-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 1686-1689
doi:10.5923/j.ajmms.20261604.29
Received: Feb. 25, 2026; Accepted: Mar. 20, 2026; Published: Apr. 8, 2026

Clinical Significance of Hemostasis Biomarkers in Preventing Post-Intervention Restenosis After Coronary Intervention in Patients with Type 2 Diabetes Mellitus and Ischemic Heart Disease
Rakhimov Nurbek Alievich1, Naimova Shohida Anvarovna2
1Bukhara Regional Multidisciplinary Medical Center, Head of the Cardiology Department; Independent Researcher, Bukhara State Medical Institute, Uzbekistan
2Bukhara State Medical Institute, Associate Professor, Department of Hematology and Clinical Laboratory Diagnostics, DSc, Uzbekistan
Correspondence to: Rakhimov Nurbek Alievich, Bukhara Regional Multidisciplinary Medical Center, Head of the Cardiology Department; Independent Researcher, Bukhara State Medical Institute, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

In patients with ischemic heart disease (IHD) comorbid with type 2 diabetes mellitus (T2DM), the risk of restenosis after percutaneous coronary intervention (PCI) is increased, and haemostatic alterations play an important role in its pathogenesis. This study aimed to assess the clinical significance of haemostasis biomarkers (D-dimer, fibrinogen, platelet count, activated partial thromboplastin time, prothrombin time, and international normalized ratio) for early prediction of post-PCI restenosis, to develop individualized preventive therapy based on these markers, and to improve the effectiveness of antiplatelet/anticoagulant treatment strategies. The findings demonstrated a significant association between prothrombotic changes in the haemostatic system and the development of restenosis, suggesting that an integrated analysis of haemostasis biomarkers may enable early prediction of post-PCI complications and support the implementation of personalized preventive approaches in clinical practice.
Keywords: Type 2 diabetes mellitus, Ischemic heart disease, Percutaneous coronary intervention, Restenosis, Haemostatic system, D-dimer, Fibrinogen, Antiplatelet therapy, Anticoagulant therapy, Prognostic biomarker
Cite this paper: Rakhimov Nurbek Alievich, Naimova Shohida Anvarovna, Clinical Significance of Hemostasis Biomarkers in Preventing Post-Intervention Restenosis After Coronary Intervention in Patients with Type 2 Diabetes Mellitus and Ischemic Heart Disease, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 1686-1689. doi: 10.5923/j.ajmms.20261604.29.
1. Introduction
- The coexistence of ischemic heart disease (IHD) and type 2 diabetes mellitus (T2DM) is considered one of the most serious challenges in contemporary cardiology. In patients with T2DM, coronary atherosclerosis develops more rapidly, endothelial dysfunction and inflammatory processes are more pronounced, and therefore the incidence of restenosis after percutaneous coronary interventions (PCI) is significantly higher than in non-diabetic or otherwise healthy patients [1,3,5]. According to various sources, restenosis after PCI in patients with T2DM has been reported to reach 25–40% [2,4,6].Activation of the hemostatic system—manifested by increased fibrinogen levels, enhanced platelet reactivity, elevated D-dimer, and changes in coagulation parameters—may intensify peristent microthrombosis and increase the risk of restenosis. For this reason, early prediction of restenosis using hemostasis biomarkers and individualization of pharmacological prophylaxis are of substantial practical importance for reducing post-PCI complications. Currently, antiplatelet/anticoagulant therapy is generally administered according to standard protocols; however, in high-risk groups such as patients with T2DM, selecting a personalized regimen based on hemostasis markers is regarded as a strategic clinical approach to preventing restenosis [10,12,15]. Tissue factor is a principal trigger of coagulation after vascular injury and has also been implicated in neointimal responses. In a prospective study of stable angina patients undergoing percutaneous transluminal coronary angioplasty (PTCA) or stent implantation, baseline whole-blood tissue factor (TF) procoagulant activity was significantly higher in those who subsequently developed restenosis, suggesting that pre-procedural TF-related activity can serve as a predictive marker of restenosis risk. [5,6]. A key link between hemostasis and restenosis is thrombin generation. In clinically stable coronary artery disease patients undergoing elective PTCA, prothrombin fragment 1+2 (a thrombin generation marker) and additional hemostatic variables distinguished restenosis vs. non-restenosis groups; importantly, late luminal loss correlated positively with prothrombin fragment 1+2, supporting thrombin generation as a mechanistically relevant predictor of renarrowing [3,5]. Fibrinogen links inflammation and thrombosis and can influence clot structure and resistance to lysis—features often abnormal in T2DM. A coronary stenting study reported that preprocedural fibrinogen had predictive value for subsequent restenosis, supporting fibrinogen as a readily available biomarker with prognostic potential. Impaired fibrinolysis can lead to prolonged fibrin persistence and enhance vascular remodeling. In a prospective cohort after elective angioplasty/stenting, serial sampling showed that patients who developed restenosis had higher urokinase plasminogen activator (uPA) antigen and lower PAI-1 antigen at baseline and across multiple post-procedural timepoints; baseline uPA (together with post-procedure obstruction diameter) emerged as a significant predictor of follow-up stenosis [1-3].Platelets contribute to thrombus formation and release mediators that promote inflammation and neointimal growth. A systematic review and meta-analysis found that mean platelet volume (MPV) was significantly higher in patients who developed restenosis after coronary angioplasty, indicating MPV’s potential utility as a simple prognostic biomarker, while also acknowledging that whether MPV should directly guide therapy remains uncertain [16]. While individual biomarkers can be informative, integrated models may better represent the multi-factorial nature of restenosis—especially in T2DM where metabolic control and thrombosis pathways intersect. A recent study developed an ISR risk scoring model in coronary artery disease patients that identified D-dimer (along with HbA1c, LDL-C and other clinical factors) as an independent risk factor incorporated into prediction.The limited number of studies in this area, the fact that biomarkers have often been evaluated separately, and the insufficient development of integrated prognostic models further increase the scientific and practical relevance of this topic.
2. Materials and Methods
- Between 2022 and 2024, a total of 110 patients aged 18–74 years who were hospitalized at the Bukhara Regional Multidisciplinary Medical Center with a diagnosis of ischemic heart disease (IHD) and underwent percutaneous coronary intervention (PCI) were enrolled in the study. Among them, 61 were men and 59 were women. A control group of 30 healthy volunteers was also recruited.The study assessed clinical and anamnestic data; laboratory biochemical parameters (blood glucose, HbA1c, insulin resistance index HOMA-IR, lipid profile, and hs-CRP); hemostasis biomarkers (D-dimer, fibrinogen, platelet count, activated partial thromboplastin time [aPTT], prothrombin time [PT], and international normalized ratio [INR]); and inflammatory cytokines (IL-6, IL-1β, TNF-α) measured by enzyme-linked immunosorbent assay (ELISA). Instrumental diagnostics included ECG, echocardiography, and vascular ultrasonography.Statistical analysis was performed using Excel 10 software and included correlation analysis, ROC-curve analysis, binary logistic regression, estimation of sensitivity/specificity, and determination of cut-off values. A p-value < 0.05 was considered statistically significant.
3. Results
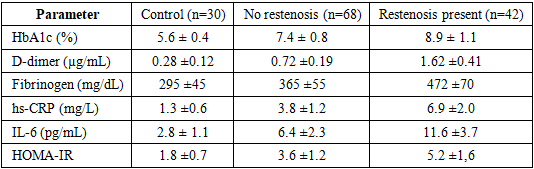
- During the 12-month follow-up period after PCI, clinical and/or biomarker-based signs of restenosis were identified in 42 of 110 patients (38.1%). In the restenosis group, levels of HbA1c, D-dimer, fibrinogen, hs-CRP, and IL-6 were significantly higher compared with patients without restenosis (p < 0.05). The insulin resistance index (HOMA-IR) was also markedly elevated in this group.
|
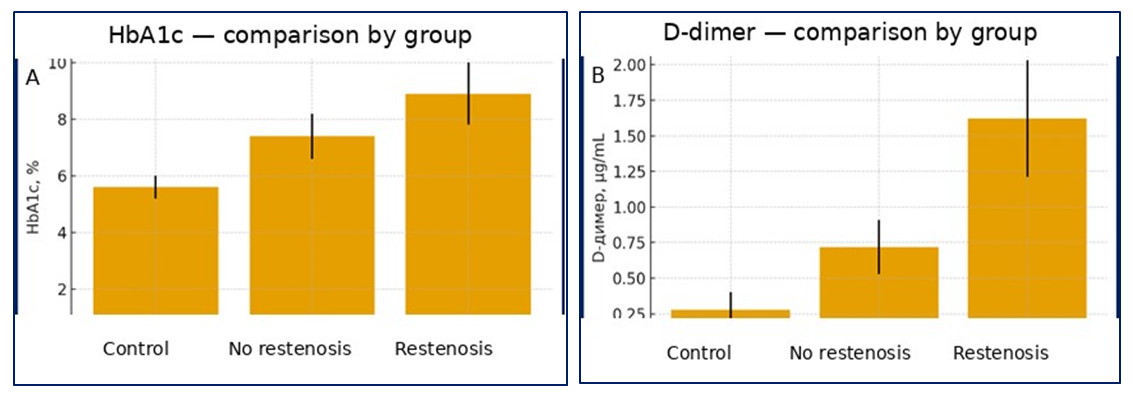 | Figure 1. Group-wise analysis of HbA1c and D-dimer |
4. Conclusions
- The obtained findings indicate that the combined activation of type 2 diabetes–related hyperglycaemia, insulin resistance, and components of the inflammatory cascade constitutes the principal pathogenetic drivers of post-PCI restenosis. According to the data presented in the table, patients who developed restenosis demonstrated an increase in HbA1c to 8.9 ± 1.1%, D-dimer to 1.62 ± 0.41 µg/mL, and fibrinogen to 472 ± 70 mg/dL. In parallel, a pronounced elevation of hs-CRP (6.9 ± 2.0 mg/L) and IL-6 (11.6 ± 3.7 pg/mL) indicates concomitant activation of inflammatory and thrombotic states in this population. The substantially lower values observed in the control group (HbA1c 5.6 ± 0.4%; D-dimer 0.28 ± 0.12 µg/mL; fibrinogen 295 ± 45 mg/dL) further confirm the high clinical relevance of these biomarker differences. Moreover, the increase of HOMA-IR to 5.2 ± 1.6 in the restenosis group provides additional support for the contributory role of insulin resistance in restenosis development.Collectively, these results suggest that elevated HbA1c may exacerbate microcirculatory impairment, endothelial injury, and thrombogenic activity—reflected by increases in fibrinogen and D-dimer—while higher hs-CRP and IL-6 levels may accelerate systemic inflammation in the diabetic milieu and promote peri-stent neointimal proliferation. Accordingly, simultaneous assessment of haemostatic markers and inflammatory cytokines appears to have high prognostic value for individualised prediction of post-PCI restenosis risk. In patients with type 2 diabetes mellitus, personalisation of antiplatelet therapy and anti-inflammatory strategies, together with maintaining HbA1c at targeted lower levels, is recommended as a clinically relevant approach to substantially reduce the likelihood of restenosis.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML