-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 1773-1779
doi:10.5923/j.ajmms.20261604.47
Received: Mar. 19, 2026; Accepted: Apr. 3, 2026; Published: Apr. 11, 2026

Diagnostic and Prognostic Significance of Imaging Modalities in Pediatric Nephroblastoma
Tojiddin Kurbonovich Mustafaev
Candidate of Medical Science, Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology, Ministry of Health of the Republic of Uzbekistan
Correspondence to: Tojiddin Kurbonovich Mustafaev, Candidate of Medical Science, Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology, Ministry of Health of the Republic of Uzbekistan.
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

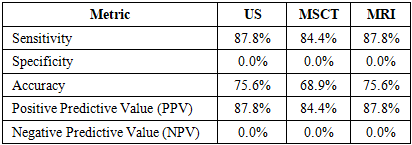
Background: Nephroblastoma is the most prevalent malignant renal tumor in children, requiring precise diagnostics for accurate prognostication and appropriate therapeutic selection. The efficacy of ultrasound (US), multislice computed tomography (MSCT), and magnetic resonance imaging (MRI) in assessing tumor invasion and the risk of metastasis remains a subject of debate. Objective: To evaluate the diagnostic and prognostic utility of US, MSCT, and MRI in pediatric nephroblastoma by correlating radiological data with intraoperative findings. Materials and Methods: An analysis was conducted on 90 patients who underwent comprehensive imaging, surgical intervention, and a subsequent 3-year follow-up. Diagnostic efficacy was assessed based on sensitivity, specificity, and predictive values. Results: Both US and MRI demonstrated a sensitivity of 87.8%, whereas MSCT exhibited a sensitivity of 84.4%. The specificity for all evaluated modalities was 0%. MRI proved superior in detecting vascular and soft-tissue invasion. Tumor invasion, as identified by MRI and MSCT, was significantly associated with the presence of metastases. Conclusion: MRI is the most informative imaging modality for assessing tumor invasion and determining prognosis in pediatric nephroblastoma. A comprehensive approach that incorporates both morphological and intraoperative data is essential.
Keywords: Nephroblastoma, Children, Invasion, Imaging, Prognosis
Cite this paper: Tojiddin Kurbonovich Mustafaev, Diagnostic and Prognostic Significance of Imaging Modalities in Pediatric Nephroblastoma, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 1773-1779. doi: 10.5923/j.ajmms.20261604.47.
1. Introduction
- Nephroblastoma (Wilms tumor) is the most prevalent malignant renal tumor in children, accounting for approximately 6% of all pediatric malignancies and up to 95% of childhood malignant renal neoplasms [2,7]. According to the Surveillance, Epidemiology, and End Results (SEER) data, the annual incidence of nephroblastoma is approximately 8 cases per million children under 15 years of age, with a peak incidence occurring at 3–4 years of age [6] (SEER Cancer Statistics Review, 1975–2020).The incidence of nephroblastoma varies by ethnicity: it is higher among African American children and lower among Asian populations, highlighting the role of genetic and population-based factors in the etiology of the tumor [4].The etiopathogenesis of nephroblastoma in the majority of cases is associated with somatic mutations occurring in embryonic renal tissue, frequently involving the WT1, WTX, and CTNNB1 genes [8] (Haber D.A., 2005). In a subset of patients, the disease is linked to hereditary syndromes, such as WAGR (Wilms tumor, Aniridia, Genitourinary anomalies, mental Retardation) syndrome, Denys-Drash syndrome, and Beckwith-Wiedemann syndrome, in which the risk of developing nephroblastoma is significantly elevated [5,10].These associations indicate that congenital genetic defects play a substantial role in tumor development in a specific patient cohort. According to the International Society of Paediatric Oncology (SIOP), the presence of congenital genitourinary anomalies and other dysplastic changes also elevates the likelihood of nephroblastoma manifestation [15].Continuing the analysis of the diagnostic aspects of imaging modalities in nephroblastoma, it is essential to emphasize both their strengths and the limitations identified in the present study.Ultrasound (US) is traditionally utilized as the primary diagnostic modality for pediatric renal tumors. According to Dome et al. (2013), US exhibits high sensitivity in detecting mass lesions, particularly during initial presentation; however, its capability to differentiate vascular and extracapsular invasion is substantially limited [3]. Davidoff (2013) emphasizes that low spatial resolution and significant operator dependence render US an unreliable tool for assessing invasive tumor growth, especially in cases of complex anatomy [2].Multislice computed tomography (MSCT), as suggested by Shamberger et al. (2001), is more effective in evaluating the anatomical boundaries of the tumor and is particularly indicated for detecting pulmonary metastases, which is crucial for accurate staging [11]. However, as demonstrated in a review by Spreafico et al. (2020), the sensitivity of MSCT in determining vascular invasion remains moderate, and even with the implementation of contrast-enhanced phases, the accuracy of the method in certain studies does not exceed 70–80% [13].Conversely, magnetic resonance imaging (MRI), according to Smets et al. (2008), provides superior visualization of soft-tissue structures and vessels, making it the preferred modality when invasion into the inferior vena cava or perinephric fat is suspected [12]. Vujanić et al. (2011) highlight that the sensitivity of MRI in assessing vascular invasion can reach 95%, particularly when specialized sequences are employed [15].Nevertheless, despite its high sensitivity, as demonstrated in our study, the specificity of all three imaging modalities remains at zero. This is attributed to the absence of reliably confirmed true-negative observations, as well as potential overdiagnosis associated with the clinical caution of the interpreters. According to Spreafico et al. (2020), this phenomenon is typical in pediatric oncology, where even minimal suspicious findings are frequently interpreted as potentially invasive to avoid underestimating the tumor stage [13].For instance, a study by Varan et al. (2007) demonstrated that signs of extracapsular extension detected by MSCT and MRI are associated with a higher risk of recurrence. The authors emphasize that preoperative evidence of invasion into the vascular bed or adjacent organs can serve as an adverse prognostic marker, especially in bilateral lesions or non-blastomatous components [14].A study by Chung et al. (2010), based on a cohort of nephroblastoma patients in the USA, also demonstrated that the detection of invasive signs via MRI significantly correlates with an increased rate of metastasis, particularly in cases where the tumor infiltrates the perinephric fat or liver [1].Concurrently, according to Rogowski et al. (2013), the utility of US in evaluating prognostic features is limited. Despite its widespread availability and safety profile, US is less informative in detecting invasive growth patterns and does not reliably predict the risk of metastasis [9]. The authors note the method's low sensitivity in recognizing vascular invasion and deep tissue penetration, which leads to an underestimation of prognosis in a portion of patients.Our work also corroborated these observations: the frequency of recurrences and metastases was significantly higher in patients with signs of tumor extension detected by MRI and MSCT, particularly with invasion into the liver, lungs, and inferior vena cava. The differences obtained via MRI reached statistical significance (p < 0.05), underscoring the method's potential not merely as a diagnostic tool, but as a prognostic instrument.Thus, current data indicate that MRI and MSCT possess defined prognostic value in nephroblastoma, particularly in assessing local invasion and its association with the risk of recurrence and metastasis. US, despite its accessibility, remains the method of primary screening but strictly requires confirmation by more accurate modalities when aggressive growth is suspected.Within the framework of our study, we also aimed to evaluate the prognostic significance of imaging modalities in the context of the risk of recurrence and distant metastasis, taking into account the specific pathways of tumor extension, which formed the basis of the presented analysis.Objective: To evaluate the diagnostic informativeness of imaging modalities—ultrasound (US), multislice computed tomography (MSCT), and magnetic resonance imaging (MRI)—in determining the direction and characteristics of tumor extension in patients with nephroblastoma. Furthermore, the study aimed to determine their prognostic significance concerning the risk of recurrence and distant metastasis based on correlation with intraoperative and morphological data.
2. Materials and Methods
- The study included 90 patients with morphologically verified nephroblastoma who underwent comprehensive treatment and follow-up at the Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology (RSSPMCOR).Inclusion criteria were: a confirmed diagnosis of nephroblastoma; completion of a comprehensive diagnostic imaging panel, including US, MSCT, and MRI; availability of a detailed surgical protocol describing intraoperative findings; completion of the treatment course including a surgical stage; and a patient follow-up period of at least 3 years post-intervention.Patients were excluded from the study if they had incomplete imaging data, lacked surgical descriptions or histological verification, experienced a fatal outcome prior to surgical treatment, or had nephroblastoma associated with congenital syndromes where an objective evaluation of independent tumor invasiveness was unfeasible.Morphological verification was established based on postoperative histological reports, specifying the nephroblastoma variant, extent of spread, and specific features of invasion. Intraoperative data recorded in surgical protocols served as the reference standard for the comparative efficacy assessment of the diagnostic imaging modalities.Statistical data processing was performed using Pearson's chi-square ($\chi^2$) test to evaluate the significance of differences between groups. The diagnostic efficacy of the imaging modalities was assessed using standard metrics: sensitivity, specificity, accuracy, positive predictive value (PPV), and negative predictive value (NPV). A 2x2 contingency table was utilized as the basis for analysis: true positives (TP), false positives (FP), true negatives (TN), and false negatives (FN).Calculations were performed using the following formulas:Sensitivity = TP / (TP + FN),Specificity = TN / (TN + FP),Accuracy = (TP + TN) / (TP + FP + FN + TN),PPV = TP / (TP + FP),NPV = TN / (TN + FN).
3. Results
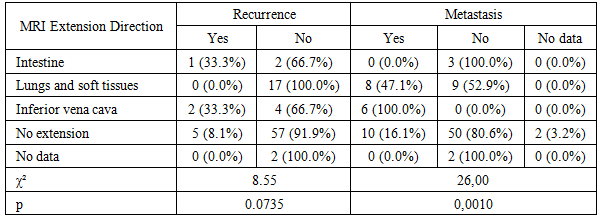
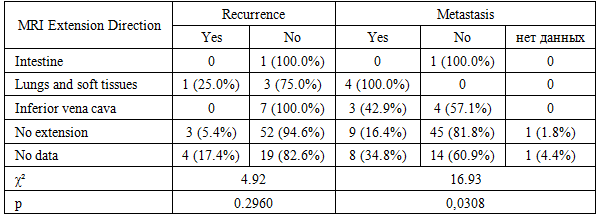
- Analysis of the clinical-demographic, morphological, and instrumental characteristics of the patients revealed several patterns reflecting the clinical course of pediatric nephroblastoma managed at a specialized oncological center. The entire cohort (n = 90) received standard therapy, encompassing stages of polychemotherapy, surgical intervention, and, when indicated, radiation therapy, ensuring the completeness of observations and the reliability of the derived conclusions.The distribution of patients by gender and age is presented in Figure 1. The majority of patients (66.7%) were children under 3 years of age, with a predominance of girls—34 cases (37.8%) compared to 26 boys (28.9%). Among children older than 3 years, there were 17 girls (18.9%) and 13 boys (14.4%), which also reflects a moderate female predominance. Thus, a pronounced accumulation of cases is observed in the younger age cohort, aligning with the well-established epidemiological characteristics of nephroblastoma, which predominantly manifests in early childhood.
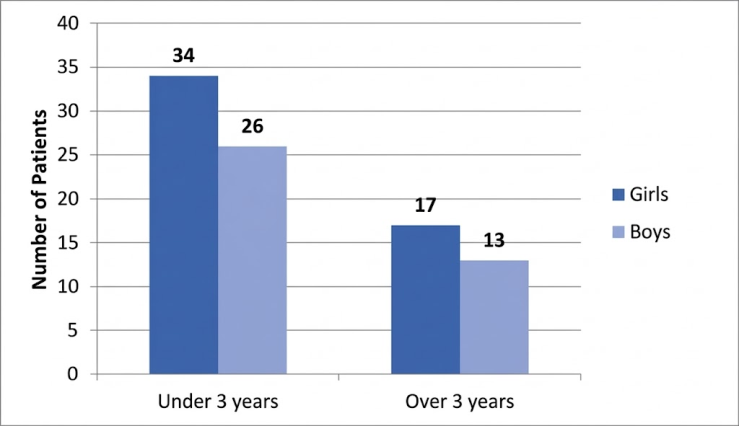 | Figure 1. Distribution of patients by gender and age groups |
|
|
|
|
|
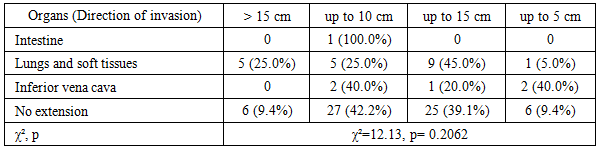
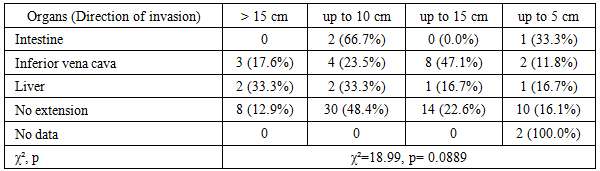
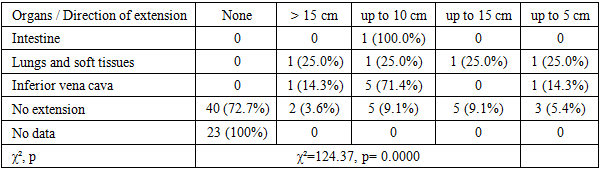
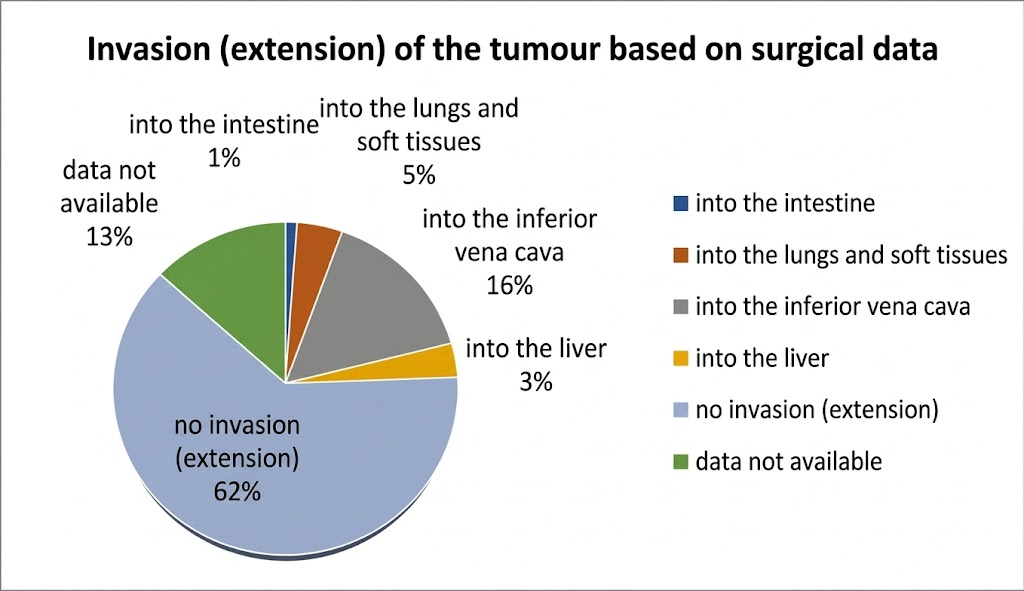 | Figure 2. Distribution of tumor extension directions according to the surgical protocol in patients with nephroblastoma (n=90) |
|
4. Discussion
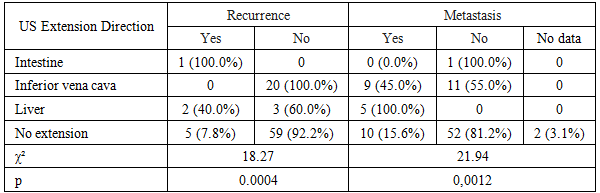
- The findings of this study reflect the clinicomorphological characteristics of nephroblastoma in pediatric patients who underwent comprehensive multimodal treatment. The observed predominance of cases in children under 3 years of age and the female preponderance align with the well-established global epidemiology of the disease [2].According to US data, the largest tumor dimensions were recorded in cases involving inferior vena cava infiltration; however, these differences did not reach statistical significance. Similar trends in tumor size assessment via MSCT are corroborated by the work of Vorobyov et al. (2017), which noted a relative advantage of MSCT in evaluating tumor volume, though not specifically in stratifying the degree of invasiveness.Our findings are consistent with the conclusions of Smith et al. (2015), who reported that MRI is superior to other imaging modalities in identifying soft-tissue and vascular invasion in pediatric renal tumors. Their results, obtained in a cohort of 72 children, demonstrated a sensitivity of 90% for this modality.The zero specificity observed for US, MSCT, and MRI in the current study may be attributed to the absence of true-negative cases within the cohort, as well as the diagnostic tendency toward overdiagnosis even when only minimal signs of invasion are present. These results align with the broader concept of early imaging as a critical component of surgical planning for renal malignancies [Vujanic et al., 2011; 13].
5. Conclusions
- The conducted analysis demonstrated that despite the high sensitivity of all three imaging modalities—US, MSCT, and MRI—in detecting the invasive growth of nephroblastoma, none possess sufficient specificity to reliably exclude extension. MRI demonstrated the highest prognostic informativeness regarding the assessment of vascular and soft-tissue invasion, particularly in cases involving the inferior vena cava and extrarenal structures.Nevertheless, the instances of overdiagnosis highlight the necessity of mandatory correlation of imaging data with the clinicomorphological profile and intraoperative findings. The findings underscore the importance of a comprehensive approach to nephroblastoma diagnostics and confirm that the evaluation of tumor extension must rely on a combination of imaging data and intraoperative verification when formulating surgical and therapeutic strategies.
References
| [1] | Chung E.M., Graeber A.R., Conran R.M. Wilms Tumor: Modern Imaging and Clinical Perspective. Radiographics. 2010; 30(5): 1257–1271. |
| [2] | Davidoff A.M., Giel D.W., Jones D.P. et al. The feasibility and outcome of nephron-sparing surgery for children with bilateral Wilms tumor: a report from the Children's Oncology Group. Cancer. 2009; 115(18): 4258–4266. |
| [3] | Dome J.S., Fernandez C.V., Mullen E.A., et al. Children's Oncology Group's 2013 blueprint for research: renal tumors. Pediatr Blood Cancer. 2013; 60(6): 994–1000. |
| [4] | Green D.M., Breslow N.E., Beckwith J.B., et al. Effect of age at diagnosis on the prognosis of patients with Wilms’ tumor. J Clin Oncol. 2019; 37(6): 531–540. |
| [5] | Grundy P.E., Breslow N.E., Li S., et al. Prognostic factors for children with recurrent Wilms’ tumor: results from the Second and Third National Wilms’ Tumor Study. J Clin Oncol. 2005; 23(29): 7312–7321. |
| [6] | Howlader N., Noone A.M., Krapcho M., et al. SEER Cancer Statistics Review, 1975–2020. National Cancer Institute. https://seer.cancer.gov. |
| [7] | National Cancer Institute. Childhood Wilms Tumor Treatment (PDQ®)–Health Professional Version. Updated July 2023. |
| [8] | Rivera M.N., Haber D.A. Wilms’ tumour: connecting tumorigenesis and organ development in the kidney. Nat Rev Cancer. 2005; 5(9): 699–712. |
| [9] | Rogowski W., Vujanić G.M., Stoneham S.J., et al. Imaging-based risk stratification in Wilms tumour: are we there yet? Eur J Cancer. 2013; 49(14): 3063–3067. |
| [10] | Scott R.H., Stiller C.A., Walker L., Rahman N. Syndromes and constitutional chromosomal abnormalities associated with Wilms tumour. J Med Genet. 2006; 43(9): 705–715. |
| [11] | Shamberger R.C., Ritchey M.L., Haase G.M., et al. Surgical complications after nephrectomy for Wilms’ tumor: report from the National Wilms Tumor Study Group. J Am Coll Surg. 2001; 192(1): 63–68. |
| [12] | Smets A.M., van Rijn R.R., Stoker J., et al. Imaging children with Wilms tumor: results of a SIOP imaging survey. Pediatr Blood Cancer. 2008; 50(2): 247–250. |
| [13] | Spreafico F., Bellani F.F., Terenziani M. Wilms tumor: current therapeutic issues and future perspectives. Pediatr Hematol Oncol. 2020; 37(6): 463–476. |
| [14] | Varan A., Akyüz C., Kutluk T., et al. Prognostic significance of radiological findings in Wilms tumor. Pediatr Hematol Oncol. 2007; 24(5): 373–380. |
| [15] | Vujanić G.M., Sandstedt B., Harms D., et al. Revised International Society of Paediatric Oncology (SIOP) working classification of renal tumors of childhood. Med Pediatr Oncol. 2002; 38(2): 79–82. |
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML