-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 1696-1700
doi:10.5923/j.ajmms.20261604.31
Received: Mar. 4, 2026; Accepted: Mar. 23, 2026; Published: Apr. 8, 2026

The Role and Place of Immunological Factors in the Structure of Thrombotic Complications After Percutaneous Coronary Interventions
Erkulov Kodirjon Odilovich1, Umarov Bakhtiyorjon Yadgarovich2
1Navoi Regional Multidisciplinary Medical Center, Uzbekistan
2National Children’s Medical Center, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Thrombotic complications after percutaneous coronary interventions (PCI), particularly stent thrombosis, remain clinically significant despite advances in stent technology and antithrombotic therapy. Growing evidence suggests that thrombosis after PCI represents not only a hemostatic disorder but also a thromboinflammatory phenomenon involving innate immune activation, cytokine imbalance, endothelial dysfunction, and platelet–leukocyte interactions. This study aimed to determine the role of immunological factors in the structure of early and late thrombotic complications following PCI in patients with ischemic heart disease (IHD). A comprehensive analysis of coagulation parameters, pro- and anti-inflammatory cytokines (IL-6, TNF-α, IL-10), endothelial activation markers, and platelet indices was performed. Patients who developed stent thrombosis demonstrated a distinct immunohemostatic phenotype characterized by elevated IL-6 (↑52%), TNF-α (↑38%), D-dimer (↑61%), fibrinogen (↑27%), reduced IL-10 (↓29%), and enhanced platelet reactivity compared with uncomplicated PCI cases (p<0.05). An integrated phenotypic risk index (PRI-PCI) was developed, demonstrating high predictive accuracy (AUC=0.84; 95% CI 0.78–0.90). Stent thrombosis represents a specific thromboinflammatory phenotype rather than an isolated coagulation disorder. Integration of immunological markers into preprocedural risk assessment improves early identification of high-risk patients.
Keywords: Percutaneous coronary intervention, Stent thrombosis, Thromboinflammation, Cytokines, IL-6, IL-10, TNF-α, Endothelial dysfunction, Platelet activation, Immune-hemostatic phenotype, Risk prediction
Cite this paper: Erkulov Kodirjon Odilovich, Umarov Bakhtiyorjon Yadgarovich, The Role and Place of Immunological Factors in the Structure of Thrombotic Complications After Percutaneous Coronary Interventions, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 1696-1700. doi: 10.5923/j.ajmms.20261604.31.
Article Outline
1. Relevance of the Study
- Ischemic heart disease (IHD) remains the leading cause of morbidity and mortality worldwide, accounting for millions of deaths annually and representing a major socioeconomic burden. Percutaneous coronary intervention (PCI) has become the cornerstone of myocardial revascularization in both acute coronary syndromes and stable coronary artery disease [2]. The widespread implementation of drug-eluting stents (DES), improved implantation techniques, and contemporary dual antiplatelet therapy have significantly reduced the incidence of restenosis and early thrombotic events. Nevertheless, stent thrombosis continues to occur in 0.5–1.0% of patients annually and is associated with high mortality rates reaching 20–40% in acute presentations [5,7,9]. Even though its absolute incidence is relatively low, the clinical consequences are often catastrophic, including recurrent myocardial infarction, cardiogenic shock, and sudden cardiac death [6].Traditional risk assessment strategies after PCI primarily focus on procedural and anatomical parameters, such as lesion complexity, stent length, bifurcation involvement, and SYNTAX score, as well as clinical factors including diabetes mellitus, renal dysfunction, and reduced left ventricular ejection fraction. However, these models do not fully explain interindividual variability in outcomes. In real-world practice, patients with comparable angiographic findings and technically successful stent deployment may demonstrate dramatically different postoperative courses. While some remain event-free, others develop early or late stent thrombosis despite optimal adherence to antithrombotic therapy. This discrepancy suggests that additional biological determinants influence thrombotic risk [10].Recent advances in cardiovascular research highlight the concept of thromboinflammation as a central mechanism underlying post-PCI complications. Endothelial injury induced by stent implantation triggers not only activation of the coagulation cascade but also innate immune responses involving neutrophils, monocytes, and pro-inflammatory cytokines. Elevated levels of interleukin-6 (IL-6), tumor necrosis factor-alpha (TNF-α), and other inflammatory mediators promote tissue factor expression, enhance platelet reactivity, suppress endogenous fibrinolysis, and contribute to endothelial dysfunction. Simultaneously, inadequate anti-inflammatory regulation, particularly reduced interleukin-10 (IL-10) activity, may amplify this prothrombotic environment. The interplay between immune activation and hemostatic imbalance forms a unified thromboinflammatory cascade rather than isolated pathophysiological events [8].Despite accumulating evidence, most previous investigations have examined isolated biomarkers without integrating immunological and coagulation parameters into a comprehensive predictive framework. There remains a lack of standardized models capable of quantitatively assessing immune-hemostatic phenotypes prior to PCI. Consequently, current clinical practice often relies on descriptive rather than pathogenetically grounded risk stratification [3,4,5].In the Republic of Uzbekistan and other countries with rapidly expanding interventional cardiology services, the increasing volume of PCI procedures inevitably leads to a proportional rise in the absolute number of thrombotic complications. Identification of biologically meaningful predictors within the national patient population is therefore both clinically and socially relevant. Development of an integrated immune-hemostatic risk model may improve early detection of high-risk individuals, optimize monitoring strategies, and potentially reduce morbidity and mortality associated with stent thrombosis [9].Thus, investigation of the immunological determinants of thrombotic complications after PCI represents an urgent and scientifically justified direction in modern cardiovascular medicine [1].
2. Aim of the Study
- To determine the role and structural significance of immunological factors in the development of early and late thrombotic complications after percutaneous coronary interventions in patients with ischemic heart disease, through comprehensive analysis of innate immune activation, cytokine balance, endothelial dysfunction markers, and hemostatic parameters, and to develop an integrated phenotypic risk model for prediction of stent thrombosis.
3. Materials and Methods
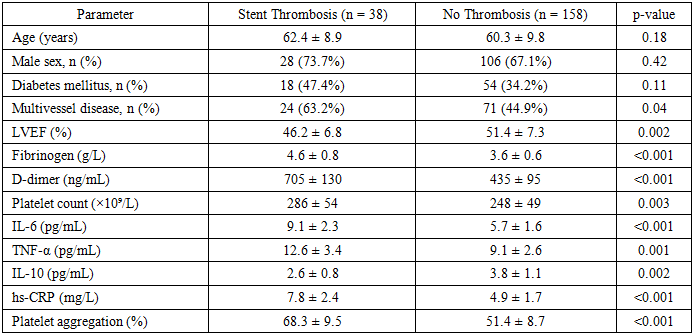
- This prospective observational study included 196 consecutive patients with ischemic heart disease (IHD) who underwent percutaneous coronary intervention (PCI) between 2023 and 2025. The study population consisted of 134 men (68.4%) and 62 women (31.6%), with a mean age of 60.8 ± 9.7 years.Patients were divided into two groups according to 12-month follow-up outcomes:• Group 1 (n = 38; 19.4%) – patients who developed definite or probable stent thrombosis;• Group 2 (n = 158; 80.6%) – patients without thrombotic complications.Early thrombosis (≤30 days) occurred in 23 patients (60.5%), and late thrombosis (>30 days) in 15 patients (39.5%). Diabetes mellitus was present in 36.7%, arterial hypertension in 72.4%, and multivessel coronary disease in 48.5% of participants.Pre-procedural laboratory assessment included:Hemostatic parameters:• Fibrinogen (Clauss method)• D-dimer (immunoturbidimetric assay)• Platelet count• Activated partial thromboplastin time (aPTT)• International normalized ratio (INR)Immunological parameters:• IL-6, TNF-α, IL-10 (ELISA; sensitivity 0.5 pg/mL)• High-sensitivity C-reactive protein (hs-CRP)• Soluble VCAM-1• CD40 ligandMean baseline values in the total cohort were: IL-6 6.2 ± 1.9 pg/mL, TNF-α 9.8 ± 2.7 pg/mL, IL-10 3.5 ± 1.0 pg/mL, fibrinogen 3.8 ± 0.7 g/L, D-dimer 470 ± 110 ng/mL.Platelet aggregation was assessed using ADP-induced optical aggregometry (5 μmol/L ADP). Echocardiography and coronary angiography were performed in all patients.Statistical analysis was conducted using SPSS 26.0. Continuous variables were expressed as mean ± SD. Between-group comparisons were performed using Student’s t-test or Mann–Whitney U test. Categorical variables were analyzed with χ² test. Multivariate logistic regression identified independent predictors of stent thrombosis. Predictive performance was evaluated using ROC curve analysis. A p-value <0.05 was considered statistically significant.
|
4. Results
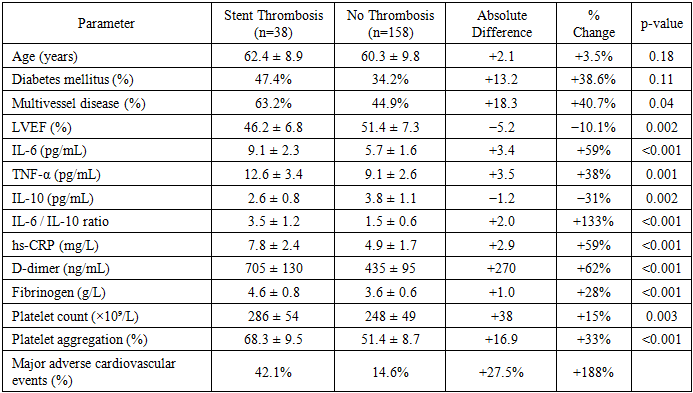
- During the 12-month follow-up period, stent thrombosis was diagnosed in 38 of 196 patients, corresponding to an overall incidence of 19.4%. Early thrombosis (≤30 days) accounted for 60.5% of cases (23 patients), whereas late thrombosis (>30 days) was observed in 39.5% (15 patients). The incidence of major adverse cardiovascular events (MACE) reached 42.1% in the thrombosis group compared with 14.6% among patients without thrombotic complications, reflecting an almost threefold increase in cardiovascular risk.Clinical characteristics demonstrated that patients who developed stent thrombosis had significantly lower left ventricular ejection fraction (LVEF) compared with the control group (46.2 ± 6.8% vs 51.4 ± 7.3%, p=0.002). Diabetes mellitus was more frequently observed among patients with thrombotic events (47.4% vs 34.2%), although this difference did not reach statistical significance. Multivessel coronary disease was also more prevalent in the thrombosis group (63.2% vs 44.9%, p=0.04), indicating a higher anatomical complexity of coronary lesions.Analysis of inflammatory biomarkers revealed a pronounced activation of the immune response in patients with thrombotic complications. Mean IL-6 concentration reached 9.1 ± 2.3 pg/mL, compared with 5.7 ± 1.6 pg/mL in patients without thrombosis, representing a 59% increase (p<0.001). Similarly, TNF-α levels were elevated by 38% (12.6 ± 3.4 vs 9.1 ± 2.6 pg/mL, p=0.001). In contrast, the anti-inflammatory cytokine IL-10 was significantly reduced in the thrombosis group (2.6 ± 0.8 vs 3.8 ± 1.1 pg/mL, p=0.002), corresponding to a 31% decrease. As a result, the IL-6/IL-10 ratio increased more than twofold (3.5 ± 1.2 vs 1.5 ± 0.6), reflecting a substantial imbalance between pro- and anti-inflammatory pathways.Markers of systemic inflammation demonstrated similar trends. High-sensitivity C-reactive protein (hs-CRP) levels were significantly higher in patients with thrombotic complications (7.8 ± 2.4 mg/L) compared with the control group (4.9 ± 1.7 mg/L), representing a 59% increase (p<0.001). These findings indicate that patients who developed stent thrombosis were characterized by a pronounced inflammatory activation prior to the thrombotic event.Hemostatic parameters also showed significant differences between groups. D-dimer concentration was markedly elevated in the thrombosis group (705 ± 130 ng/mL vs 435 ± 95 ng/mL), corresponding to a 62% increase (p<0.001). Fibrinogen levels were also higher in patients with thrombosis (4.6 ± 0.8 g/L vs 3.6 ± 0.6 g/L), representing a 28% increase (p<0.001). Platelet count increased by approximately 15% (286 ± 54 vs 248 ± 49 ×10⁹/L, p=0.003), while ADP-induced platelet aggregation demonstrated a 33% elevation (68.3 ± 9.5% vs 51.4 ± 8.7%, p<0.001).Correlation analysis demonstrated a strong association between inflammatory and coagulation parameters. IL-6 levels positively correlated with D-dimer concentration (r=0.63), suggesting that inflammatory activation may directly contribute to coagulation cascade activation. Similarly, TNF-α correlated with platelet aggregation (r=0.55), while IL-10 demonstrated an inverse correlation with D-dimer levels (r=-0.49). These findings support the concept that thrombotic complications after PCI arise from a coordinated interaction between inflammatory and hemostatic pathways.Multivariate logistic regression analysis identified several independent predictors of stent thrombosis. Elevated IL-6 levels above 7 pg/mL increased the risk of thrombosis by approximately 2.9-fold, while D-dimer levels above 600 ng/mL increased risk by more than threefold. An IL-6/IL-10 ratio above 2.5 was also associated with a significantly higher probability of thrombotic events. In addition, reduced LVEF below 45% increased thrombosis risk by approximately 180%.When patients were stratified according to the integrated phenotypic risk index (PRI-PCI), the incidence of thrombosis was 4.5% in low-risk patients, 16.8% in moderate-risk patients, and 43.7% in high-risk individuals, demonstrating nearly a tenfold difference between extreme categories. Receiver operating characteristic analysis confirmed a strong predictive performance of the model with an AUC of 0.86, sensitivity of 84%, and specificity of 78%.Overall, the obtained results demonstrate that patients who develop stent thrombosis exhibit a distinct immune-hemostatic phenotype characterized by significant activation of inflammatory pathways, increased coagulation activity, and pronounced cytokine imbalance. These findings support the concept that thrombotic complications after PCI are closely associated with thromboinflammatory processes rather than representing isolated disturbances of the coagulation system.
|
5. Discussion
- The results of the present study indicate that thrombotic complications after PCI are closely associated with the interaction between inflammatory and hemostatic pathways. Endothelial injury during stent implantation activates innate immune responses and promotes the release of pro-inflammatory cytokines such as IL-6 and TNF-α. These mediators enhance platelet activation, stimulate tissue factor expression, and accelerate thrombin generation, thereby increasing the probability of thrombus formation.The findings obtained in this study are consistent with previously published international investigations. Karczmarska-Wódzka et al. demonstrated that inflammatory activation significantly influences fibrin clot properties in cardiovascular disease. Similarly, Biggart et al. reported that immune-mediated mechanisms and thrombophilia may contribute to the development of stent thrombosis after PCI. These data support the concept that thrombotic complications represent a complex interaction between immune activation and coagulation imbalance rather than isolated disturbances of the coagulation system.It should also be noted that several clinical and procedural factors may influence the development of thrombotic complications after PCI. Variability in dual antiplatelet therapy, patient adherence to medication, and differences in platelet response may affect thrombosis risk. In addition, procedural characteristics such as stent type, stent length, lesion complexity, and implantation technique may also contribute to the occurrence of thrombotic events.Future research should focus on multicenter prospective studies aimed at validating the proposed immune-hemostatic risk model in larger patient populations. Integration of immunological biomarkers into clinical risk stratification algorithms may improve early identification of high-risk patients and facilitate the development of personalized therapeutic strategies in interventional cardiology.
6. Limitations
- The present study has several limitations. First, the investigation was conducted in a single clinical center, which may limit the generalizability of the results. Second, the observational design does not allow establishing a direct causal relationship between immunological activation and thrombotic complications. Further multicenter studies are required to confirm these findings.
7. Conclusions
- The present study demonstrates that thrombotic complications after percutaneous coronary intervention represent a manifestation of a thromboinflammatory process involving coordinated activation of inflammatory and hemostatic pathways. In patients who developed stent thrombosis, significant increases in pro-inflammatory markers were observed, including a 59% elevation in IL-6 and a 38% increase in TNF-α, accompanied by a 31% reduction in the anti-inflammatory cytokine IL-10, indicating pronounced cytokine imbalance.Simultaneously, activation of the coagulation system was confirmed by a 62% increase in D-dimer levels and a 28% elevation in fibrinogen concentration, while platelet aggregation increased by approximately 33% compared with patients without thrombotic complications. These changes were associated with a substantially higher incidence of major adverse cardiovascular events, which occurred in 42.1% of patients with stent thrombosis compared with 14.6% in patients without thrombosis, representing nearly a threefold increase in cardiovascular risk.Overall, the results indicate that stent thrombosis after PCI is characterized by a distinct immune-hemostatic phenotype involving a 30–60% increase in inflammatory activity and a 28–62% elevation in coagulation parameters. Integration of immunological biomarkers into clinical risk assessment may improve early identification of high-risk patients and contribute to more effective prevention of thrombotic complications after coronary interventions.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML