Khodjaeva Nodira Vokhidovna1, Abdinazarova Maftuna Zokhid kizi2, Khodjayeva Feruza Sodikovna3, Dadakhanova Maryam Bakhtiyarovna4
1Assistant of the Department of Endocrinology with Pediatric Endocrinology, Tashkent State Medical University, Candidate of Medical Sciences, Uzbekistan
2Master's Degree Student, 2nd year, Tashkent State Medical University, Uzbekistan
3Assistant of the Department of Endocrinology with Pediatric Endocrinology, Tashkent State Medical University, Uzbekistan
4Assistant of the Department of Endocrinology with Pediatric Endocrinology, Tashkent State Medical University, PhD, Uzbekistan
Correspondence to: Khodjaeva Nodira Vokhidovna, Assistant of the Department of Endocrinology with Pediatric Endocrinology, Tashkent State Medical University, Candidate of Medical Sciences, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
According to the World Health Organization, obesity and type 2 diabetes mellitus have reached the scale of a 21st century epidemic [2]. All countries are now affected by obesity and the impact is predicted to become even more prominent, resulting in more years of lost healthy life, disability and death [12]. In fact, it is known that up to 85.2% of people with type 2 diabetes mellitus have an overweight or obesity problem, and by 2025, more than 300 million people will have obesity-related type 2 diabetes mellitus [11]. Obesity and type 2 diabetes mellitus are chronic diseases that are on the rise worldwide, requiring new approaches to treat and prevent diabetes in obese people. Therefore, it is important to understand the mechanistic relationship between the two and develop a comprehensive approach to increase life expectancy and improve the quality of life of patients. This literature review provides detailed information of the indispensable role of obesity in the pathophysiology of type 2 diabetes mellitus and their mechanistic link [5].
Keywords:
Obesity, Type 2 diabetes mellitus, Fatty acids, Lipids, Inflammation, Adipocytokines, Leptin, Adiponectin, Gut microbiota, Beta cells, Insulin
Cite this paper: Khodjaeva Nodira Vokhidovna, Abdinazarova Maftuna Zokhid kizi, Khodjayeva Feruza Sodikovna, Dadakhanova Maryam Bakhtiyarovna, The Vicious Circle of Obesity and Diabetes: Pathogenesis and Relationship, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 1782-1787. doi: 10.5923/j.ajmms.20261604.49.
1. Introduction
Type 2 diabetes mellitus is one of the most common metabolic disorders worldwide. Its development is primarily due to a combination of two major factors: defective insulin secretion by pancreatic β-cells and insulin resistance. Insulin secretion and action must precisely match metabolic needs. Consequently, the molecular mechanisms involved in insulin synthesis and secretion, as well as the insulin response in tissues, must be tightly regulated. Thus, defects in any of the mechanisms involved can lead to metabolic imbalances that result in the development of type 2 diabetes mellitus [6]. Obesity is a condition of abnormal increase in the amount of adipose tissue, which increases the risk of numerous diseases such as cardiovascular disease, certain cancers and type 2 diabetes mellitus. There are a number of reasons that lead to the development of obesity, including genetic and environmental factors. Studies have shown that not only the amount of calories consumed, but also the type of diet, frequency and timing of meals influence the development of obesity and related metabolic disorders [8]. Obesity plays an inevitable role in the increasing prevalence of type 2 diabetes mellitus, in which the body does not produce enough insulin or cannot utilize insulin efficiently, resulting in elevated blood glucose levels. It is the fastest growing pandemic and public health emergency in the world [3].Consequently, this literature review will examine several theories explaining the pathogenetic relationship between obesity and type 2 diabetes mellitus.
2. Main Text
Lipotoxicity theoryIncreased gluconeogenesis in obese individuals causes fasting and post-meal hyperglycemia. Increased hepatic gluconeogenesis is due to impaired suppression of adipose tissue lipolysis, which increases the delivery of free fatty acids to the liver. In obese individuals, increased lipid oxidation causes high plasma concentrations of free fatty acids, leading to insulin resistance. This "lipid spillover" limits glucose oxidation. Studies show that glucose uptake is the rate-limiting step in fatty acid-induced insulin resistance, not intracellular glucose metabolism. According to this model, fatty acids, together with metabolites such as acyl-CoA, ceramides, and diacylglycerols, serve as signaling molecules that activate protein kinases. These protein kinases disrupt insulin signaling by increasing the phosphorylation of insulin receptor substrate serine, which are key mediators of insulin receptor signaling [5].A striking feature in determining the risk of developing obesity-related type 2 diabetes mellitus is the pattern of fat distribution throughout the body. For example, obese individuals with abdominal subcutaneous fat, intra-abdominal fat, intrahepatic triglycerides and pancreatic fat are at higher risk of developing type 2 diabetes mellitus compared to those with less body fat accumulation. The role of intramyocellular lipids (IMCL) is increasingly recognized as an important factor in the regulation of insulin action. IMCL accumulation in obese patients is due to increased levels of lipid peroxidation and the production of lipid peroxidation by-products such as 4-hydroxynonenal (4-HNE), as well as reduced fatty acid beta-oxidation in muscle, which affects insulin sensitivity.Ectopic lipid accumulation is associated with insulin resistance due to reactive oxygen species (ROS) activation, mitochondrial dysfunction or endoplasmic reticulum (ER) stress. The persistent imbalance between ROS and antioxidant production is a major cause of oxidative stress leading to fat accumulation in humans. Increased ROS in prediabetes is caused by increased fatty acids, which leads to oxidative stress due to increased mitochondrial uncoupling and beta-oxidation subsequently leading to increased ROS production. In insulin resistance, ectopic fat accumulation is associated with decreased mitochondrial oxidative activity and ATP synthesis [5]. Lipotoxicity, has been considered as a major factor in the onset of type 2 diabetes mellitus, contributing to tissue inflammation and insulin resistance in peripheral tissues. Moreover, it has been suggested that it may additionally contribute to the disease by directly altering ß-cell function and differentiation.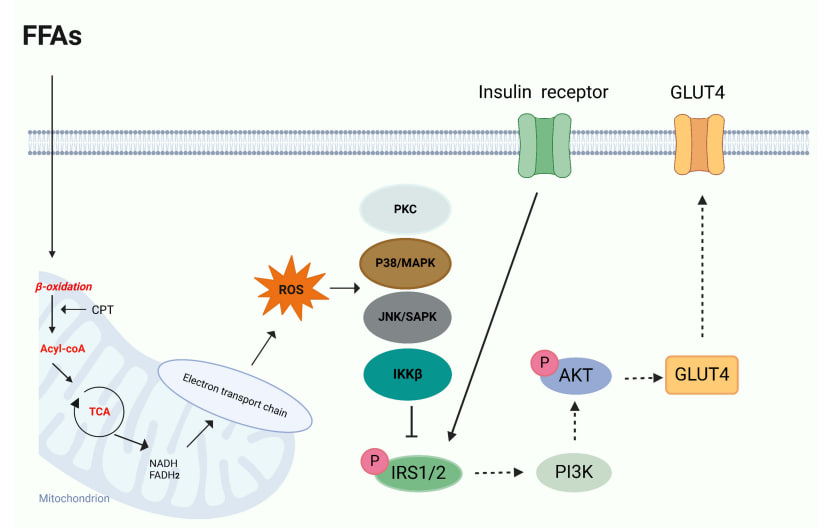 | Figure 1. Lipotoxicity activates OS and mitochondrial dysfunction in pancreatic β-cells. The accumulation of FFAs leads to increased beta oxidation, resulting in the production of excess acetyl-CoA, which subsequently enters the tricarboxylic acid (TCA) cycle. This cycle becomes overloaded, generating elevated levels of NADH and FADH2, which enhance the activity of the electron transport chain. This heightened activity results in the overproduction of ROS, contributing to oxidative stress and mitochondrial dysfunction. The excessive generation of ROS adversely affects insulin signaling by activating stress kinases, including c-Jun N-terminal kinase (JNK), IκB kinase beta (IKKβ), p38 MAPK, and protein kinase C (PKC). Consequently, the phosphorylation of insulin receptor substrate (IRS) at serine residues inhibits insulin signaling and induces insulin resistance [16] |
Most studies of lipotoxicity and ß-cell viability have examined the effects of palmitate and oleate; the former saturated fatty acid was more harmful and promoted ß-cell death. Palmitate is a precursor to ceramide biosynthesis, a form of fat that may contribute to lipotoxicity. Intracellular ceramide concentrations increase significantly after exposure to palmitate, which is associated with ß-cell dysfunction and apoptosis [7]. Chronic inflammation theoryAdipose tissue produces numerous adipokines such as adiponectin, leptin, visfatin, resistin, apelin, omentin, retinol-binding protein (RBP4), vaspin and many others that affect the overall activity of various organs including liver, pancreas, intestine, brain and skeletal muscles. These processes include adipocyte hypoxia, which is caused by increased oxygen demand and stimulates fibrogenesis and macrophage chemotaxis. This leads to increased plasma concentrations of branched-chain amino acids, increased numbers of adipocyte-derived macrophages and T cells, decreased adipose tissue and adiponectin (an insulin-sensitive hormone) production, increased adipose tissue lipolytic activity leading to the release of free fatty acids into the bloodstream and altered exosomes derived from adipose tissue macrophages. Among these factors, it has been suggested that adipose tissue inflammation is a major driver of insulin resistance in obese individuals. Activated macrophages and hypertrophied adipocytes increase levels of pro-inflammatory cytokines such as TNF-α, IL-1β, monocyte chemoattractant protein-1 (MCP-1) and IL-6, ultimately leading to the development of metabolic inflammation. This chronic inflammatory state is a key factor contributing to the pathogenesis of insulin resistance, which reduces glucose uptake in muscle, leads to increased glucose production in the liver, provokes β-cell dysfunction in the pancreas and leads to endocrine dysfunction in adipose tissue [5].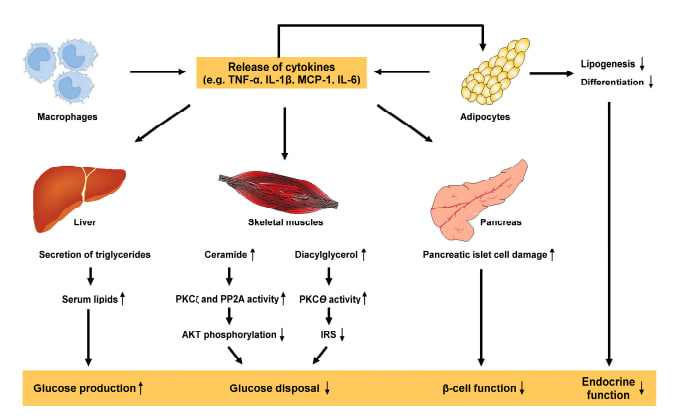 | Figure 2. Pro-inflammatory cytokines in the pathogenesis of metabolic inflammation and insulin resistance [5] |
Adipocytokine theoryLeptin. Lack of leptin or its receptors leads to uncontrollable hunger, potentially leading to obesity. Numerous studies have demonstrated a strong correlation between leptin levels and body fat percentage, with an increase in adipocytes triggering elevated leptin levels as an adaptive response to control energy balance. Elevated leptin levels are associated with insulin resistance and type 2 diabetes. In this particular population, there is an association between hyperleptinemia and a higher risk of cardiovascular disease, as well as more high rates of obesity, hypertension and endothelial dysfunction. Leptin plays a role in the regulation of proinflammatory cytokines, which are also associated with insulin resistance [14]. Adiponectin. An important biomarker of adipose tissue health is plasma adiponectin, which is directly related to insulin sensitivity and has an inverse relationship with body fat percentage [10]. Adiponectin has multiple functions. It is well known that adipose tissue maintains energy homeostasis of the whole body. Obesity, which occurs due to accumulation of WAT in internal organs, results in lack of angiogenesis in subcutaneous adipose tissue. This results in an inability to store excess energy, leading to insulin resistance in obese individuals. Hence, there may be a link between adipose tissue angiogenesis, vascular function and insulin sensitivity [9]. The gut microbiota theoryThe gut microbiota refers to the complex ecosystem colonized in the human intestinal tract, which includes a large number of microorganisms including bacteria, fungi, viruses, archaea, protozoa, etc.If there is an imbalance in bacterial composition, changes in bacterial metabolic activity, or changes in the distribution of bacteria in the gut, this is defined as "dysbiosis". Gut dysbiosis in obesity is a major contributor to lipid accumulation. The altered gut microbiota in obese individuals leads to higher concentrations of lipopolysaccharides, which triggers a number of inflammatory reactions and causes metabolic endotoxemia. Leads to increased expression of pro-inflammatory cytokines in adipose tissue (including interleukin-6 and TNF-α), which can lead to insulin resistance. In addition, the gut microbiota also favors lipid accumulation through induction of leptin resistance and inhibition of fat-suppressing neuropeptides [4]. Studies have shown that the gut microbiota plays an important role in weight regulation, which may influence fat and energy storage through both energy regulation and gene expression [15]. Although there are diverse bacterial species in the gut microbiota, it was recently found that Firmicutes (F) and Bacteroidetes (B) are the major phyla in the gut microbiota. In light of the growing concerns regarding obesity and type 2 diabetes, emerging studies have found that different species, a combination of strains, or the ratio of these two phyla could potentially impact obesity through multiple mechanisms. The ratio of Firmicutes to Bacteroidetes (F/B) is increasingly considered as a crucial biomarker for obesity regulation and metabolic health [17].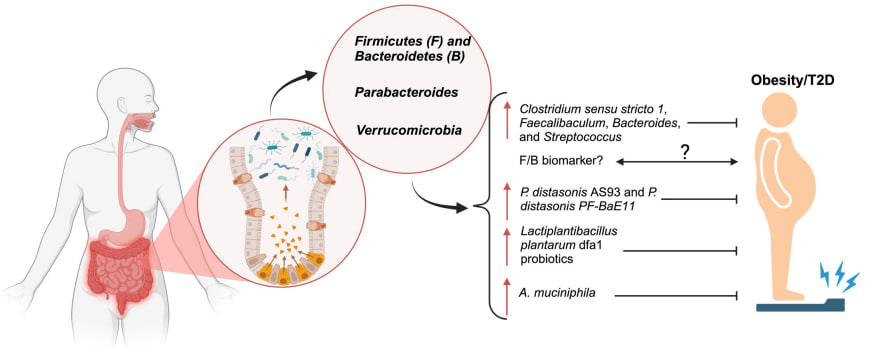 | Figure 3. The composition and balance of four major gut microbiota and their roles in obesity. The composition and balance of gut microbiota are pivotal in regulating metabolic processes and influencing the development and progression of obesity. The illustration highlights key bacterial phyla and genera, including Firmicutes, Bacteroidetes, Parabacteroides, and Verrucomicrobia, and their roles in obesity and type 2 diabetes mellitus [17] |
Hyperinsulinemia and β-cell depletion theoryBeta cells produce insulin, which is then sent to the liver via the portal vein. The balance of insulin in the bloodstream depends on a delicate balance between its secretion and the liver's ability to clear it. After release by beta cells, the liver utilizes more than half of this insulin. Factors such as increased insulin secretion in the pancreas and reduced partial extraction and filtration of insulin as it enters the cells combine to result in elevated basal plasma insulin levels in obese individuals after meals. The persistent decline in pancreatic beta cell performance causes a consistent weakening of blood sugar control, paving the way for prediabetes and eventually the onset of type 2 diabetes [1]. To compensate for increased insulin resistance, β-cells initially increase insulin secretion. However, over time, the number and secretory response of healthy β-cells decrease due to glucolipotoxicity, endoplasmic reticulum stress, mitochondrial dysfunction, and inflammation, which overlap with genetic and epigenetic factors [13]. The anti-aging gene Sirtuin 1Unhealthy diets inactivate the calorie sensitive gene Sirtuin 1 (Sirt 1) involved in epigenetic processes that promote immune system alterations, mitochondrial apoptosis, Non-alcoholic Fatty Liver Disease (NAFLD), diabetes and Nitric Oxide (NO) modification with relevance to core body temperature involved with appetite regulation, glucose homeostasis and hepatic xenobiotic metabolism [18].Sirt 1 is important to SCN function that maintains synchrony between neurons that is critical to circadian rhythm and glucose clearance in the brain and periphery. Metabolic disturbances in neurons may be linked to other anti-aging genes associated glucose dyshomeostasis and dyslipidemia. Interests in calorie restriction, appetite regulation and neurodegeneration that involve Sirt 1 mediated regulation of other anti-aging genes has accelerated and involve p53 and FOXO deacetylation in relation to autonomous disease of the brain and liver. Nutritional interventions that maintain Sirt 1 activity are important to T3D, T2D and T1D with Sirt 1 down regulation in NAFLD relevant to the defective metabolism of caffeine, glucose, fatty acids, cholesterol, amyloid beta, bile acids, and xenobiotics [19].Sirt 1 interactions with forkhead transcription factor O1 (FOXO1), C/EBP alpha may involve Klotho C/EBP alpha and peroxisome Proliferator-Activated Receptor (PPAR) interactions important to mitochondrial function and adipocyte differentiation. Furthermore miR-122 and miR-132 have been shown directly inhibit Sirt 1 and may interfere with adipose tissue adiponectin release.Sirt 1 is connected to immunometabolism and adipogenesis disorders with adipose tissue release of adipokines, inflammatory cytokines, heat shock proteins and natural killer cells relevant to mitophagy in diabetes and MODS [18].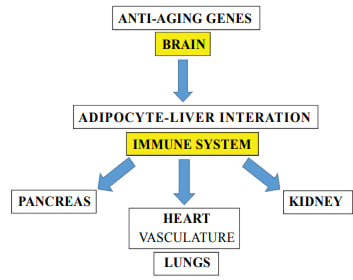 | Figure 4. Nutritional diets and environmental xenobiotics are now involved with the repression of anti-aging genes with epigenetic alterations linked to the global chronic disease epidemic. Circadian dyssynchrony and immune system imbalances involve mitochondrial apoptosis in many tissues with relevance to diabetes and Multiple Organ Disease Syndrome (MODS) |
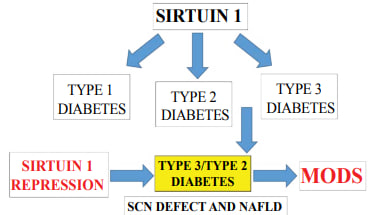 | Figure 5. Nutritional regulation of Sirt 1 is important to prevent insulin resistance and mitophagy in diabetic individuals. Individuals with Type 3/Type 2 diabetes have SCN defects with accelerated NAFLD and MODS associated with hepatic xenobiotic metabolism and mitophagy |
Specific genes that are involved in epigenetics are sensitive to nutritional regulation, oxidative stress and the development of insulin resistance that can result from changes in cellular chromatin structure, DNA methylation and histone modifications with relevance to the global chronic disease epidemic [18].
3. Conclusions
In conclusion, the results of this literature review confirm the close relationship between obesity and type 2 diabetes mellitus due to common pathogenetic mechanisms and risk factors. A better understanding of the relationship and causality between these factors may enable risk to be predicted, modified and controlled, as there is substantial evidence that weight loss interventions can reduce blood glucose levels. This review also emphasizes the need for further in-depth studies to investigate these diseases, risk factors and mechanisms of pathogenesis, which in turn will improve the management of these conditions and the quality of life of patients.
References
| [1] | Albaji, Maitam Abdallah, Dan Florin Mihailescu. "Obesity and diabetes mellitus 2: an interrelated relative relationship". International Journal of Research and Review (2023): n. p. cm. |
| [2] | Saini, Simmi, Mohinder Pal Sachdeva and Vipin Gupta. "Association between obesity and type 2 diabetes mellitus." The Asian Man - An International Journal 12 (2018): 196-202. |
| [3] | Chandrasekaran, Priti, Ralph Weisskirchen. "Role of obesity in type 2 diabetes mellitus - a review". International Journal of Molecular Sciences 25 (2024): n. p. cm. |
| [4] | Shorouk Gamal Salah Ibrahim, Abd El-Rauf Mohamed El-Deib, Sameh Tolba Abu-Elela, Mohamed Osama Abdallah, Ahmed Mohamed Mossad. Egyptian Reviews of Medical and Health Sciences (2024). |
| [5] | Chandrasekaran, Preethi, and Ralf Weiskirchen. “The Role of Obesity in Type 2 Diabetes Mellitus-An Overview.” International journal of molecular sciences vol. 25, 3 1882. 4 Feb. 2024, doi: 10.3390/ijms25031882. |
| [6] | Galicia-Garcia, Unai et al. "Pathophysiology of Type 2 Diabetes Mellitus." International journal of molecular sciences vol. 21, 17 6275. 30 Aug. 2020, doi: 10.3390/ijms21176275. |
| [7] | Jiménez-Sánchez, C., Oberhauser, L., Maechler, P. Role of fatty acids in the pathogenesis of ß-cell failure and Type-2 diabetes. Atherosclerosis. 2024 Nov; 398: 118623. doi: 10.1016/j.atherosclerosis.2024.118623. |
| [8] | Kojta, I., Chacińska, M., Błachnio-Zabielska, A. Obesity, Bioactive Lipids, and Adipose Tissue Inflammation in Insulin Resistance. Nutrients. 2020 May 3; 12(5): 1305. doi: 10.3390/nu12051305. PMID: 32375231; PMCID: PMC7284998. |
| [9] | Lemoine, A.Y.; Ledoux, S.; Quéguiner, I.; Caldérari, S.; Mechler, C.; Msika, S.; Corvol, P.; Larger, E. Link between adipose tissue angiogenesis and fat accumulation in severely obese subjects. J. Clin. Endocrinol. Metab. 2012, 97, E775-E780. |
| [10] | Li, N.; Zhao, S.; Zhang, Z.; Zhu, Y.; Gliniak, C.M.; Vishvanath, L.; An, Y.A.; Wang, M.Y.; Deng, Y.; Zhu, Q.; et al. Adiponectin preserves metabolic fitness during aging. eLife 2021, 10, e65108. |
| [11] | Ortega, M. A., Fraile-Martínez, O., Naya, I., García-Honduvilla, N., Álvarez-Mon, M., Buján, J., Asúnsolo, Á., & de la Torre, B. (2020). Type 2 Diabetes Mellitus Associated with Obesity (Diabesity). The Central Role of Gut Microbiota and Its Translational Applications. Nutrients, 12(9), 2749. |
| [12] | Ruze, R., Liu, T., Zou, X., Song, J., Chen, Y., Xu, R., Yin, X., & Xu, Q. (2023). Obesity and type 2 diabetes mellitus: connections in epidemiology, pathogenesis, and treatments. Front. Endocrinol. 14: 1161521. doi: 10.3389/fendo.2023.1161521. |
| [13] | Strati, M., Moustaki, M., Psaltopoulou, T., Vryonidou, A., Paschou, S.A. Early onset type 2 diabetes mellitus: an update. Endocrine. 2024 Sep; 85(3): 965-978. doi: 10.1007/s12020-024-03772-w. Epub 2024 Mar 12. PMID: 38472622; PMCID: PMC11316703. |
| [14] | Vilariño-García, T., Polonio-González, M. L., Pérez-Pérez, A., Ribalta, J., Arrieta, F., Aguilar, M., Obaya, J. C., Gimeno-Orna, J. A., Iglesias, P., Navarro, J., Durán, S., Pedro-Botet, J., & Sánchez-Margalet, V. (2024). Role of Leptin in Obesity, Cardiovascular Disease, and Type 2 Diabetes. International journal of molecular sciences, 25(4), 2338. |
| [15] | Zhang, L., Wang, P., Huang, J., Xing, Y., Wong, F. S., Suo, J., & Wen, L. (2024). Gut microbiota and therapy for obesity and type 2 diabetes. Frontiers in endocrinology, 15, 1333778. |
| [16] | Chen B, Li T, Wu Y, et al. Lipotoxicity: A New Perspective in Type 2 Diabetes Mellitus. Diabetes Metab Syndr Obes. 2025; 18: 1223-1237. Published 2025 Apr 24. doi: 10.2147/DMSO.S511436. |
| [17] | Liu, Siman et al. “The Functions of Major Gut Microbiota in Obesity and Type 2 Diabetes.” Metabolites vol. 15, 3 167. 1 Mar. 2025, doi: 10.3390/metabo15030167. |
| [18] | Martins IJ (2017) Single Gene Inactivation with Implications to Diabetes and Multiple Organ Dysfunction Syndrome. J Clin Epigenet. Vol. 3 No. 3: 24. |
| [19] | Martins IJ (2017) Nutrition Therapy Regulates Caffeine Metabolism with Relevance to NAFLD and Induction of Type 3 Diabetes. J Diabetes Metab Disord 4: 019. |



 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML


