Karimova Nargiza Sunnatillayevna1, 2
1PhD, Associate Professor, Republican Specialized Scientific-Practical Medical Center of Oncology and Radiology of the Republic of Uzbekistan
2Center for the Development of Professional Qualification of Medical Workers, Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: Karimova Nargiza Sunnatillayevna, PhD, Associate Professor, Republican Specialized Scientific-Practical Medical Center of Oncology and Radiology of the Republic of Uzbekistan.
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
Background. Malignant glial brain tumors are characterized by high aggressiveness, significant disability, and poor prognosis. In the Republic of Uzbekistan, the incidence in 2023 was 3.3 per 100,000 population, ranking sixth among all oncological pathologies. Objective. To identify the clinico-morphological and demographic characteristics of malignant gliomas based on the example of patients treated at the Republican Specialized Scientific-Practical Medical Center of Oncology and Radiology. Materials and Methods. Data from 174 patients diagnosed with WHO grade G2–G4 gliomas who underwent neurosurgical intervention and radiotherapy were analyzed. Statistical, morphological, and clinical-diagnostic methods were employed. Results. The majority of patients were males (55.75%) aged 20–39 years. The frontal, temporal, and parietal lobes were the most commonly affected regions. The predominant histological type was anaplastic astrocytoma (68.97%), with grade G3 being most prevalent (73.56%). In 66% of cases, patients sought medical attention within the first six months of symptom onset. The main surgical approach was subtotal resection (75.3%). Conclusion. The disease demonstrates distinct demographic and clinico-morphological patterns. Efforts should focus on early detection and personalized therapeutic strategies.
Keywords:
Malignant gliomas, Brain, Uzbekistan, Astrocytoma, Glioblastoma
Cite this paper: Karimova Nargiza Sunnatillayevna, Clinico-Morphological and Demographic Profile of Brain Tumors: A Comprehensive Analysis Based on Rsspmcor Data, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 1754-1759. doi: 10.5923/j.ajmms.20261604.43.
1. Introduction
Brain tumors represent one of the most complex nosological entities in oncology, both in terms of their clinical presentation and in the challenges associated with diagnosis, treatment, and prognosis. Malignant glial tumors occupy a leading position among primary tumors of the central nervous system (CNS), accounting, according to various sources, for 40% to 60% of all intracranial neoplasms in adults [8,10]. According to data from the Central Brain Tumor Registry of the United States (CBTRUS, 2022), the annual incidence of malignant CNS tumors averages 7.1 per 100,000 population, while glioblastoma remains the most common glioma, accounting for up to 48.6% of all malignant brain tumors in adults [4]. In Europe and the CIS countries, the figures are comparable: according to EUROCARE and RUSONCO, the incidence of glial tumors ranges from 6 to 8 per 100,000 population per year [2,5]. According to data from the Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology of the Republic of Uzbekistan, in 2023 the overall incidence of malignant neoplasms was 73.5 cases per 100,000 population. In the structure of cancer incidence, the highest proportions were attributed to breast cancer (12.3 per 100,000), colorectal cancer (5.6), gastric cancer (5.3), cervical cancer (5.1), and malignant neoplasms of the bronchopulmonary system (4.6). Malignant brain neoplasms ranked sixth, with an incidence of 3.3 cases per 100,000 population [1]. Despite the relatively low proportion of malignant brain tumors in the overall structure of cancer incidence, these pathologies are of particular clinical and social significance. This is обусловлено their predominant localization in functionally important regions of the CNS, the high degree of patient disability they cause, the difficulties of early diagnosis, and the limited therapeutic potential. The modern classification of glial tumors proposed by the World Health Organization (WHO) in 2021 has substantially changed the approach to morphological and molecular verification of the diagnosis. Primary emphasis is placed on molecular markers (IDH, ATRX, 1p/19q, MGMT, TERT, and others), which makes it possible to stratify tumors according to malignancy grade and prognosis, including within the same morphological type [2]. Thus, in the new classification, glioblastoma is defined exclusively as glioblastoma, IDH-wildtype, whereas IDH-mutant forms have been reclassified into the category of grade 4 astrocytomas. Despite advances in surgery, radiotherapy, and chemotherapy, the prognosis of malignant gliomas remains extremely unfavorable: the median overall survival for glioblastoma is 12-15 months, while for grade 3 gliomas it is 2-3 years [5,6]. Thus, the need for a comprehensive analysis of the clinical, morphological, and demographic characteristics of malignant gliomas, taking into account current concepts of morphogenesis and molecular biology, determines the high relevance of the present study, especially in the context of practical healthcare in Uzbekistan. Objective. To perform a comprehensive analysis of the clinical, morphological, and demographic characteristics of malignant glial brain tumors based on data from patients treated at the Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology, in order to identify patterns in their clinical course, morphological structure, and factors influencing diagnosis and treatment.
2. Materials and Methods
The study included the clinical, morphological, and demographic data of 174 patients with verified malignant glial brain tumors who were treated at the departments of the Republican Specialized Scientific and Practical Medical Center of Oncology and Radiology and its branches. Inclusion criteria were histologically confirmed malignant glioma (grades G2-G4), complete clinical and diagnostic documentation, and comprehensive treatment including a mandatory neurosurgical stage followed by external beam radiotherapy. All patients received a course of external beam radiotherapy using Elekta Infinity and Terabalt systems, in accordance with an individualized treatment plan developed with consideration of the anatomical and topographic features of the tumor, its volume, and extent of spread. Radiotherapy was delivered in a fractionated regimen using CT- and MRI-based treatment planning, with the aim of achieving maximal sparing of healthy brain tissue. The analysis was performed with regard to the following parameters: age, sex, place of residence, tumor localization, degree of tumor differentiation, histological type, extent of surgical intervention, general condition at admission, severity of postoperative neurological deficit, and spectrum of clinical manifestations. Morphological verification of the diagnosis was carried out on the basis of histological examination of tumor material obtained during surgery or stereotactic biopsy, using standard staining methods (hematoxylin and eosin) and, when necessary, immunohistochemical analysis. Histological classification of tumors was performed in accordance with WHO recommendations. The clinical assessment included anamnesis data, results of neuroimaging studies (MRI with and without contrast enhancement), clinical and neurological examination findings, as well as laboratory and instrumental investigations. The general condition of patients at admission was assessed using a clinical severity scale. Statistical analysis was performed using descriptive and analytical statistical methods. Absolute and relative frequencies, mean values, standard errors (SE), and 95% confidence intervals (CI) were calculated. The critical level of significance was set at p < 0.05.
3. Results of the Study
The retrospective and prospective analysis of the clinical, morphological, demographic, and epidemiological characteristics of malignant glial brain tumors in 174 patients made it possible to identify significant patterns. The principal statistically significant findings for each analytical domain are presented below.The highest number of tumors was recorded in the 20-29 and 30-39 year age groups, confirming the high incidence of these neoplasms in young and middle-aged individuals. The gradual decrease in frequency in older age cohorts may be associated with diagnostic limitations, comorbidity burden, and natural selection. The differences between age groups were statistically significant (p < 0.0001) (Table 1).Table 1. Distribution of Patients by Age
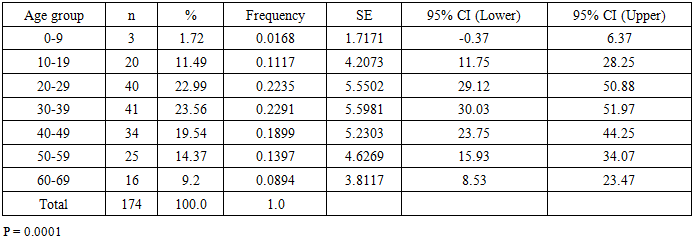 |
| |
|
Male patients predominated (55.75%), which may reflect sex-related differences in the pathogenesis of central nervous system tumors (Table 2).Table 2. Distribution of Patients by Sex and Their Mean Age
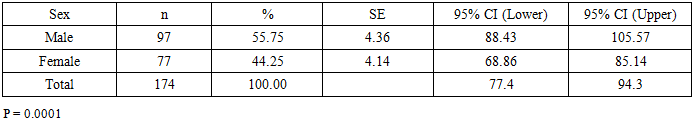 |
| |
|
Urban residents accounted for 60.34% of the cohort, which may be explained both by greater accessibility of oncological care and by the potential influence of urban-related risk factors. The differences by place of residence were statistically significant (p = 0.0001) (Table 3).Table 3. Distribution of Patients According to Place of Residence
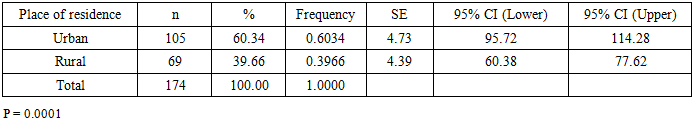 |
| |
|
The frontal (25.86%), temporal (21.84%), and parietal (17.24%) lobes were the most frequently affected anatomical sites. These regions are characterized by high metabolic activity and major functional importance, which may render them more vulnerable to oncogenesis. Tumors of the deep brain structures and brainstem were less common but were associated with a poorer prognosis (Table 4).Table 4. Distribution of Patients by Anatomical Tumor Localization
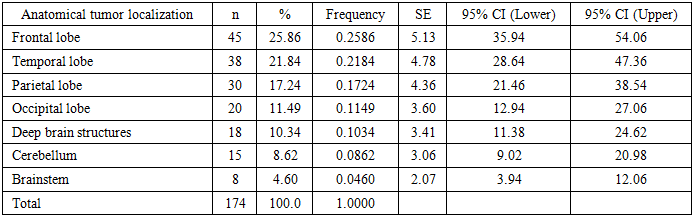 |
| |
|
Grade G3 tumors predominated (73.56%), demonstrating a moderate degree of differentiation and aggressive biological behavior. Grade G4 tumors (17.82%) represented highly aggressive forms associated with an unfavorable prognosis (Table 5).Table 5. Distribution of Patients According to Tumor Differentiation Grade
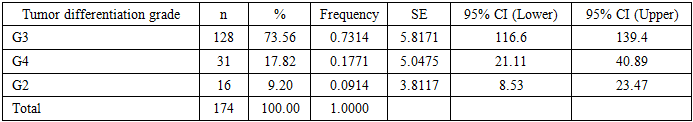 |
| |
|
Anaplastic astrocytoma (68.97%) and glioblastoma (12.64%) predominated in the histological structure, reflecting the epidemiological significance of the astrocytic lineage of tumor growth (Table 6).Table 6. Distribution of Patients by Histological Group
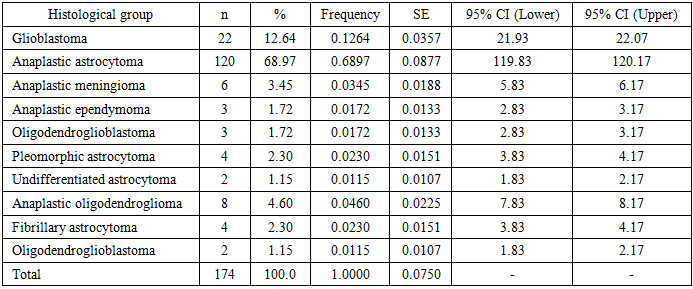 |
| |
|
В In 66.09% of cases, patients sought medical attention within the first 6 months after the onset of symptoms; however, 15% presented only after more than 6 months. This indicates the presence of both clinically overt and subclinical forms of the disease and underscores the need for increased diagnostic vigilance (Table 7).Table 7. Duration of Medical History Before the First Visit to a Physician
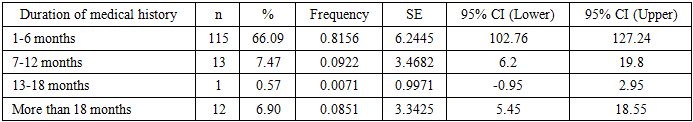 |
| |
|
The principal surgical approach was subtotal resection (75.3%), representing a compromise between oncological radicality and preservation of neurological function. The frequencies of partial and total resections (6.9% and 17.8%, respectively) were determined by individual anatomical and clinical factors (Table 8).Table 8. Extent of Surgery Prior to Radiotherapy
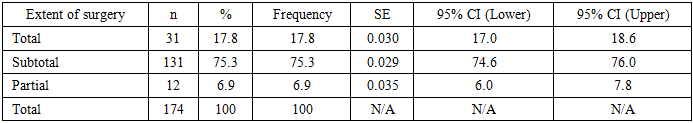 |
| |
|
Patients in moderate general condition predominated (46.55%), which is typical for oncology patients at the preoperative stage. However, 18.39% demonstrated clinical deterioration over time, underscoring the need for early intervention (Table 9).Table 9. Distribution of Patients According to General Condition at Admission
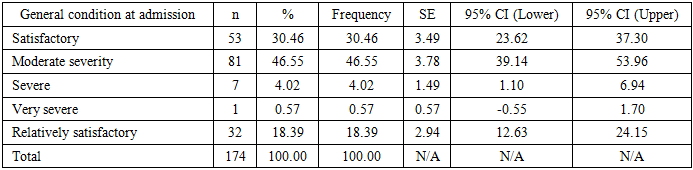 |
| |
|
The most common complaints were headache (21.7%) and seizures (11.3%), reflecting intracranial hypertension and cortical irritation. Focal neurological symptoms, as well as cognitive and autonomic disturbances, also played a significant role in the clinical presentation of the disease (Table 10).Table 10. Clinical Distribution of Patient Complaints at Hospital Admission
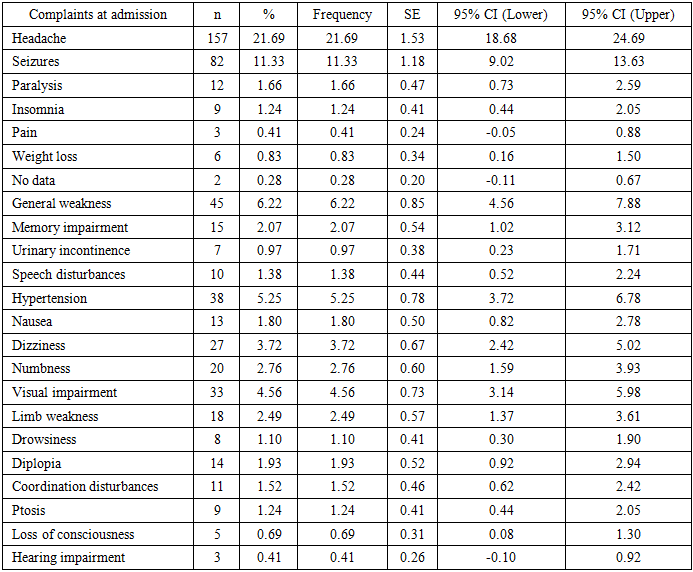 |
| |
|
4. Discussion
The findings obtained in the present study confirm the importance of a comprehensive approach to the analysis of the clinical, morphological, and demographic characteristics of malignant glial tumors of the brain. The patient cohort was predominantly composed of young and middle-aged individuals, mainly males, which is consistent with the results of a number of epidemiological studies. Thus, according to the analytical review by Ostrom et al., the peak incidence of gliomas is observed in the age groups of 35-44 and 45-54 years, with a moderate male predominance [10]. At the same time, the shift of the age peak toward younger cohorts (20-39 years) identified in the present study may be related to regional demographic and environmental characteristics, as well as differences in access to medical care.The sex asymmetry with male predominance (55.75%) also confirms the existing literature data, according to which the male sex is associated with a higher frequency of high-grade gliomas, including glioblastomas [4]. It has been suggested that sex-related differences in the expression of gonadotropin receptors and sex hormones may play a role in central nervous system carcinogenesis, as noted by McKinney [9]. The predominance of patients living in urban areas (60.34%) may indicate not only better detection of tumors in this group, but also the possible influence of urbanization-related factors, such as radiation exposure, electromagnetic fields, and environmental pollution, which is also supported by the findings of Huss et al. and Zarnescu et al. [7,15].The anatomical localization of tumors predominantly in the frontal, temporal, and parietal lobes is in agreement with studies indicating the selective vulnerability of these regions to mutation-mediated oncogenesis [8]. At the same time, tumors of the deep brain structures and the brainstem, although less common, are associated with a more severe clinical course and limited surgical options, which necessitates a comprehensive multidisciplinary approach. The predominance of grade G3 (73.56%) and G4 (17.82%) tumors reflects the general trend toward delayed diagnosis and high proliferative activity of central nervous system tumors. The high proportion of anaplastic astrocytomas (68.97%) indicates the predominance of tumors of intermediate-to-high malignancy requiring aggressive combined treatment.The results of the meta-analysis by Hervey-Jumper et al. also emphasize that survival in patients with G3 gliomas varies substantially depending on the extent of resection and the molecular profile of the tumor [6]. Subtotal resection was the predominant type of surgical intervention (75.3%), reflecting the effort to maintain a balance between oncological radicality and preservation of neurological function. This approach is supported by the EANO 2021 clinical guidelines, which emphasize that complete resection is feasible only when the risk of neurological deficits is minimal [13]. Clinical manifestations such as headache, epileptic seizures, focal neurological deficits, and cognitive disturbances correspond to the typical neuro-oncological profile and are consistent with the findings of Yordanova et al. (2018), who demonstrated the dependence of clinical symptomatology on the anatomical zone involved [14].Thus, the results of the present study are consistent with contemporary international data and underscore the need for timely diagnosis, an individualized treatment approach, and multidisciplinary collaboration in the management of patients with glial tumors of the brain.
5. Conclusions
The present study made it possible to identify significant demographic, clinical, and morphological patterns of malignant glial tumors of the brain. The disease was found to occur more frequently in young and middle-aged individuals, predominantly in males, with a higher prevalence among the urban population. The most common anatomical localizations were the frontal, temporal, and parietal lobes, while the histological spectrum was represented mainly by anaplastic astrocytomas and glioblastomas. The predominance of G3 tumors underscores the aggressive nature of most of these neoplasms and highlights the need for comprehensive treatment with the highest possible oncological radicality while preserving neurological function.Early presentation was observed in the majority of patients; however, the presence of a substantial proportion of cases diagnosed at a later stage indicates the need to improve oncological alertness among primary care specialists. The findings emphasize the importance of a multidisciplinary and personalized approach to the management of this patient population, as well as the need to further refine strategies for early detection and differential diagnosis of central nervous system tumors.
References
| [1] | “The State of Oncological Care for the Population of the Republic of Uzbekistan in 2023” / edited by M.N. Tillyashaykhov, Sh.N. Ibragimov, S.M. Djanklich. Tashkent: IPTD “Uzbekistan”, 2024. 184 p. |
| [2] | RUSONCO: Russian Cancer Research Center. Analytical Materials on Cancer Incidence. 2022. |
| [3] | Balducci L., Extermann M. Management of cancer in the older person: A practical approach. The Oncologist, 2020, 25(2), 115–123. |
| [4] | CBTRUS Statistical Report. Neuro-Oncology, 2022, 24(Suppl 5), v1–v95. |
| [5] | EUROCARE-5 Working Group. European cancer registry–based study on survival and care of cancer patients. Lancet Oncology. 2015. |
| [6] | Hervey-Jumper S.L., Berger M.S. Role of surgical resection in low- and high-grade gliomas. Current Treatment Options in Neurology, 2019, 21(8), 34. |
| [7] | Huss A. et al. Environmental risk factors and brain tumors: A review. Current Environmental Health Reports, 2022, 9(1), 35–49. |
| [8] | Louis D.N. et al. The 2021 WHO Classification of Tumors of the Central Nervous System: a summary. Acta Neuropathol, 2021, 142(1), 11–13. |
| [9] | McKinney P.A. Central nervous system tumors in children: epidemiology and risk factors. Bioelectromagnetics Suppl, 2004, 7, S60–S68. |
| [10] | Ostrom QT, et al. CBTRUS Statistical Report: Primary brain and other CNS tumors diagnosed in the United States, 2015–2019. Neuro-Oncology. 2021. |
| [11] | Stupp R, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005; 352: 987–996. |
| [12] | Turgut M. et al. Diagnostic delay in brain tumors: a multidisciplinary problem. Surgical Neurology International, 2020, 11, 180. |
| [13] | Weller M. et al. EANO guidelines on the diagnosis and treatment of diffuse gliomas of adulthood. Lancet Oncology, 2021, 22(9), e370–e384. |
| [14] | Yordanova Y.N. et al. Cognitive and neurological impairment in adult brain tumor patients. Neuropsychology Review, 2018, 28(4), 408–420. |
| [15] | Zarnescu R. et al. Urban environmental exposures and brain tumor risk: a systematic review. Environmental Research, 2023, 220, 114919. |


 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML








