-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 1701-1704
doi:10.5923/j.ajmms.20261604.32
Received: Mar. 4, 2026; Accepted: Mar. 28, 2026; Published: Apr. 8, 2026

Care Tactics for Children with Primary Congenital Glaucoma with Sturge-Weber Syndrome
Turakulova Dilfuza Mukhitdinovna1, Nazirova Zulfiya Rustamovna2, Karabaeva Iroda Murodzhonovna3
1Candidate of Medical Sciences, Department of Ophthalmology, Pediatric Ophthalmology, Associate Professor, Tashkent State Medical University, Uzbekistan
2Doctor of Medical Sciences, Department of Ophthalmology, Pediatric Ophthalmology, Associate Professor, Tashkent State Medical University, Uzbekistan
3Master's Student, Department of Ophthalmology, Pediatric Ophthalmology, Tashkent State Medical University, Uzbekistan
Correspondence to: Turakulova Dilfuza Mukhitdinovna, Candidate of Medical Sciences, Department of Ophthalmology, Pediatric Ophthalmology, Associate Professor, Tashkent State Medical University, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The article presents a retrospective and prospective analysis of the examination and treatment outcomes of 21 children with primary congenital glaucoma associated with Sturge–Weber syndrome. Clinical and instrumental features of the disease course are analyzed, including vascular abnormalities of the anterior chamber angle and episcleral vessels and their impact on intraocular pressure. The effectiveness of a combined surgical approach aimed at simultaneous influence on the main aqueous humor outflow pathways, along with preoperative hemostatic preparation, is evaluated. The results demonstrate that the proposed surgical technique provides effective intraocular pressure control in most patients; however, it is associated with a high risk of prolonged postoperative hypotony and choroidal complications. The study concludes that management of glaucoma in children with Sturge–Weber syndrome requires an individualized surgical strategy and careful postoperative monitoring.
Keywords: Sturge–Weber syndrome, Port-wine stain, Etamsylate, Maklakov tonometry, iCare 100 tonometry
Cite this paper: Turakulova Dilfuza Mukhitdinovna, Nazirova Zulfiya Rustamovna, Karabaeva Iroda Murodzhonovna, Care Tactics for Children with Primary Congenital Glaucoma with Sturge-Weber Syndrome, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 1701-1704. doi: 10.5923/j.ajmms.20261604.32.
Article Outline
1. Relevance
- Sturge-Weber syndrome (encephalotrigeminal angiomatosis) is a rare congenital neurocutaneous disorder often associated with glaucoma in children. [1,5]Sturge-Weber syndrome is a rare congenital disorder, occurring in approximately 1 in 20,000–50,000 live births. Glaucoma is observed in a significant proportion of patients with this syndrome—according to the literature, in 30–70% of cases. [3,6,7]Glaucoma in this syndrome is characterized by early onset, a progressive course, and a high probability of irreversible vision loss already in childhood. According to clinical observations, glaucoma in Sturge-Weber syndrome is detected in a significant proportion of patients and is often resistant to standard treatment methods. [4,8,10]A characteristic feature of glaucoma associated with Sturge-Weber syndrome is its complex pathogenesis, associated with developmental anomalies of the anterior chamber angle, increased episcleral venous pressure, and vascular malformations. This results in the low effectiveness of conservative therapy and a high complication rate after surgical interventions. Furthermore, in young children, diagnosis and monitoring of intraocular pressure are difficult, leading to late detection of the disease and a worse prognosis. [9,11,12]Despite the existence of various medical and surgical approaches to the treatment of glaucoma associated with Sturge-Weber syndrome, there are still no uniform standards for the care of these patients, and treatment outcomes remain variable. The high risk of progression of glaucomatous optic neuropathy, the need for repeated surgeries, and long-term follow-up emphasize the social and medical significance of this problem. [13,14,15]In this regard, an analysis of current glaucoma treatment methods in children with Sturge-Weber syndrome is relevant and necessary. This will allow us to evaluate the effectiveness of various therapeutic strategies, determine optimal patient management tactics, and improve functional outcomes, helping to preserve visual function and improve children's quality of life. [2]The aim of the study is to examine current approaches to the treatment of glaucoma in children with Sturge-Weber syndrome, evaluate their effectiveness, and determine optimal management tactics.
2. Materials and Methods
- A retrospective and prospective study was conducted involving 21 children diagnosed with Sturge–Weber syndrome and associated primary congenital glaucoma. The study was carried out at the Department of Ophthalmology, Tashkent State Medical University, during the period from 2018 to 2025.The sample included 12 boys (55%) and 9 girls (45%). According to age distribution, 16 patients (76%) were infants, 4 (19%) were young children, and 1 (5%) was of preschool age.Inclusion criteria: confirmed diagnosis of Sturge–Weber syndrome with glaucoma.Exclusion criteria: absence of complete clinical data or follow-up.All patients underwent comprehensive ophthalmological and general clinical examination, including: visometry; biomicroscopy; ophthalmoscopy; gonioscopy; echobiometry; Maklakov tonometry; iCare 100 tonometry; tonography. Magnetic resonance imaging (MRI) of the brain was performed to assess vascular abnormalities.Tonometry was performed under general anesthesia 3–5 minutes after induction. Premedication included diphenhydramine 1% and atropine sulfate 0.1%. During induction, a combination of 0.5% sibazon, 40% sodium oxybutyrate (GHB), and 0.005% fentanyl was used.All children underwent antiglaucoma surgery developed in our department using a combined method that involves simultaneous interventions on the outflow pathways in three directions: Burian sinusotrabeculotomy into the scleral sinus, cyclodialysis-cycloretraction with an autoscleral pedicle into the suprachoroidal space, and basal iridectomy with sclerectomy under the scleral flap into the episcleral venous system (Patent for Invention "Method for Surgical Treatment of Congenital Glaucoma" No. IAP 04890 dated May 12, 2014).Ethical considerations. The study was conducted in accordance with the principles of the Declaration of Helsinki. Participation was voluntary. Written informed consent was obtained from the parents or legal guardians of all patients. Patient confidentiality and anonymity were strictly maintained.Statistical analysis was performed using standard statistical methods. Quantitative data are presented as mean ± standard deviation.
3. Results
- The study included 21 patients (12 boys and 9 girls). The majority were infants (76%). All patients had a congenital capillary malformation of the facial skin (nevus flammeus), localized predominantly to one side of the face. The lesion appears as a flat, pink-red spot with indistinct borders, without signs of infiltration or elevation.The coloration is relatively uniform, with a tendency to increase in intensity in the central areas.The lesion extends into the innervation zone of the first (ophthalmic) and partially the second (maxillary) branches of the trigeminal nerve, including the periorbital area and buccal surface. The skin in the affected area is smooth, without scaling or ulceration.
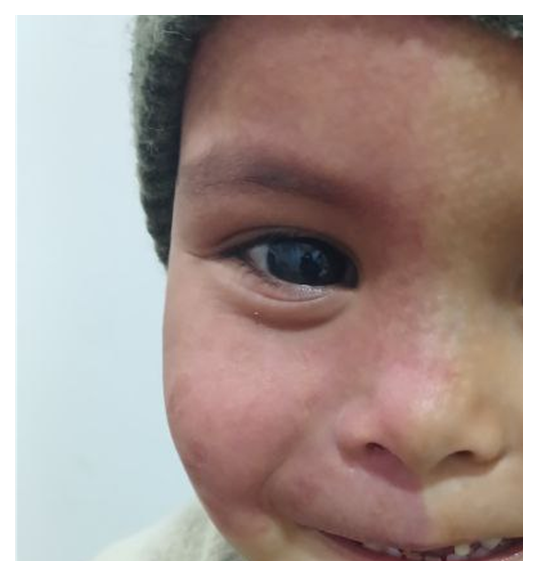 | Figure 1. Congenital capillary malformation of the facial skin (nevus flammeus), localized predominantly in the area of one half of the face |
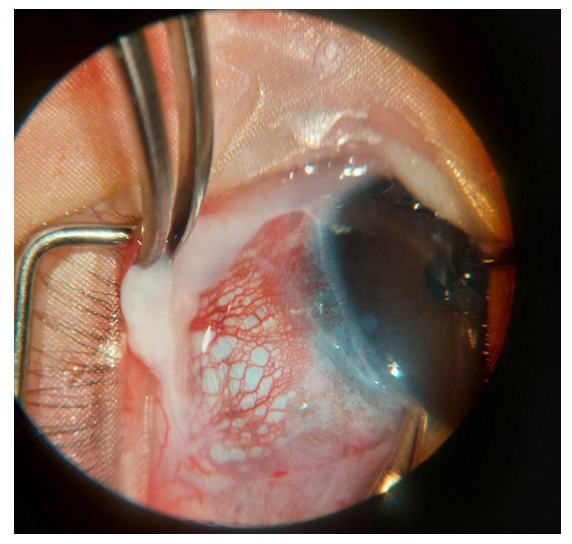 | Figure 2. Intraoperative image of the scleral surface in a patient with Sturge-Weber syndrome |
4. Discussion
- The aim of this study was to evaluate the effectiveness of surgical management of glaucoma in children with Sturge–Weber syndrome. The results demonstrated that the combined surgical approach provides effective intraocular pressure control in most cases. However, a high incidence of postoperative hypotony and choroidal complications was observed. These findings are consistent with previous studies reporting high surgical complexity and complication rates in Sturge–Weber-associated glaucoma [6,7]. However, some authors report lower rates of postoperative hypotony, which may be related to differences in surgical techniques. The strengths of this study include a combined retrospective and prospective design and detailed clinical analysis. Limitations include a relatively small sample size and the absence of long-term follow-up in some patients.
5. Conclusions
- The study demonstrated that combined surgical treatment is effective in controlling intraocular pressure in children with Sturge–Weber syndrome.From a healthcare perspective, early diagnosis and individualized surgical strategy are essential to improve visual outcomes and reduce complications.It is recommended to ensure careful postoperative monitoring and further research with larger patient groups.
ACKNOWLEDGEMENTS
- The authors would like to thank the staff of the ophthalmology department for their assistance.
Financial Support and Sponsorship
- None.
Author Contributions
- T.D.M. – study design, supervisionN.Z.R. – data analysis, manuscript editingK.I.M. – data collection, manuscript preparation
Conflict of Interest
- The authors declare no conflict of interest.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML