-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 1799-1803
doi:10.5923/j.ajmms.20261604.52
Received: Feb. 25, 2026; Accepted: Mar. 20, 2026; Published: Apr. 13, 2026

Predictive Significance of Hemostatic System Markers for Coronary Restenosis
Mekhmanov Feruz Shamsievich1, Naimova Shohida Anvarovna2
1Bukhara Regional Multidisciplinary Medical Center, Head of the Cardiology Department, Independent Researcher, Bukhara State Medical Institute, Uzbekistan
2Bukhara State Medical Institute, Associate Professor, Department of Hematology and Clinical Laboratory Diagnostics, DSc, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

In patients with ischemic heart disease (IHD) who undergo percutaneous coronary intervention (PCI), the risk of coronary artery restenosis may be elevated. This study included 103 patients (48 men and 55 women) aged 18–74 years who underwent coronary intervention and were treated at the Bukhara Regional Multidisciplinary Medical Center during 2021–2024. A control group consisted of 30 healthy volunteers. Clinical assessment, biochemical testing (lipid profile), hemostasis markers (D-dimer, fibrinogen, platelet count, activated partial thromboplastin time, prothrombin time, international normalized ratio), enzyme-linked immunosorbent assay for VEGF, and instrumental examinations (ECG, echocardiography, ultrasonography) were performed. The results demonstrated that alterations in the hemostatic system were significantly associated with the development of restenosis. These findings support early clinical risk assessment of restenosis and enable the implementation of personalized antiplatelet/anticoagulant therapy.
Keywords: Ischemic heart disease, Percutaneous coronary intervention, Restenosis, Hemostatic system, D-dimer, Fibrinogen, Prognostic index
Cite this paper: Mekhmanov Feruz Shamsievich, Naimova Shohida Anvarovna, Predictive Significance of Hemostatic System Markers for Coronary Restenosis, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 1799-1803. doi: 10.5923/j.ajmms.20261604.52.
1. Introduction
- Ischaemic heart disease (IHD) is currently recognised as one of the greatest threats to human health, remaining the leading cause of mortality and disability worldwide. According to the World Health Organization (WHO), more than 17 million people die each year globally as a consequence of IHD, and over half of these deaths occur in association with myocardial infarction and its complications [1]. In the treatment of IHD, contemporary interventional approaches—particularly percutaneous coronary intervention (PCI), i.e., transcutaneous instrumental dilation of the coronary vessels—are considered effective for relieving ischaemia and restoring myocardial perfusion [2,14]. However, the development of restenosis after PCI (in-stent restenosis, ISR) continues to represent a major challenge in clinical practice. Various studies report that restenosis occurs in approximately 10–30% of cases [3,4]. Coronary restenosis after percutaneous coronary intervention is a multifactorial process classically driven by vascular injury, inflammation, smooth-muscle cell proliferation, and extracellular matrix remodelling; however, haemostatic activation may contribute by linking early thrombus formation to later neointimal growth and adverse vessel healing. Contemporary reviews of in-stent restenosis (ISR) note that, beyond traditional clinical and lesion-related predictors, several circulating biomarkers—including components of coagulation and fibrinolysis—have been investigated as potential tools for risk stratification [15].Balloon angioplasty and stent implantation disrupt the endothelium and expose subendothelial procoagulant surfaces, which can trigger thrombin generation, platelet activation, and fibrin formation. These early events may influence late angiographic outcomes that are commonly used as quantitative restenosis endpoints and correlate monotonically with binary restenosis risk in interventional trials [16].The pathogenesis of restenosis is multifactorial and complex, involving endothelial dysfunction, inflammation, thrombogenesis, fibrinolysis, and neointimal hyperplasia. Within these processes, the haemostasis system plays a pivotal role. Parameters such as platelet activity, fibrinogen concentration, D-dimer levels, activated partial thromboplastin time (APTT), prothrombin time (PT), and the international normalised ratio (INR) reflect disturbances in microcirculation within the vascular wall and the risk of thrombosis [5,11]. Contemporary evidence suggests that subtle (subclinical) alterations in haemostasis may have important prognostic value for predicting the risk of restenosis following PCI [6,14]. In particular, elevated fibrinogen levels may amplify vascular wall inflammation and prothrombotic activity, whereas D-dimer reflects fibrinolytic activity and may be considered an indicator of restenosis probability [7,8].A key clinical study in stable coronary artery disease evaluated haemostatic markers after angioplasty and compared patients with and without restenosis using both categorical (>50% diameter stenosis) and continuous endpoints (late luminal loss). In that cohort (n=60; restenosis n=18), prothrombin fragment 1+2 (a marker of thrombin generation) helped discriminate restenosis status and showed a positive correlation with late luminal loss (reported r≈0.41). The authors interpreted these findings as consistent with a hypercoagulable profile—particularly enhanced thrombin generation—characterising a higher-risk restenosis phenotype.Tissue factor (TF), a potent initiator of the extrinsic coagulation pathway, has also been evaluated as a predictive marker. In a clinical study of PTCA patients, higher baseline TF levels were reported as a predictor of restenosis. This supports the concept that upstream “trigger” signals of coagulation initiation may carry prognostic value beyond routine coagulation times [17]. Among fibrin-related biomarkers, soluble fibrin (sF) has been repeatedly highlighted as a candidate “prethrombotic” marker reflecting ongoing fibrin formation. In one study of coronary angioplasty patients (n=92; restenosis history n=33), sF levels were reported to be elevated overall, with significantly higher sF concentrations in those with restenosis compared with those without occlusion; the authors concluded that a combined assessment (including sF and routine indices) was more informative than isolated tests [18]. In a subsequent prospective observational report with 18-month follow-up (n=91; history of late restenosis/stent thrombosis n=32), baseline haemostasis profiling included sF, D-dimer, fibrinogen, and protein C. The study described a pattern in thrombotic-complication patients characterised by markedly increased sF alongside relatively low D-dimer, interpreted as “depressed” fibrinolytic response; the authors also emphasised the clinical relevance of the sF/D-dimer relationship and concurrent reduction in anticoagulant capacity reflected by protein C [19]. From a broader haemostasis perspective, independent work on fibrin-related markers has shown that soluble fibrin and D-dimer reflect thrombotic activity and may differ in diagnostic utility across thrombotic conditions, supporting the biological plausibility of sF as a sensitive indicator of coagulation activation [20]. Platelet and fibrinolysis pathways have also been implicated. A cohort study reported that higher PAI-1 (a major inhibitor of fibrinolysis) and higher P-selectin (a platelet/endothelial activation marker), together with lower plasmin–α2-antiplasmin complex (PIC, reflecting plasmin activity), were associated with post-angioplasty restenosis. This pattern is mechanistically coherent with reduced fibrinolytic capacity plus enhanced platelet activation contributing to restenosis biology in at least a subset of patients [18].Fibrinogen is widely available and biologically relevant because it links inflammation, thrombosis, and blood viscosity. In a Circulation study, fibrinogen measured at follow-up after angioplasty was higher in restenosis groups and was associated with higher restenosis rates when above a specified threshold, suggesting fibrinogen may function as a risk marker in some settings. At the same time, literature across eras and procedural types shows heterogeneity—likely reflecting differences in endpoints, timing of sampling, antithrombotic therapy, stent technology, and patient comorbidity (e.g., diabetes). Therefore, fibrinogen (and similarly accessible parameters such as PT/INR, aPTT, platelet count, D-dimer) may be best interpreted as part of a multivariable risk profile rather than as definitive single predictors [21].VEGF as an adjunct biomarker connecting injury response and restenosis. Because restenosis involves vascular repair processes, angiogenesis-related mediators have been explored. In patients treated with drug-eluting stents, VEGF dynamics (increase after PCI) were reported to be associated with restenosis during follow-up, suggesting that post-procedural biomarker change may be more informative than baseline levels alone in some contexts [22]. Therefore, determining the prognostic significance of haemostatic markers in coronary artery restenosis represents a clinically and scientifically relevant objective. The findings of such research may contribute to developing individualised risk-assessment systems for patients after PCI, personalising antiplatelet and anticoagulant therapy, and ultimately reducing the incidence of restenosis.
2. Materials and Methods
- A total of 103 patients aged 18–74 years (48 men and 55 women) with ischaemic heart disease (IHD) who underwent percutaneous coronary intervention (PCI) at the Bukhara Regional Multidisciplinary Medical Center between 2021 and 2024 were included in this observational study. The control group comprised 30 apparently healthy volunteers.According to angiographic findings after PCI, the patients were stratified into restenosis and non-restenosis groups. Angiographic restenosis was defined as ≥50% luminal diameter stenosis within the stented segment or within the 5-mm proximal or distal peri-stent region.The following assessments were performed: clinical evaluation, including presenting complaints, auscultatory findings, disease stage, and PCI-related procedural characteristics; biochemical testing, including lipid profile analysis; haemostasis assessment, including D-dimer, fibrinogen, platelet count, activated partial thromboplastin time (APTT), prothrombin time (PT), and international normalised ratio (INR); and enzyme-linked immunosorbent assay (ELISA) for vascular endothelial growth factor (VEGF). Instrumental examinations included electrocardiography (ECG), echocardiography (EchoCG), and ultrasonography (US).Statistical analysis included correlation analysis, logistic regression modelling, and evaluation of diagnostic performance using sensitivity and specificity indices.
3. Results
- In the group that developed restenosis, D-dimer and fibrinogen concentrations were significantly higher than in the comparison group (p < 0.05). Other haemostatic components, including platelet count, APTT, and PT, also deviated from reference ranges. Based on regression analysis, an integral prognostic index was developed, demonstrating an estimated sensitivity of ~84% and specificity of ~79%. These findings indicate that restenosis risk can be predicted using the proposed marker set, potentially reducing the need for repeat PCI in clinical practice.
4. Discussion
- The present study demonstrated that patients who developed restenosis after PCI had more pronounced abnormalities in hemostasis-related parameters than both post-PCI patients without restenosis and healthy controls. As shown in Table 1, overall, between-group differences were statistically significant for all analyzed variables, including D-dimer, fibrinogen, platelet count, APTT, PT, and INR.
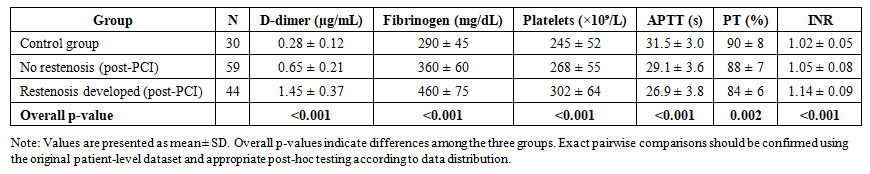 | Table 1. Hemostasis parameters in control subjects and post-PCI patients with and without coronary restenosis |
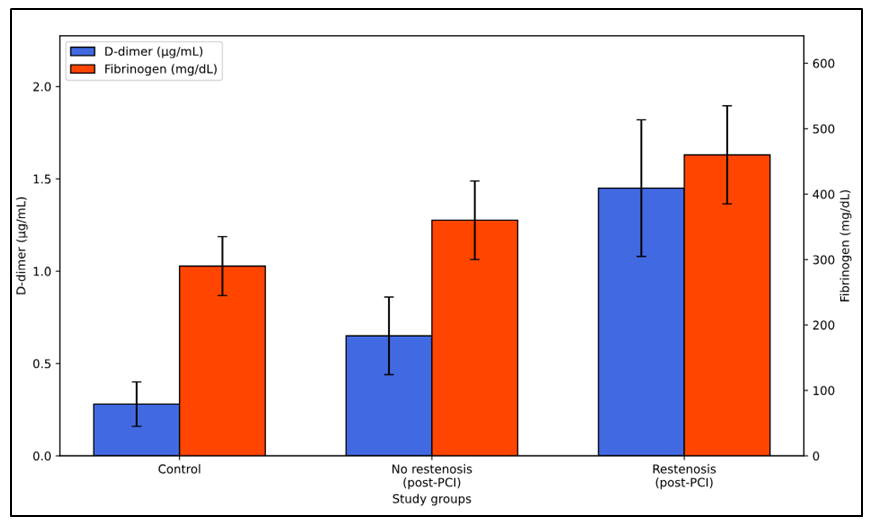 | Figure 1. Dual-axis comparison of D-dimer and fibrinogen levels across the study groups |
5. Conclusions
- The present study demonstrates that haemostasis system markers (D-dimer, fibrinogen, platelets, APTT, and PT) are reliably associated with restenosis development in patients with ischaemic heart disease undergoing percutaneous coronary intervention (PCI). In the restenosis group, markedly elevated D-dimer and fibrinogen levels (1,45 ± 0,37 µg/mL and 460 ± 75 mg/dL, respectively), APTT shortening (26,9 ± 3.8 s), and reduced PT (84 ± 6%) indicate activation of prothrombotic changes within the haemostasis system. These findings support the notion that endothelial microthrombosis, inflammation, and platelet-mediated pathological responses constitute key links in restenosis pathogenesis. Accordingly, the integral prognostic index developed through comprehensive assessment of these markers—showing clinically meaningful sensitivity (84%) and specificity (79%)—may serve as an effective tool for early detection of restenosis risk. Thus, implementation of routine monitoring of haemostatic parameters and biomarker-guided personalized antiplatelet/anticoagulant strategies may help reduce post-PCI complications, decrease the need for repeat interventions, and improve long-term patient outcomes.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML