-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 1760-1763
doi:10.5923/j.ajmms.20261604.44
Received: Mar. 17, 2026; Accepted: Apr. 2, 2026; Published: Apr. 11, 2026

T786C of the eNOS Gene in the Development of Chronic Obstructive Pulmonary Disease in a Comorbid Status Importance
Tilloeva Sh. Sh.1, Rakhimova D. A.2, Hamidova M. H.1
1Bukhara State Medical Institute, Bukhara, Uzbekistan
2Republican Specialized Scientific and Practical Medical Center of Therapy and Rehabilitation, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The article presents an analysis of modern research on programming and positional mapping methods in the study of genetic determinants of chronic obstructive pulmonary disease. The frequency of alleles and genotypes of the eNOS gene (T786C) polymorphism in the presence of this disease in a comorbid state was analyzed. Also, based on the results of the study, the views of leading scientists on the methods of processing and analyzing genomic data obtained using programming tools and the use of genome-wide analysis of these methods to identify predisposition to the disease and the use of new treatment methods are expressed.
Keywords: Chronic obstructive pulmonary disease, eNOS gene (T786C) polymorphism, Heterozygous, Homozygous, Allele gene, Genotype, Arterial hypertension
Cite this paper: Tilloeva Sh. Sh., Rakhimova D. A., Hamidova M. H., T786C of the eNOS Gene in the Development of Chronic Obstructive Pulmonary Disease in a Comorbid Status Importance, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 1760-1763. doi: 10.5923/j.ajmms.20261604.44.
1. Introduction
- Chronic obstructive pulmonary disease (COPD) is one of the leading causes of disability and mortality worldwide, and its incidence is increasing day by day. In recent years, Uzbekistan, among developed countries, has been experiencing an increase in the prevalence and mortality of chronic obstructive pulmonary disease among respiratory and lung diseases [1,2]. According to the World Health Organization, COPD is one of the most common and disabling diseases of the 21st century. This disease has a significant impact not only on health, but also on the economic and social spheres [3,4,5].Its prevalence among the adult population can reach 14.9% in European countries, 15.3% in the Russian Federation, 17.4% in Uzbekistan, and even 20% in some countries. According to JCCT data, RI of the upper respiratory tract (respiratory infections) took the 3rd and 4th place in the developed countries in terms of death rate between 2015-2019. In countries with pact economic opportunities, eca, y took the 1st place [6,7].Currently, the role of genetic factors in the development of SCD remains one of the most complex problems in medical genetics [12,13]. SLE is considered to be a multifactorial disease, and therefore identifying genetic factors in the course and development of the disease plays an important role in preventing the consequences of the disease [8,9,10].Modern views on genetics show how closely the concepts of susceptibility or hereditary predisposition to diseases are related to the development of diseases [11,12,13,14]. Today, thanks to the success of molecular genetics and the development of the concept of positional and candidate mapping, it is possible to determine the localization and characterization of specific gene polymorphisms responsible for the formation of a predisposition to certain bronchial obstructive diseases [16,17]. The last decades have been marked by great achievements in the diagnosis and treatment of COPD [15,18].The presence of comorbidities such as cardiovascular, metabolic, and oncological diseases in patients with SLE can negatively affect the clinical course of the disease, leading to a worsening prognosis and a decrease in quality of life [19]. Scientific sources indicate that the relationship between SLE and these comorbidities is associated with a prolonged systemic inflammatory process, in which C-reactive protein (CRP) and markers such as endothelin-1 and eNOS are involved as key pathogenetic factors [15,20].In addition to environmental factors (dust, toxins, smoking), genetic and molecular mechanisms also play an important role in the development and exacerbation of COPD [21]. In some cases, the predisposition to the disease is determined genetically. Therefore, the study of molecular-genetic indicators in patients with COPD is of great importance for early detection of the disease, the formation of an individual approach and the development of effective treatment methods.Research objective: The study included patients with chronic obstructive pulmonary disease (COPD) and patients with chronic obstructive pulmonary disease (COPD) + emphysema. and analysis of allele and genotype frequencies for the eNOS gene polymorphism (T786C) in groups of physiologically healthy people.
2. Material and Methods
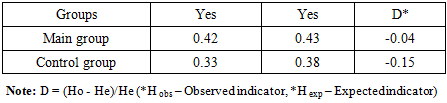
- Genetic studies were conducted on 101 patients in the main group and 95 healthy people involved in our study, and the eNOS gene (T786C) was identified, polymorphism we studied the polymorphism of the gene. In turn, we divided 101 patients from the main group into 2 groups. Group I included patients with chronic obstructive pulmonary disease 60 patients, group II was suffering from comorbid chronic obstructive pulmonary disease and arterial hypertension there were 41 patients.Inspection results. In the main group of patients, T/T homozygous genotype was 47.52%, T/S heterozygous genotype was 41.58% and S/S homozygous mutant genotype was 10.89%. In the control group, these genotypes were found proportionally at the level of 57.89%, 32.63% and 9.47%. The prevalence of T (wild type) allele was 68.32% and S (mutant) allele was 31.68% in the main group. In the control group, T and S alleles were 74.21% and 25.79%, respectively (Figure 1).
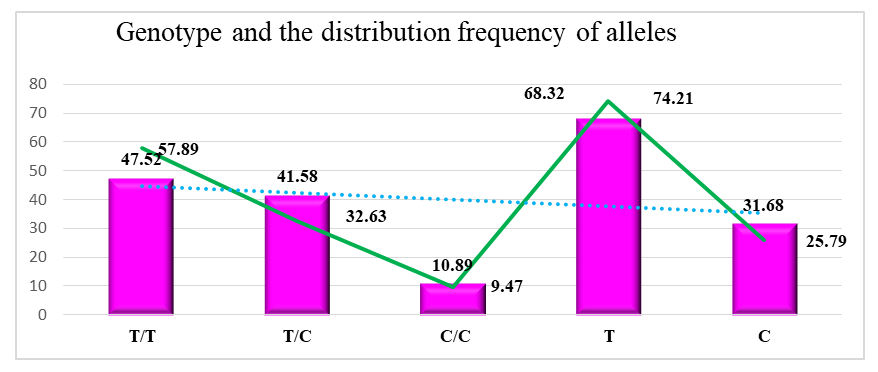 | Figure 1. In the main and control groups Distribution of eNOS gene genotypes and alleles |
|
3. Research Results
- According to the results of our study, eNOS was significantly higher in women enrolled in group II. (T786C) gene, T and S mutant alleles were detected at a frequency of 65.9% and 34.1%, respectively. In the control group taken for comparison, these indicators are corresponding It was 74.2%, 25.8%.Differences between the incidence rates of genotypes and alleles of the T786C locus in the eNOS gene in group II (n=41) and the control group (probability -control model). eNOS According to the results of the analysis of the (T786C) polymorphism, There were some differences in the prevalence of the wild-type T allele in groups I and II. In particular, the T allele was found at a frequency of 65.9% in patients in group II, while in group I this figure was 70.0%. These differences were not statistically significant. (χ2 = 0.4; r=0.72; RR =0.9; 95% CI: 0.48-1.86; OR=0.8; 95% CI: 0.45-1.51), but the slight predominance of the T allele in group I may indicate a possible protective role. Also, the mutant C allele was detected at a frequency of 30.0% in group I and 34.1% in group II. This difference is also not statistically significant (χ2 = 0.4; p=0.72; RR =1.1; 95% CI: 0.65-1.75; OR=1.2; 95% CI: 0.66-2.21), but the C allele is more common in group II The higher prevalence does not negate its association with chronic lung disease and cardiovascular disease.The predictive value of the mutant allele C of the eNOS gene in the main group of patients was AUC=0.6 3 (SE=0.74; SP=0.52; OR=1.33; 95% CI= 0.86-2.05; p=0.43). In patients in group 1, this indicator was AUC=0.62; SE=0.74; SP=0.53; OR= 1.23; 95% CI= 0.75-2.03; p = 0.58, and in patients in group 2, this indicator was AUC = 0.64; SE = 0.7 4; SP =0.54; OR= 1.49; 95% CI= 0.85-2.6; p=0.64. The mutant C allele of the eNOS gene under study may be a risk factor for the development of chronic lung diseases and cardiovascular diseases, AUC (Area Under ARGL Nerve) in the main group was AUC= 0.63, in group I AUC= 0.62, in group II AUC= 0.64. Thus, since the AUC was on average 0.6 3, the mutant C allele of the eNOS gene has a high prognostic efficiency in predicting the development of the disease.The predictive efficiency of the eNOS gene mutant C/C genotype in the main group of patients was AUC=0.71 (SE=0.71 8; SP=0.91; OR= 1.17; 95% CI= 0.46- 2.99; p=0.51). In group 1 patients, this indicator was AUC=0.61; SE=0.71; SP=0.61; OR= 1.06; 95% CI= 0.37 -3.05; p=0.39, and in group 2 patients, this indicator was AUC=0.62; SE=0.62; SP =0.91; OR= 1.33; 95% CI= 0.41 - 4.27; p=0.3. In the main group and in both subgroups, the mutant form of the genotype is S/S We can conclude that it is important in the development of chronic obstructive pulmonary disease. Thus, the eNOS gene is important as a marker for predicting genital prolapse, since its AUC is 0.65.
4. Conclusions
- The studies conducted and a number of meta-analyses have shown that our study also Statistical analyses have proven that the eNOS gene polymorphism (T786C) plays an important role in the development of chronic lung diseases and cardiovascular diseases. These are the results of our research proved that in the Uzbek population, patients with chronic lung diseases have a higher incidence of eNOS gene mutations (T786C) polymorphism We observed that the mutant homozygous genotype was more common than the control group. According to the OR and AUC data in our statistical studies, the mutant allele C and mutant C/C homozygous genotypes of the eNOS gene in the Uzbek population indicate an increased risk of disease.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML