-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 2161-2163
doi:10.5923/j.ajmms.20261604.124
Received: Apr. 8, 2026; Accepted: Apr. 26, 2026; Published: Apr. 29, 2026

Optimization of Prevention of Purulent-Septic Complications After Cesarean Section
Nishonova Diloramxon Axmatjanovna1, Asrankulova Diloramxon Bakhtiyarovna2, Nadjmitdinov Otabek Bakhritdinovich3
1Assistant, Department of Obstetrics and Gynecology No.1, Andijan State Medical Institute, Andijan, Uzbekistan.
2Doctor of Medical Sciences, Andijan State Medical Institute, Professor, Head of 1-Department of Obstetrics and Gynecology, Andijan, Uzbekistan
3Head of the Department of Medical Radiology, Interventional Cardiology, Nephrology-Hemodialysis and Clinical and Laboratory Diagnostics, Andijan State Medical Institute, Andijan, Uzbekistan
Correspondence to: Nadjmitdinov Otabek Bakhritdinovich, Head of the Department of Medical Radiology, Interventional Cardiology, Nephrology-Hemodialysis and Clinical and Laboratory Diagnostics, Andijan State Medical Institute, Andijan, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Cesarean section remains one of the most commonly performed procedures in obstetrics and is simultaneously a significant risk factor for postoperative infectious complications. It is well established that the incidence of infections in women after cesarean delivery is 5–20 times higher compared to vaginal birth, highlighting the clinical importance of effective preventive strategies. Routine antibiotic prophylaxis reduces the rate of postoperative infections by approximately 60–70%; however, there remains a need for additional approaches aimed at enhancing tissue repair and reducing inflammatory responses. In recent years, particular attention has been paid to autoplasma therapy, which, due to its high concentration of growth factors, promotes tissue regeneration, improves microcirculation, and reduces the risk of infectious complications.The aim of the study was to optimize a comprehensive strategy for the prevention of purulent-septic complications following cesarean section through the combined use of antibiotic prophylaxis and autoplasma therapy.
Keywords: Cesarean section, Purulent-septic complications, Prevention, Autoplasma therapy, Antibiotic prophylaxis, Postpartum period, Endometritis, Uterine subinvolution
Cite this paper: Nishonova Diloramxon Axmatjanovna, Asrankulova Diloramxon Bakhtiyarovna, Nadjmitdinov Otabek Bakhritdinovich, Optimization of Prevention of Purulent-Septic Complications After Cesarean Section, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 2161-2163. doi: 10.5923/j.ajmms.20261604.124.
1. Introduction
- Cesarean section (CS) is one of the most frequently performed surgical interventions in modern obstetrics, with a steadily increasing global rate. Despite advances in surgical techniques, anesthesia, and perioperative care, cesarean delivery remains associated with a higher risk of postoperative complications compared to vaginal birth, particularly infectious morbidity. Purulent-septic complications, including endometritis, wound infection, and pelvic inflammatory processes, continue to be among the leading causes of maternal morbidity in the postoperative period [2,8,11].It has been reported that the incidence of infectious complications following cesarean section is significantly higher-by 5 to 20 times-than after spontaneous vaginal delivery. This underscores the importance of effective preventive strategies, especially in patients with identified risk factors such as prolonged labor, premature rupture of membranes, anemia, or existing genital tract infections [3,4,7].Antibiotic prophylaxis is widely recognized as a standard of care and has been shown to substantially reduce the incidence of postoperative infections. However, despite its proven effectiveness, antibiotic prophylaxis alone does not completely eliminate the risk of complications, particularly in high-risk groups. This necessitates the development and implementation of additional therapeutic approaches aimed at improving tissue healing, enhancing local immune responses, and minimizing inflammatory processes [11,14].In this context, increasing attention has been given to regenerative methods, including autoplasma therapy, also known as platelet-rich plasma (PRP). Due to its high concentration of biologically active substances and growth factors, PRP has demonstrated the potential to stimulate tissue regeneration, improve microcirculation, and accelerate wound healing. Its application in obstetrics, particularly in surgical delivery, represents a promising direction for reducing postoperative complications [4,12].Therefore, the optimization of comprehensive preventive strategies combining antibiotic prophylaxis with regenerative approaches such as autoplasma therapy is of considerable clinical relevance and may significantly improve postoperative outcomes in women undergoing cesarean section.
2. Materials and Methods
- Clinical investigations were carried out in 110 pregnant women delivered by cesarean section in the lower uterine segment at the Republican Specialized Scientific and Practical Medical Center for Maternal and Child Health (Andijan branch) between 2024 and 2025.At the first stage, a retrospective analysis of clinical data from 2023 was performed, including the review of medical records of 130 women who had undergone cesarean section, in order to assess the frequency and structure of postoperative complications.At the second stage, patients were allocated into two groups:• Group I – 45 patients receiving standard therapeutic management; • Group II – 65 high-risk patients treated with the proposed comprehensive approach (local antibiotic therapy combined with autoplasma therapy).All patients underwent ultrasound examination, Doppler assessment of uterine blood flow, and standard clinical and laboratory tests (complete blood count, urinalysis, smear microscopy, and coagulation profile). In the main group, additional parameters were evaluated, including D-dimer levels, C-reactive protein, and blood lactate.The third stage involved a comparative analysis of the effectiveness of standard and proposed treatment methods. At the fourth stage, an algorithm for the prevention of postoperative complications was developed based on the obtained results.During the postoperative period, patients were assessed for general condition, body temperature, lactation, uterine status, characteristics of lochia, and the condition of the postoperative wound.
3. Results
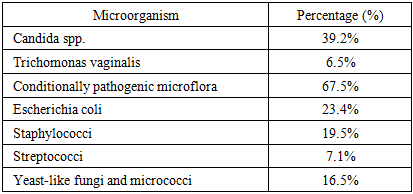
- A retrospective analysis was conducted on the medical records of 130 women who underwent cesarean section. Patients belonging to the high-risk group were included in the study. According to the results obtained, the mean age of the patients was 25 ± 4.5 years, and the following findings were revealed.
|
|
|
4. Discussion
- The findings of the present study indicate a high clinical efficacy of the proposed approach for the prevention of purulent-septic complications following cesarean section. The high prevalence of infectious risk factors identified during the retrospective analysis highlights the need for enhanced preventive strategies in this patient population.Local administration of antibiotics ensures a high concentration of the drug directly at the surgical site, thereby significantly improving the effectiveness of infection prevention [7,9,15]. At the same time, autoplasma therapy appears to promote tissue regeneration, enhance microcirculation, and reduce inflammatory responses.The absence of uterine subinvolution, hematomas, and purulent-inflammatory complications in the main group demonstrates the clear clinical advantage of the proposed method. The few cases of subfebrile temperature were not associated with infectious processes, further supporting the safety of this approach.In conclusion, the developed comprehensive strategy can be considered an effective method for preventing postoperative infectious complications and may be recommended for implementation in clinical practice, particularly in high-risk patients.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML