichia spp., Staphylococcus spp., Proteus spp., Enterococcus spp., Bacteroides spp., Lactobacillus spp., Bifidobacterium spp.) were identified at the genus level. Additionally, impression smears of internal organs were prepared for bacteriological analysis. The percentage of microbial germination (PMG) was assessed in accordance with the method described by Nuraliev N.A. and Suvonov K.J. [4].
3. Results
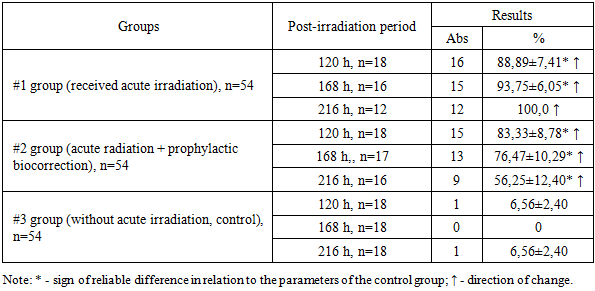
- The proportion of microorganisms migrating from the large intestine to the mesenteric lymph nodes varied between the three groups throughout the observation period. (Table 1).
|
|
|
4. Discussion
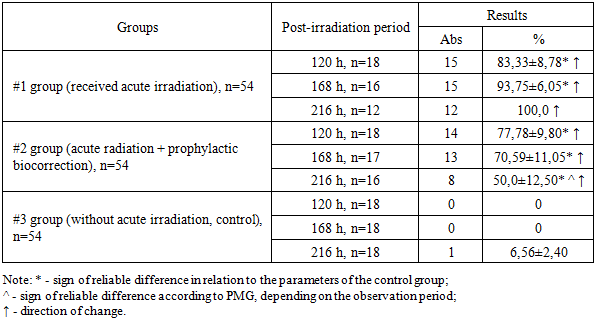
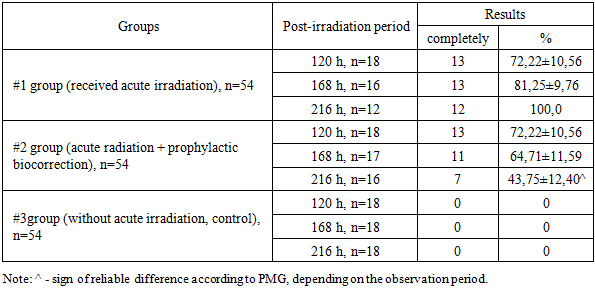
- Throughout the observation period, the PMG (microbial translocation) from the large intestine to the mesenteric lymph nodes in Group 1 increased steadily, reaching a peak of 100.0% by 216 hours after exposure to acute irradiation. In animals that received prophylactic biocorrection, PMG levels were initially high but demonstrated a gradual decline over time, dropping to 56.25% by 216 hours. This was significantly lower—by a factor of 1.78—compared to Group 1 (P<0.001).The liver of irradiated laboratory animals showed a similar trend in microbial translocation, with a marked increase (P<0.001) in PMG levels in both experimental groups relative to the control. Despite the similarity in translocation trends, the intensity of microbial colonization in the liver was lower than in the mesenteric lymph nodes. Notably, the results obtained from the biocorrected animals (Group 2) were inversely related to those of Group 1. Moreover, while no statistically significant differences were found in Group 1 over time, Group 2 exhibited a significant reduction in liver PMG values depending on the duration of observation (P<0.05).In the spleen, PMG values remained at 0% in non-irradiated control animals throughout the observation period. In contrast, acutely irradiated animals showed translocation in 50.0–100.0% of cases. Among the animals subjected to both irradiation and prophylactic biocorrection, PMG levels in the spleen declined progressively with time and decreased by 1.65 times by the end of the 216-hour period (P<0.05).Regarding the lungs, PMG was not detected in non-irradiated animals (0%), whereas in irradiated animals, microbial translocation ranged from 55.56±11.71% to 91.67±7.98%, increasing over the 216-hour observation period. In biocorrected animals, PMG levels in the lungs showed a decreasing trend, although the differences were not statistically significant.In peripheral blood, PMG following acute irradiation increased over time in Group 1. Conversely, in Group 2 animals that received biocorrection, the percentage of microorganism germination decreased with time. In both irradiated groups, the germination levels remained significantly higher than those observed in the control group.
5. Conclusions
- 1. Acute irradiation markedly intensified bacterial translocation from the large intestine to sterile internal organs in white outbred rats. The highest frequency of microbial detection was observed in the mesenteric lymph nodes, where the proportion of animals with translocated microorganisms progressively increased during the observation period and reached 100% at 216 hours after irradiation. This finding confirms that the mesenteric lymphatic system acts as a primary pathway and initial barrier for the spread of intestinal microorganisms during radiation-induced impairment of the intestinal barrier.2. In the liver of irradiated animals, microbial translocation from the large intestine also demonstrated a progressive increase over time. However, the intensity of microbial proliferation in the liver remained lower than that recorded in the mesenteric lymph nodes, indicating that although the liver participates in the systemic dissemination of intestinal microorganisms, its microbial burden develops more slowly compared with lymphatic structures. This observation highlights the role of the mesenteric lymph nodes as the main site of early microbial accumulation following radiation exposure.3. In the spleen and lungs of irradiated rats, translocated microorganisms from the large intestine were detected with frequencies ranging from 37.5±12.10% to 100.0%. The presence of microorganisms in these organs reflects the systemic spread of intestinal bacteria following disruption of the intestinal mucosal barrier caused by radiation. In addition, the detection of microorganisms in peripheral blood confirmed the development of systemic microbial dissemination associated with acute radiation exposure.4. Preventive biocorrection demonstrated a pronounced protective effect against radiation-induced bacterial translocation. Although the initial level of microbial detection in animals receiving prophylactic biocorrection was elevated after irradiation, a gradual and consistent decline in microbial proliferation and translocation was observed throughout the experimental period. This trend was recorded in the mesenteric lymph nodes, liver, spleen, lungs, and peripheral blood.5. Overall, the obtained results indicate that acute irradiation significantly disrupts the intestinal barrier and enhances the migration of viable microorganisms from the large intestine into the lymphatic system, internal organs, and bloodstream. At the same time, the application of prophylactic biocorrection contributes to the reduction of microbial germination and limits the intensity of bacterial dissemination, suggesting its potential value as a preventive approach for controlling radiation-induced microbial translocation and its systemic consequences.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML