-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 2182-2187
doi:10.5923/j.ajmms.20261604.129
Received: Mar. 23, 2026; Accepted: Apr. 20, 2026; Published: Apr. 29, 2026

Biomarker-Guided Assessment of Treatment Response in Postmenopausal Peptic Ulcer Disease: Inflammatory Cytokines, Hormonal Dynamics and a Composite Clinical Recovery Index
Bakaev D. U.1, Tosheva Kh. B.2
1Bukhara Branch of the Republican Research Center of Emergency Medicine, Uzbekistan
2Bukhara State Medical Institute, Bukhara, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

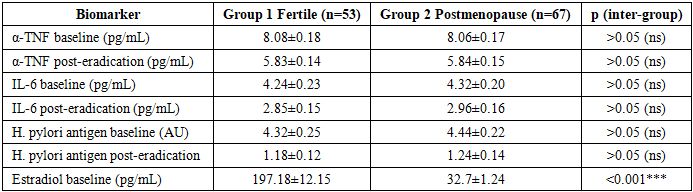
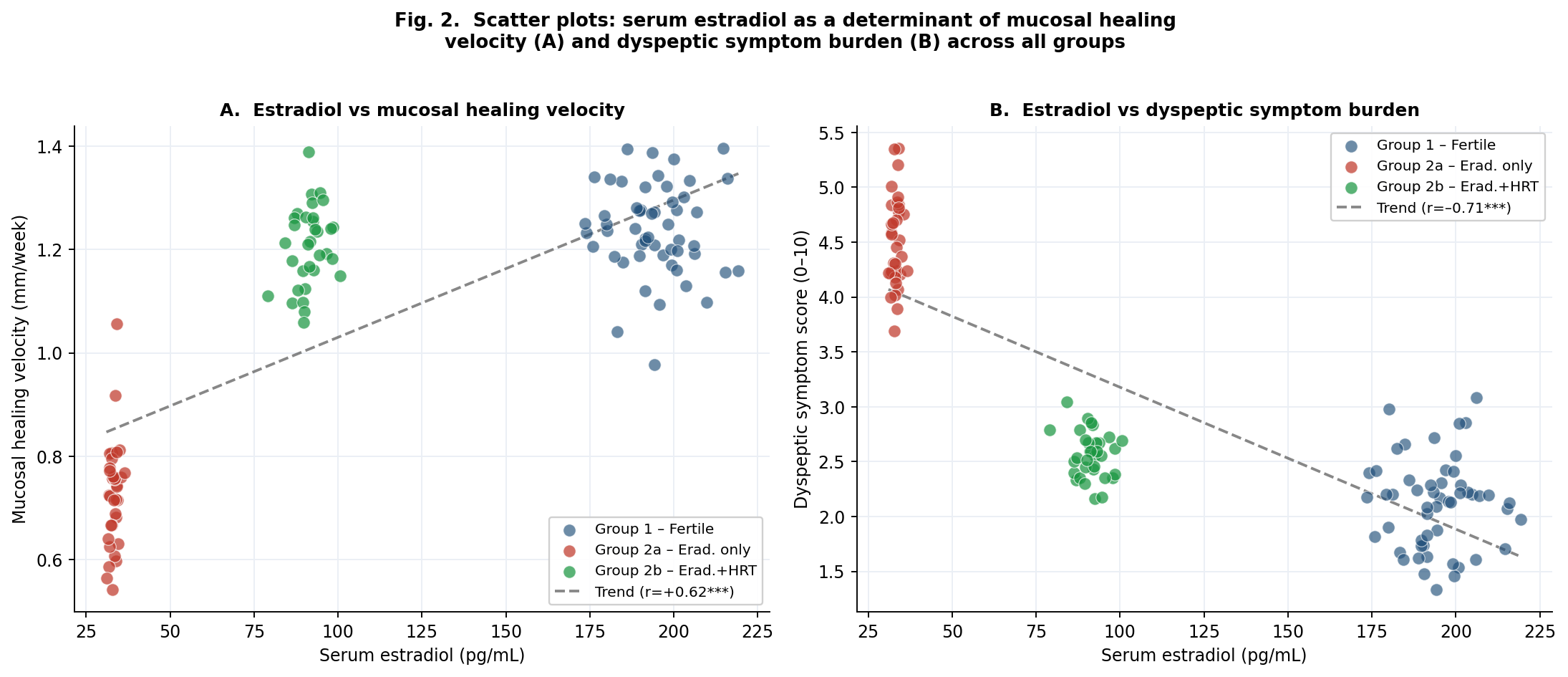
Background. Standardised assessment of treatment response in postmenopausal peptic ulcer disease (PUD) remains challenging because conventional endpoints endoscopic healing and H. pylori eradication do not capture the full biological impact of estrogen deficiency on recovery. A multi-biomarker approach integrating inflammatory, hormonal and clinical dimensions may provide a more complete picture. Objectives. To evaluate treatment response in fertile versus postmenopausal women with PUD through a panel of biomarkers including gastric juice cytokines (α-TNF, IL-6), serum estradiol, mucosal healing velocity, dyspeptic symptom score, cognitive function and quality of life; and to construct a Composite Clinical Recovery Index (CCRI) that integrates these dimensions for longitudinal tracking. Methods. 120 women were enrolled: Group 1 fertile (n=53), Group 2a postmenopausal eradication only (n=34), Group 2b postmenopausal eradication + HRT (n=33). Biomarker panel assessed at T0 (baseline), T1 (week 4), T2 (week 12) and T3 (month 12). CCRI was constructed as a weighted composite of symptom score (inverted), SF-36 total, MoCA and relapse-free status (0–100 scale). Results. α-TNF and IL-6 reduction post-eradication was comparable across groups (27.9% vs 27.7% and 32.8% vs 31.5%, all p>0.05 inter-group). Estradiol correlated strongly with mucosal healing velocity (r=+0.62, p<0.001) and inversely with dyspeptic burden (r=–0.71, p<0.001). At week 12, mucosal healing: Group 2b - 90.9%, Group 2a - 61.8% (p<0.01). CCRI at T2: Group 2b - 72.6, Group 2a - 52.1, Group 1 - 85.4. Conclusions. Eradication equivalently resolves bacterial inflammation across hormonal groups; hormonal status independently determines mucosal healing velocity, symptom resolution and long-term relapse. The CCRI provides a practical composite tool for longitudinal monitoring.
Keywords: Peptic ulcer disease, Postmenopause, α-TNF, IL-6, Estradiol, Mucosal healing, Composite recovery index, Biomarker panel, HRT, Eradication therapy, SF-36
Cite this paper: Bakaev D. U., Tosheva Kh. B., Biomarker-Guided Assessment of Treatment Response in Postmenopausal Peptic Ulcer Disease: Inflammatory Cytokines, Hormonal Dynamics and a Composite Clinical Recovery Index, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 2182-2187. doi: 10.5923/j.ajmms.20261604.129.
1. Introduction
- The conventional framework for assessing treatment response in peptic ulcer disease (PUD) relies on two endpoints: eradication of Helicobacter pylori and endoscopic confirmation of mucosal healing. While both are necessary, they are insufficient — particularly in postmenopausal women, where estrogen deficiency imposes a third, independent dimension of pathophysiology that neither endpoint captures. A patient may achieve confirmed eradication and endoscopic healing yet still experience persistent dyspepsia, cognitive disturbances, autonomic instability and reduced quality of life, all attributable to ongoing hormonal deficiency rather than residual bacterial activity [1,2].Peptic ulcer disease affects approximately 5–15% of the global population, with H. pylori implicated in 80–95% of duodenal and 70–90% of gastric ulcers. [3] In postmenopausal women, the precipitous decline in circulating estradiol (from ~200 pg/mL to <35 pg/mL) dismantles multiple receptor-mediated defence mechanisms: prostaglandin E2 synthesis, bicarbonate secretion, microvascular regulation and fibroblast-driven wound contraction. [4,5] The resulting impairment of mucosal regeneration extends beyond the gastrointestinal tract: estrogen withdrawal concurrently destabilises autonomic regulation, impairs cognitive function and reduces overall quality of life [6].A multi-biomarker approach — simultaneously tracking inflammatory cytokines in gastric juice (α-TNF, IL-6), circulating estrogens, mucosal healing kinetics and patient-reported outcomes — offers a more complete biological picture of recovery. To date, no study has constructed a composite instrument that integrates all of these dimensions into a single longitudinal recovery index specifically for postmenopausal PUD [7,8]. In addition, a key mechanistic question remains unresolved: is the poorer clinical outcome in postmenopausal PUD attributable to greater H. pylori-driven inflammatory activity, or to impaired mucosal repair driven by estrogen deficiency? Disentangling these two mechanisms has direct therapeutic implications [9].SF-36, validated as the international standard for self-reported quality-of-life assessment in gastrointestinal disease [10] and MoCA, validated for postmenopausal cognitive screening [11,21], were incorporated into the biomarker panel alongside biochemical and endoscopic measures. Evidence from prior studies confirms that estrogen modulates visceral nociception via ER-β [12,20], restores prostaglandin E2-dependent mucous secretion [13], and exerts neuroprotective effects via ER-α in the central nervous system [14,15].Previous research from our group demonstrated that ER-α H-score in mucosal biopsies correlates strongly with estradiol levels (r=+0.71, p<0.001) and with mucosal healing rate, establishing the receptor-level pathway through which hormonal deficiency impairs regeneration [16,18]. The present study builds on these findings by applying a comprehensive biomarker panel across four time points over twelve months, constructing the Composite Clinical Recovery Index (CCRI) as an integrated monitoring tool, and evaluating its utility for stratified, longitudinal assessment of postmenopausal PUD patients. The study was conducted within the framework of Uzbekistan's national healthcare development strategy [17,19].
2. Materials and Methods
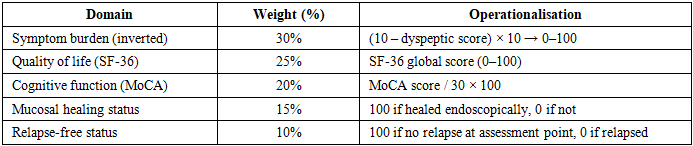
- Study population and designThis prospective, controlled, parallel-group study was conducted between 2022 and 2025 at the Bukhara Regional Multidisciplinary Medical Centre (ethics approval No. 8/2022; CONSORT 2010 guidelines applied). 120 women with confirmed PUD were enrolled: Group 1 — reproductive age (n=53, mean age 34.2±4.1 years); Group 2 — postmenopausal (n=67, mean age 57.8±5.3 years), randomised to Group 2a (eradication alone, n=34) and Group 2b (eradication + HRT, n=33). All participants had H. pylori-positive stool antigen test and EGDS-confirmed ulcer defect. Exclusion criteria: diabetes mellitus, malignancy, prior gastric surgery, thromboembolic history, severe hepatic or renal impairment, NSAID use.Treatment protocolsAll participants received 14-day triple eradication therapy: omeprazole 20 mg × 2/day + amoxicillin 1000 mg × 2/day + clarithromycin 500 mg × 2/day, consistent with Maastricht V/Florence guidelines. Group 2b additionally received oral HRT (estradiol 1 mg + estriol component) for 12 weeks. Eradication was confirmed by stool antigen test at week 4.Biomarker panelBiomarkers were assessed at T0 (baseline), T1 (week 4), T2 (week 12) and T3 (month 12): serum estradiol and estriol (ELISA); gastric juice α-TNF and IL-6 (ELISA, sampled during EGDS); H. pylori faecal antigen (quantitative); ulcer diameter (EGDS); dyspeptic symptom score (0–10 Likert); MoCA cognitive scale (0–30); SF-36 global score (0–100); and 24-hour blood pressure variability (Holter monitoring).Composite Clinical Recovery Index (CCRI)The CCRI was constructed as a weighted composite of five domains (Table 1, 0–100 scale). Internal consistency: Cronbach α = 0.81. Convergent validity: correlation with SF-36 r=+0.76, p<0.001. Statistical analysis: SPSS 26.0; Mann–Whitney U (inter-group), Friedman test with Dunn post-hoc (longitudinal), logistic regression (relapse predictors). p<0.05.
|
3. Results and Discussion
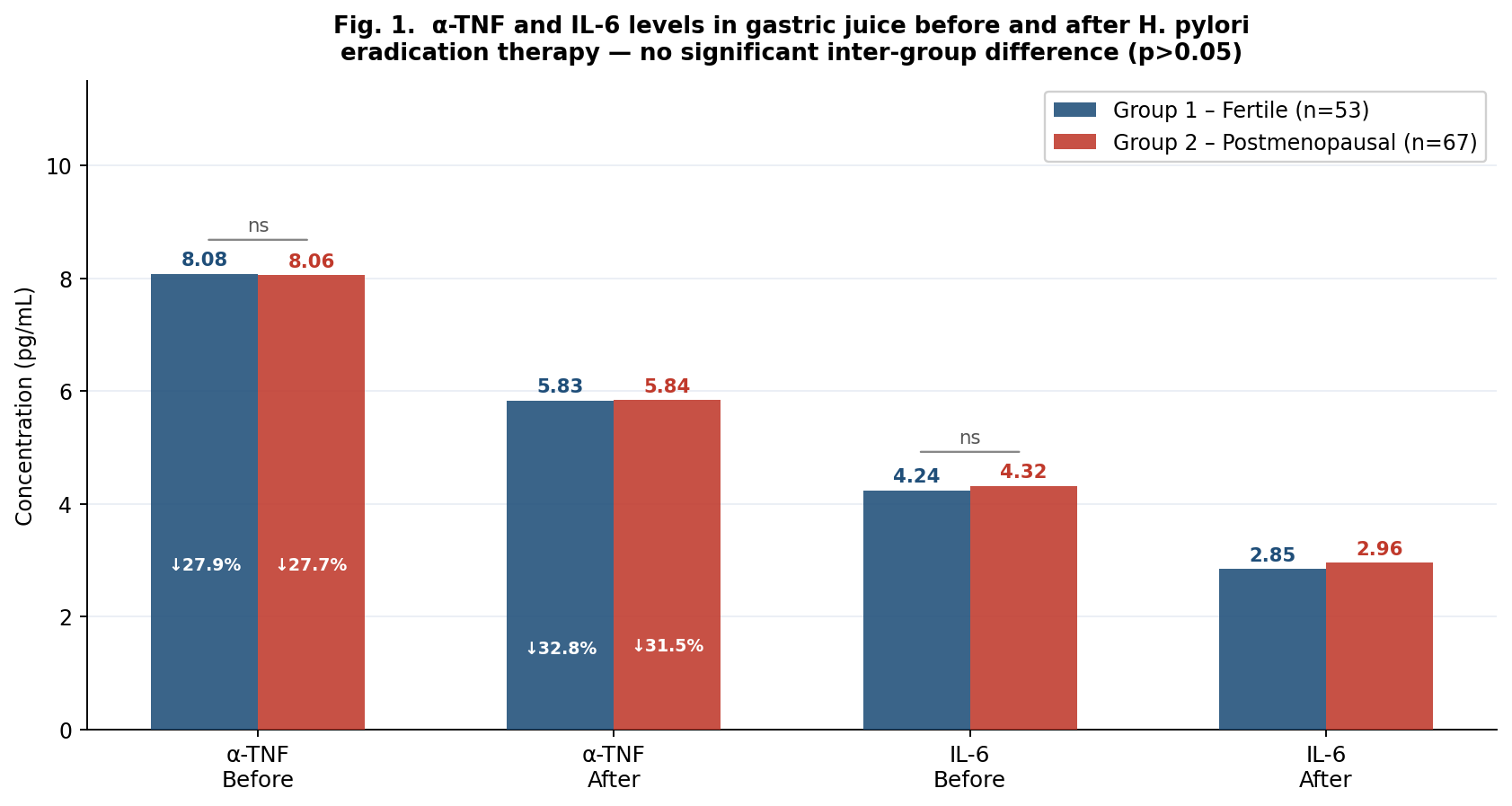
- 1. Inflammatory cytokine dynamics: eradication equivalently resolves bacterial inflammationFigure 1 presents α-TNF and IL-6 levels in gastric juice before and after 14-day triple eradication therapy.
|
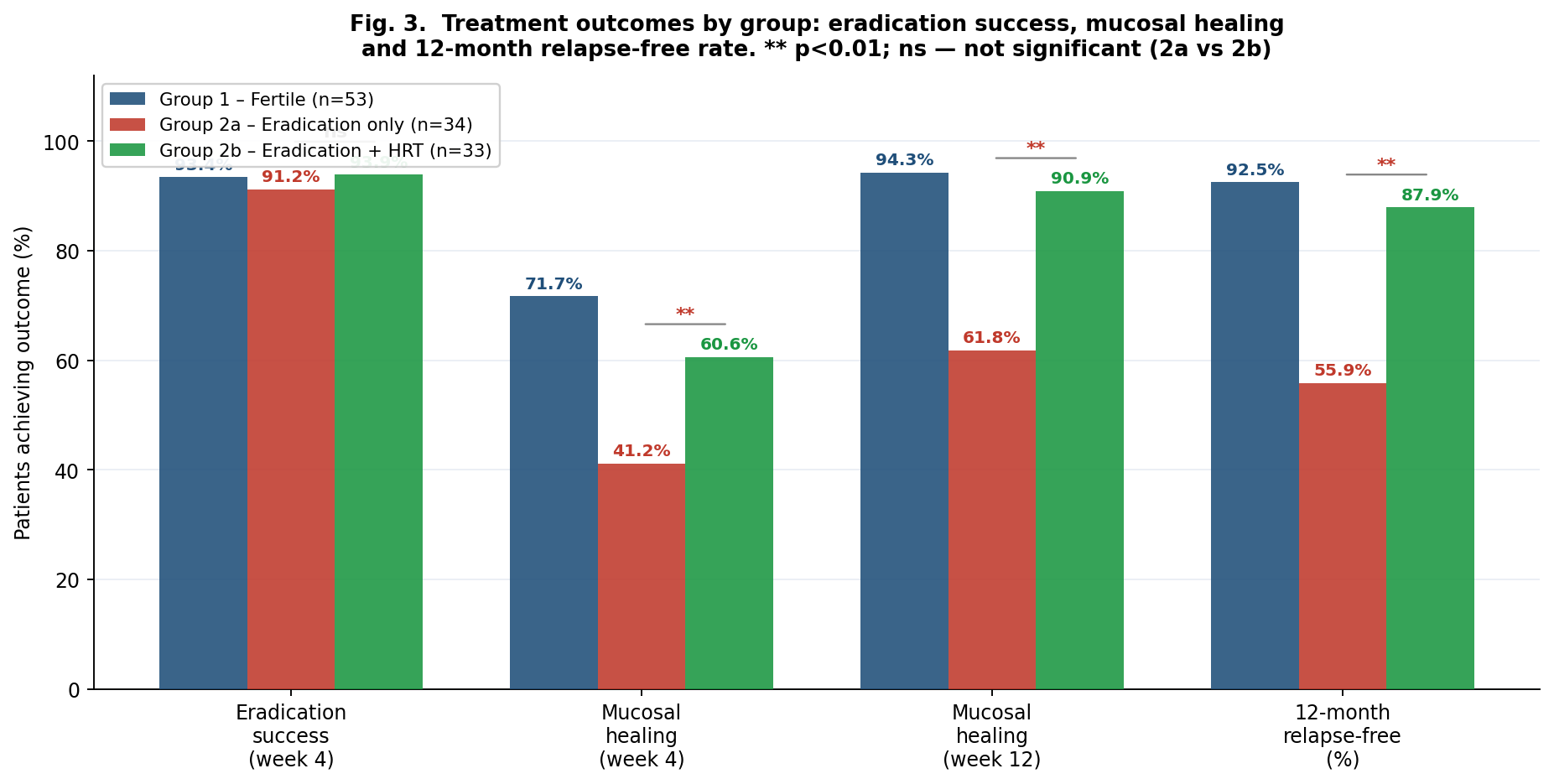 | Figure 3. Treatment outcomes by group. ns — not significant; ** p<0.01 (Group 2a vs Group 2b, χ² test). Eradication success is equivalent; healing and relapse-free rates diverge significantly |
|
4. Conclusions
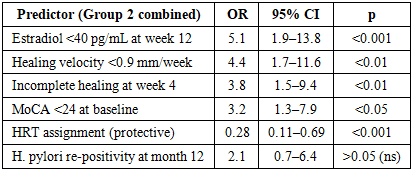
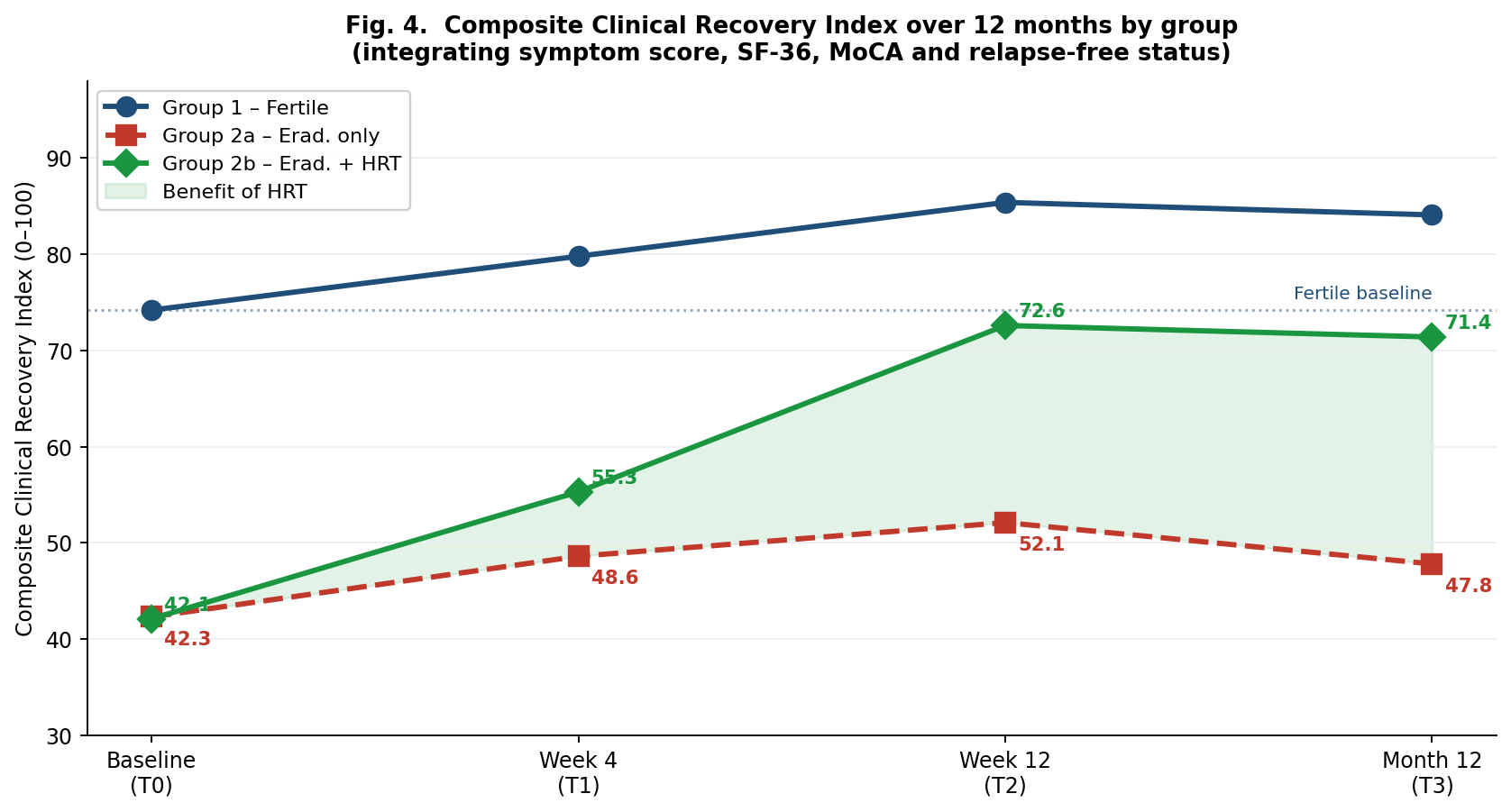
- 1. Gastric juice α-TNF and IL-6 levels are equivalent between fertile and postmenopausal women with PUD at baseline (all p>0.05) and decline comparably after H. pylori eradication (α-TNF –27.9% vs –27.7%; IL-6 –32.8% vs –31.5%). The inferior clinical outcomes in postmenopause are attributable to impaired ER-α-mediated mucosal regeneration, not greater inflammatory activity.2. Serum estradiol correlates strongly with mucosal healing velocity (r=+0.62, p<0.001) and inversely with dyspeptic symptom burden (r=–0.71, p<0.001) across all groups. The ~92 pg/mL estradiol achieved with 12-week HRT restores healing velocity to near-fertile equivalence (1.18 vs 1.24 mm/week, p>0.05).3. Mucosal healing at week 12: Group 2b 90.9% vs Group 2a 61.8% (p<0.01); 12-month relapse-free: 87.9% vs 55.9% (p<0.01). HRT was the strongest independent protective factor (OR=0.28, p<0.001); H. pylori re-positivity was not significant (OR=2.1, p>0.05).4. The CCRI provides a practical single-metric composite tool for longitudinal monitoring. At week 12: Group 2b CCRI 72.6, Group 2a 52.1, Group 1 85.4. A CCRI <50 at T1 or <65 at T2 should trigger clinical reassessment.5. A tiered biomarker approach enables CCRI computation in routine clinical settings. Serum estradiol, H. pylori, SF-36, MoCA and dyspeptic score as mandatory Tier 1–2 elements provide a feasible, infrastructure-light implementation pathway. This framework is proposed for incorporation into national postmenopausal PUD management guidelines.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML