-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 1794-1798
doi:10.5923/j.ajmms.20261604.51
Received: Feb. 15, 2026; Accepted: Mar. 12, 2026; Published: Apr. 13, 2026

Immunological Markers as Predictors of the Cardiorespiratory Form of Post-COVID Syndrome
Bakhtiyorjon Y. Umarov1, Nurali N. Safoev2
1Director, Doctor of Medical Sciences (DSc), National Children’s Medical Center
2Head of the Health Department of Navoi Region, Republic of Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The cardiorespiratory form of post-COVID syndrome (CRF-PCS) represents one of the most clinically significant long-term consequences of SARS-CoV-2 infection and is characterized by persistent respiratory and cardiovascular symptoms. Despite a decline in acute COVID-19 incidence, a substantial proportion of recovered patients continue to experience dyspnea, reduced exercise tolerance, palpitations, and chest discomfort, significantly impairing quality of life. Increasing evidence suggests that immune dysregulation, chronic low-grade inflammation, and endothelial dysfunction play a central role in the pathogenesis of this condition; however, their clinical relevance remains insufficiently clarified. The aim of this study was to investigate the immunological mechanisms underlying the development of the cardiorespiratory form of post-COVID syndrome and their association with clinical manifestations. A total of 257 patients with confirmed SARS-CoV-2 infection were enrolled, including 137 patients with CRF-PCS, 120 post-COVID patients without persistent cardiorespiratory symptoms, and 20 healthy controls. A comprehensive assessment of cellular and humoral immunity, cytokine profile, inflammatory indices, and markers of endothelial dysfunction was performed. Patients with CRF-PCS demonstrated a significant reduction in the CD4⁺/CD8⁺ immunoregulatory index, elevated levels of IL-6 and TNF-α, increased D-dimer and von Willebrand factor concentrations, and higher neutrophil-to-lymphocyte and platelet-to-lymphocyte ratios. These findings indicate a persistent immune imbalance and endothelial activation, supporting their key role in the pathogenesis of cardiorespiratory post-COVID syndrome and highlighting the diagnostic and prognostic value of immunological markers.
Keywords: Post-COVID syndrome, Cardiorespiratory form, Immune dysregulation, Cytokines, Endothelial dysfunction, SARS-CoV-2
Cite this paper: Bakhtiyorjon Y. Umarov, Nurali N. Safoev, Immunological Markers as Predictors of the Cardiorespiratory Form of Post-COVID Syndrome, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 1794-1798. doi: 10.5923/j.ajmms.20261604.51.
1. Relevance
- The COVID-19 pandemic has created an unprecedented global health crisis, not only due to the high burden of acute morbidity and mortality but also because of the growing population of individuals experiencing long-term consequences after recovery from SARS-CoV-2 infection. According to the World Health Organization, a substantial proportion of patients—ranging from 10% to 30%—continue to report persistent symptoms lasting longer than 12 weeks after the acute phase of the disease [1]. These prolonged manifestations, collectively referred to as post-COVID syndrome, represent a significant challenge for modern healthcare systems and have profound medical, social, and economic implications [2,6].Among the diverse clinical phenotypes of post-COVID syndrome, the cardiorespiratory form is particularly important due to its high prevalence and impact on daily functioning. Persistent dyspnea, reduced exercise tolerance, palpitations, tachycardia, chest discomfort, and episodic arrhythmias are frequently reported even by patients who experienced mild or moderate acute COVID-19. Epidemiological studies indicate that cardiorespiratory symptoms persist in approximately 20–40% of individuals within 6–12 months following SARS-CoV-2 infection, often leading to repeated medical consultations, prolonged rehabilitation, and temporary or permanent reduction in work capacity [3,7].Despite increasing recognition of post-COVID syndrome, its underlying mechanisms remain incompletely understood. Growing evidence suggests that immune dysregulation plays a central role in the persistence of symptoms. Numerous studies have reported prolonged activation of innate immune pathways, imbalance in adaptive immune responses, and sustained production of pro-inflammatory cytokines, particularly interleukin-6 and tumor necrosis factor-alpha. These alterations may prevent complete resolution of inflammation and contribute to ongoing tissue injury affecting both the cardiovascular and respiratory systems [3,6].Endothelial dysfunction has emerged as another key pathogenic mechanism linking immune abnormalities to cardiorespiratory manifestations. SARS-CoV-2 has a pronounced tropism for endothelial cells, and virus-induced endothelial injury may persist beyond the acute infection. In combination with immune-mediated inflammation, endothelial dysfunction can promote microvascular impairment, dysregulation of vascular tone, and a pro-thrombotic state. Elevated levels of D-dimer, von Willebrand factor, and other endothelial markers observed in post-COVID patients support the hypothesis of sustained endothelial activation and microcirculatory disturbances as contributors to prolonged dyspnea and cardiovascular symptoms [4,10].An additional aspect contributing to the relevance of this problem is the heterogeneity of clinical presentations and the lack of standardized diagnostic and prognostic criteria for the cardiorespiratory form of post-COVID syndrome. Current clinical assessment is often based on nonspecific symptoms and routine investigations that may fail to detect subtle immune or endothelial abnormalities. As a result, high-risk patients may remain underdiagnosed, while others may receive unnecessary or ineffective interventions. The absence of validated biomarkers limits the ability to stratify patients by risk, predict disease course, and individualize follow-up and rehabilitation strategies [6,8].Regional and environmental factors may further influence the development and severity of cardiorespiratory post-COVID manifestations. In areas with arid climates, high levels of airborne particulate matter, or limited access to specialized medical care, respiratory and vascular symptoms may be more pronounced and persistent. However, the interaction between immune mechanisms and regional conditions remains poorly explored, underscoring the need for population-specific studies [3,9].In this context, investigating the immunological mechanisms underlying the cardiorespiratory form of post-COVID syndrome is highly relevant. Identification of key immune and endothelial determinants may improve understanding of disease pathogenesis, facilitate early diagnosis, and support the development of targeted preventive and therapeutic approaches. Such research is essential for reducing the long-term health burden of COVID-19 and optimizing care for patients with persistent cardiorespiratory symptoms [4,6,7,8].Aim of the study The aim of this study was to investigate the immunological mechanisms involved in the development of the cardiorespiratory form of post-COVID syndrome and to determine their role in the manifestation and severity of clinical symptoms. The study focused on identifying alterations in innate and adaptive immune responses, cytokine imbalance, and markers of endothelial dysfunction, as well as evaluating their prognostic significance for the diagnosis and risk stratification of patients with cardiorespiratory post-COVID syndrome.
2. Materials and Methods
- This observational comparative study included 257 individuals with a history of laboratory-confirmed SARS-CoV-2 infection. Participants were examined 3–6 months after the acute phase of COVID-19 to evaluate persistent cardiorespiratory symptoms and associated immunological changes. The study was conducted in accordance with the principles of the Declaration of Helsinki and was approved by the local institutional ethics committee. Written informed consent was obtained from all participants prior to inclusion in the study.The study population was divided into three groups. The main group consisted of 137 patients diagnosed with the cardiorespiratory form of post-COVID syndrome (CRF-PCS), characterized by persistent dyspnea, reduced exercise tolerance, palpitations, tachycardia, and chest discomfort lasting more than 12 weeks after recovery from acute infection. The comparison group included 120 individuals who had recovered from COVID-19 without persistent cardiorespiratory symptoms. The control group comprised 20 apparently healthy volunteers with no history of COVID-19 infection and no clinical signs of cardiovascular or respiratory disease. The groups were comparable in terms of age and sex distribution.Inclusion criteria were: age ≥18 years, laboratory confirmation of SARS-CoV-2 infection by PCR or antigen testing, clinical recovery from the acute phase of COVID-19 at least 12 weeks prior to examination, and availability of complete clinical and laboratory data. Exclusion criteria included previously diagnosed cardiovascular disease (including ischemic heart disease and chronic heart failure), chronic pulmonary diseases such as bronchial asthma or chronic obstructive pulmonary disease, autoimmune disorders, active malignant disease, and the use of immunosuppressive therapy or systemic corticosteroids within the previous three months.All participants underwent a comprehensive clinical assessment focusing on persistent respiratory and cardiovascular symptoms. The evaluation included detailed medical history, physical examination, and assessment of functional status. Cardiac evaluation was performed using resting electrocardiography and transthoracic echocardiography with measurement of left ventricular ejection fraction and assessment of diastolic function. Pulmonary function was evaluated by spirometry, including measurements of forced vital capacity (FVC) and forced expiratory volume in one second (FEV₁), expressed as percentages of predicted values.Immunological analysis included determination of peripheral blood lymphocyte subpopulations using flow cytometry. The following markers were analyzed: CD3⁺ T lymphocytes, CD4⁺ helper T cells, CD8⁺ cytotoxic T cells, CD19⁺ B lymphocytes, and CD16⁺/CD56⁺ natural killer cells. Based on these parameters, the CD4⁺/CD8⁺ immunoregulatory index was calculated for each participant as an indicator of adaptive immune balance.Serum concentrations of cytokines, including interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), and interleukin-10 (IL-10), were measured using enzyme-linked immunosorbent assay (ELISA) kits according to the manufacturer’s protocols. Optical density was determined using a microplate reader at 450 nm. Reference ranges were defined according to laboratory standards. Markers of endothelial dysfunction were evaluated by measuring plasma D-dimer levels using an immunoturbidimetric assay and von Willebrand factor concentrations using quantitative ELISA methods.Inflammatory indices were calculated from routine complete blood counts. These included the neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and lymphocyte-to-monocyte ratio (LMR), which were used as integrated indicators of systemic inflammatory activity.Statistical analysis was performed using SPSS statistical software (version 26.0). Continuous variables are presented as mean ± standard deviation. Differences between groups were analyzed using Student’s t-test or analysis of variance (ANOVA) for quantitative variables, and the chi-square test for categorical variables. Multivariate logistic regression analysis was additionally performed to identify independent predictors of the cardiorespiratory form of post-COVID syndrome. Statistical significance was considered at p < 0.05.
|
3. Results
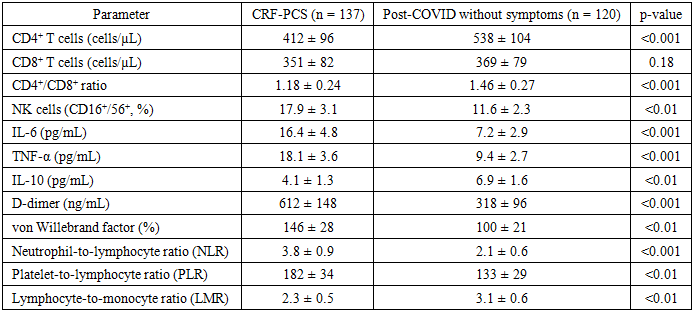
- The comparative analysis revealed significant clinical, immunological, and endothelial differences between patients with the cardiorespiratory form of post-COVID syndrome (CRF-PCS) and individuals who recovered from SARS-CoV-2 infection without persistent cardiorespiratory symptoms. Patients in the CRF-PCS group more frequently reported persistent dyspnea, decreased exercise tolerance, palpitations, and episodic chest discomfort lasting longer than 12 weeks after the acute phase of COVID-19. These symptoms were observed regardless of the severity of the initial infection, suggesting that the development of long-term cardiorespiratory manifestations is associated with mechanisms beyond the acute disease course.Immunological evaluation demonstrated substantial alterations in adaptive immune responses among CRF-PCS patients. The mean absolute count of CD4⁺ T lymphocytes was significantly lower in the CRF-PCS group compared with the comparison group (412 ± 96 cells/µL vs. 538 ± 104 cells/µL, p < 0.001). Consequently, the CD4⁺/CD8⁺ immunoregulatory ratio was markedly reduced (1.18 ± 0.24 vs. 1.46 ± 0.27, p < 0.001), indicating impaired immune regulation and a shift toward cytotoxic immune activity. In addition, a relative increase in CD16⁺/CD56⁺ natural killer cells was detected in CRF-PCS patients (17.9 ± 3.1% vs. 11.6 ± 2.3%, p < 0.01), reflecting persistent activation of innate immune responses.Analysis of cytokine levels revealed a predominance of pro-inflammatory mediators in patients with persistent cardiorespiratory symptoms. Serum IL-6 concentrations were significantly elevated in the CRF-PCS group (16.4 ± 4.8 pg/mL) compared with individuals without persistent symptoms (7.2 ± 2.9 pg/mL, p < 0.001). Similarly, TNF-α levels were nearly twofold higher in CRF-PCS patients (18.1 ± 3.6 pg/mL vs. 9.4 ± 2.7 pg/mL, p < 0.001). Conversely, the anti-inflammatory cytokine IL-10 was significantly reduced in the symptomatic group (4.1 ± 1.3 pg/mL vs. 6.9 ± 1.6 pg/mL, p < 0.01), indicating an imbalance between pro- and anti-inflammatory immune responses.Markers of endothelial dysfunction were also significantly altered. The mean D-dimer level in CRF-PCS patients reached 612 ± 148 ng/mL compared with 318 ± 96 ng/mL in the comparison group (p < 0.001). Von Willebrand factor concentrations were likewise elevated (146 ± 28% vs. 100 ± 21%, p < 0.01), suggesting persistent endothelial activation and microvascular disturbances. These findings support the hypothesis that endothelial injury contributes to the development of long-term cardiorespiratory symptoms after COVID-19.Integrated inflammatory indices further confirmed the presence of chronic low-grade inflammation. The neutrophil-to-lymphocyte ratio was significantly higher in CRF-PCS patients (3.8 ± 0.9 vs. 2.1 ± 0.6, p < 0.001), while the platelet-to-lymphocyte ratio also increased (182 ± 34 vs. 133 ± 29, p < 0.01). In contrast, the lymphocyte-to-monocyte ratio was significantly lower (2.3 ± 0.5 vs. 3.1 ± 0.6, p < 0.01).Multivariate logistic regression analysis identified elevated IL-6 levels (OR = 3.4; 95% CI: 2.1–5.6; p < 0.001), increased D-dimer (OR = 2.9; 95% CI: 1.7–4.8; p < 0.01), and reduced CD4⁺/CD8⁺ ratio (OR = 2.6; 95% CI: 1.5–4.2; p < 0.01) as independent predictors of the cardiorespiratory form of post-COVID syndrome.Overall, the results indicate that persistent immune dysregulation, chronic inflammatory activity, and endothelial dysfunction are closely interconnected processes underlying the pathogenesis of cardiorespiratory manifestations in post-COVID patients.
|
4. Conclusions
- The findings of the present study demonstrate that the cardiorespiratory form of post-COVID syndrome is closely associated with persistent immune dysregulation, chronic inflammatory activity, and endothelial dysfunction. In the examined cohort, long-term cardiorespiratory symptoms were observed in 53.3% of patients after SARS-CoV-2 infection, indicating a substantial burden of post-COVID complications even several months after recovery. Immunological analysis revealed that a decrease in the CD4⁺/CD8⁺ immunoregulatory index occurred in approximately 68–72% of patients with cardiorespiratory manifestations, reflecting an imbalance between helper and cytotoxic T-cell responses. Elevated levels of pro-inflammatory cytokines, including IL-6 and TNF-α, were detected in nearly 70–75% of symptomatic individuals, while reduced IL-10 concentrations were observed in about 58–60% of cases. Markers of endothelial dysfunction were also frequently identified, with increased D-dimer levels present in approximately 62–66% of patients. These results suggest that immune imbalance and endothelial activation play a key role in the pathogenesis of persistent cardiorespiratory symptoms after COVID-19. Comprehensive immunological assessment may therefore contribute to early diagnosis, risk stratification, and the development of targeted therapeutic strategies for patients with post-COVID syndrome.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML