-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 2151-2155
doi:10.5923/j.ajmms.20261604.122
Received: Mar. 27, 2026; Accepted: Apr. 22, 2026; Published: Apr. 29, 2026

Significance of Clinical Factors and Endothelial Dysfunction in Tge Development of Femoral Head Osteonecrosis
Ulmasova Sh. R., Musaev T. S., Amonov E. I., Ibragimov Z. Z.
Central Military Hospital, Tashkent, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: Avascular necrosis of the femoral head develops in the context of impaired microcirculation and endothelial dysfunction, which play a key role in the disease pathofenesis. Objective: This study analyzed the allele and genotype frequencies of rs1799983, rs1549758, and rs2070744 polymorphisms of the NOS3 gene in patients with avascular necrosis of the femoral head and assessed their asosiashin with clinical risk factors. Methods: Allele and genotype frequencies were analyzed in patients with avascular necrosis of the femoral head using molecular genetic methods, and their association with clinical risk factors was evaluated. Results: Obtained result indicate that genetic variants can contribute to increased risk of development of disease and supports their potential relevance as genetic markers for early diagnosis and provides prevention of avascular necrosis of age femoral head. Conclusion: Polymorphisms of the gene encoding endothelial nitric oxide synthase may serve as potential markers for early diagnosis and prevention of avascular necrosis of the femoral head.
Keywords: Avascular necrosis of the femoral head, NOS3 gene, Endothelial dysfunction, Nitric oxide, Microcirculation, COVID-19
Cite this paper: Ulmasova Sh. R., Musaev T. S., Amonov E. I., Ibragimov Z. Z., Significance of Clinical Factors and Endothelial Dysfunction in Tge Development of Femoral Head Osteonecrosis, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 2151-2155. doi: 10.5923/j.ajmms.20261604.122.
1. Introduction
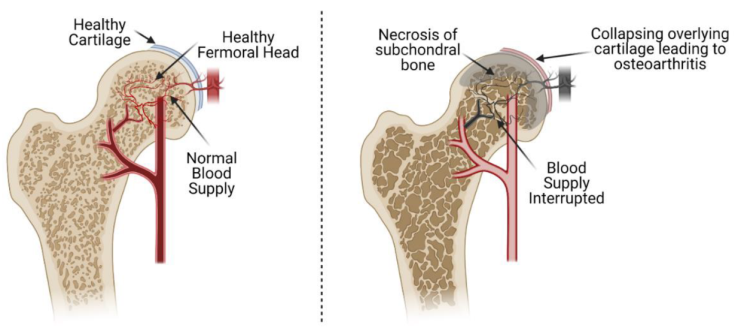
- Avascular osteonecrosis (AVN) is one of most severe degenerative diseases in orthopedics/traumatolofy, particularly femoral head avascular osteonecrosis (FHAVN). It is characterized by osteocyte necrosis due to impaired blood supply in bone tissue, subchondral fractures, and during disease profression, structural collapse of the femoral head, ultimately leading to the development of hip joint osteoarthritis [1,2,3]. In clinical practice, FHAVN predominantly affect working population, making it a condition of significant sociale and economic concern. In the early stages, nonspecific clinical symptoms and delayed diagnosis often result in severe impairment of hip joint function and necessitate joint replacement [3,4].The etiolofy of FHAVN is multifactorial. Risk factors include corticosteroid use, alcohol consumption, trauma, coagulation disorders, metabolic and endocrine patholofies, as well as comorbid somatic diseases [5,6]. Microcirculatory disturbances and endothelial dysfunction play a decisive role in the pathofenesis of FHAVN [7,8,12]. Endothelial nitric oxide synthase (eNOS), encoded by the NOS3 gene, is a key regulator of nitric oxide (NO) synthesis. In individuals with polymorphisms in this gene, reduced NO production can result with vascular spasms, hyper coagulation, and ischemic changes in bone tissue, representing an important pathofenetic factor (Figure 1).
2. Materials and Methods
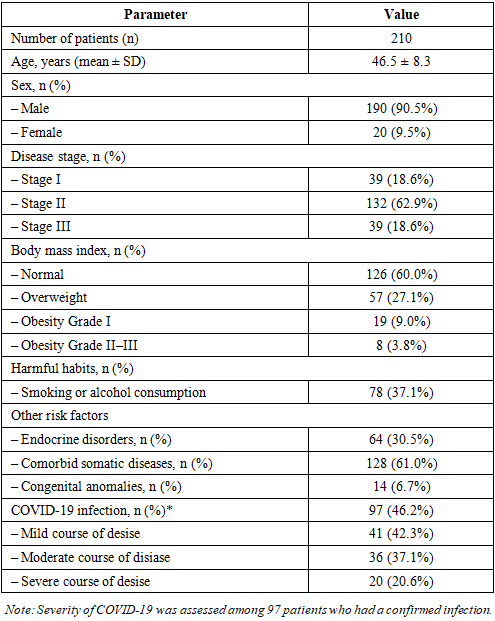
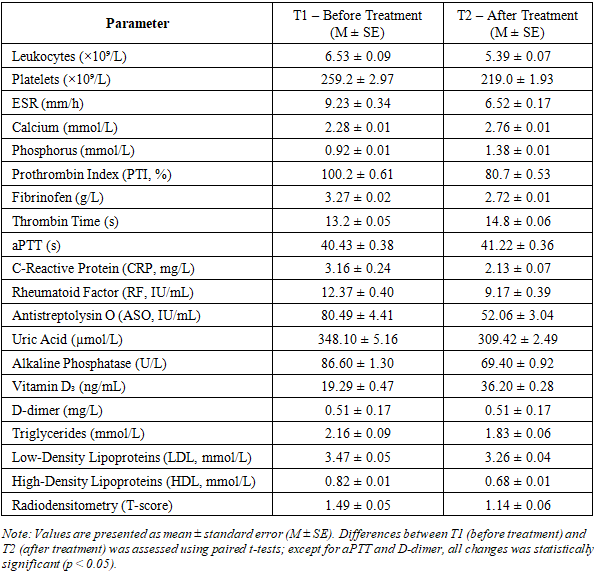
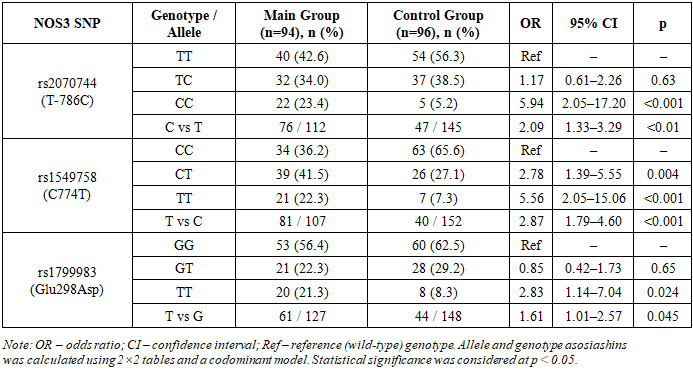
- The study was conducted at the Central Military Hospital from 2021 to 2025. It was an observational analytical study aimed at assessing the asosiashin of NOS3 gene polymorphisms with clinical indicators in patients with femoral head osteonecrosis. A total of 210 patient was diagnosed with femoral head osteonecrosis was included. Diagnosis was confirmed based on clinical data and instrumental examinations (X-ray and MRI). Disease stages was classified I to III.Clinical and Laboratory Examinations: Clinical and laboratory parameters was assessed in patients with femoral head osteonecrosis. All patients was monitored according to a unified standard clinical protocol. Clinical indicators included age, disease duration, body mass index (BMI), osteonecrosis stage, laterality (right or left), and comorbidities.Laboratory Examinations: Blood samples was collected from the peripheral vein in the morning after fasting. The following parameters was analyzed:Complete blood count: Platelet count, leukocyte count, erythrocyte sedimentation rate (ESR); Biochemical parameters: Calcium, phosphorus, alkaline phosphatase, uric acid, total cholesterol, triglycerides, lipoproteins;Hemostasis parameters: Prothrombin index (PTI), fibrinofen, thrombin time, activated partial thromboplastin time (aPTT), international normalized ratio (INR), D-dimer; Inflammatory markers: C-reactive protein (CRP), rheumatoid factor (RF), antistreptolysin O (ASO); Hormonal parameters: Parathyroid hormone, vitamin D3.Instrumental Examinations: Bone tissue status was evaluated using radiodensitometry. Intima- media thickness (IMT) of major arteries was measured using color Doppler scanning to asses vascular wall status and analyzed dynamically.Molecular Genetic Examinations: Main and control groups was formed. The main group included 94 inpatients diagnosed with femoral head osteonecrosis, while the control group consisted of 96 apparently healthy individuals. Peripheral venous blood (2–5 mL) was collected into EDTA tubes. Genomic DNA was extracted using a selective detergent precipitation method with the Genomic DNA Purification Kit (Thermo Fisher Scientific, USA), yielding 2–10 µg of high-quality DNA of approximately 50 kb from 0.2 mL of blood within 35 minutes. DNA quality and concentration was assessed spectrophotometrically (A260/A280). Polymorphisms rs1799983, rs1549758, and rs2070744 of the NOS3 gene was analysed using real-time PCR with the “SNP-Screen” kit. Allele-specific dual probes were used fi r detection, witg results recorded via two fluorescent channels to differentiate each allele. PCR reactions was performed using the qTOWER³ Real-Time PCR Thermal Cycler (Germany).Statistical Analysis: Quantitative variants were expressed as mean ± standard error (M ± m), and analyzed using Doctor Stat 2013 (Version 1.9). Qualitative data was presented as n (%), quantitative data as mean ± SD. Genotype and allele frequencies was calculated, and Hardy– Weinberg equilibrium (HWE) was tested. Differences between groups was assessed with the χ² test or Fisher exact test. Lofistic regression (OR, 95% CI) was used to identify factors associated with osteonecrosis severity, adjusting for age, sex, BMI, harmful habits, comorbidities, and COVID-19 history. A p-value < 0.05 was considered statistically significant.Ethical Considerations: The study protocol was approved by the internal ethics committee. All participants have written agreement and confidentiality was ensured.Scientific Novelty: Fir the first time, the asosiashin between clinical-laboratory parameters, NOS3 gene polymorphisms, and osteonecrosis pathofenesis was analyzed in 210 patients with osteonecrosis.Clinical Factors: The mean age of participants was 46.5 ± 8.3 years, indicating that osteonecrosis predominantly affects working-age adults (Table 1). Male patients predominated (90.5%), highlighting sex- specific risk factors such as occupational load, harmful habits, and hormonal influences. Stage analysis showed that most patients was at stage II (62.9%), indicating late clinical presentation. Stages I and III each comprised 18.6%, reflecting limited early diagnostic opportunities and declared medical attention. BMI analysis revealed that approximately 40% of patients was overweight or obesity, emphasizing the role of metabolic disturbances, microcirculatory changes, and impaired bone tissue tropism in osteonecrosis pathogenesis. Harmful habits, including smoking or alcohol consumption, was present in 37.1% of patients, representing risk factors that exacerbate endothelial dysfunction, vascular spasm, and ischemic changes in bone tissue. Endocrine disorders was present in 30.5%, and 61.0% had one or more comorbid somatic conditions, confirming the multifactorial nature of osteonecrosis and the importance of systemic pathologies in its profession. Congenital anomalies were rare (6.7%), indicating that the primary pathogenic mechanisms mostly related to acquired risk factors.
|
|
|
3. Discussion
- Impaired microcirculation, endothelial dysfunction, and hypercoagulation among the leading pathogenetic factors in the development of femoral head osteonecrosis [3,5,7,13]. In recent years, interest has increased in the role of tg e endothelial nitric oxide (NO) system in the pathogenesis of this disease [7]. Functional polymorphisms in the NOS3 gene have been shown in multiple studies to play a key role in regulating vascular tone, platelet aggregation, and tissue perfusion [8].Based on literature data and genetic, clinical, and laboratory results got in this study, a pathogenetic algoritm fir the development of aseptic femoral head osteonecrosis, dependent on endothelial dysfunction proposed ( Figure 2).
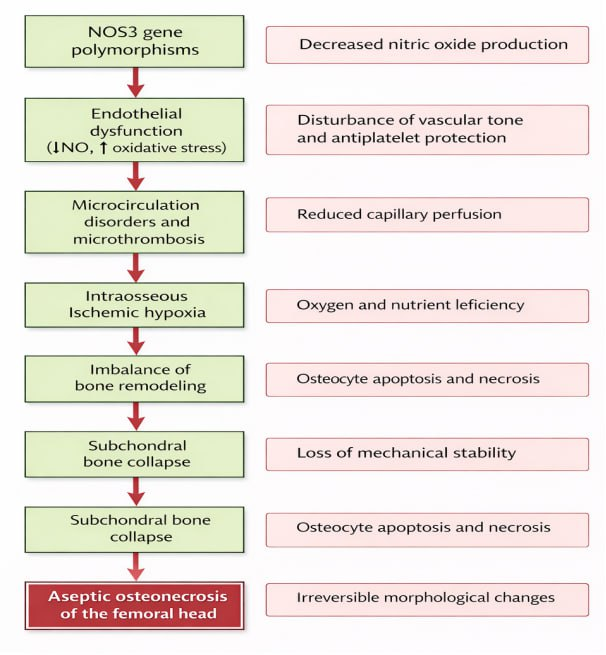 | Figure 2. Pathogenetic Algorithm of Aseptic Femoral Head Osteonecrosis Associated with NOS3 Gene Polymorphisms |
4. Conclusions
- 1. In the studied group, NOS3 gene polymorphisms rs2070744, rs1549758, and rs1799983 was significantly associated witg an increased risk of n femoral head osteonecrosis.2. Genetic variations manifested in clinical-laboratory changes characteristic of endothelial dysfunction, including inflammatory markers, coagulation parameters, and impaired microcirculation.3. In the patient group, disturbances in bone mineral metabolism, vitamin D₃ deficiency, and remodeling imbalance was observed as important factors in the pathogenesis of femoral head osteonecrosis.4. Genotype distributions fir all studied polymorphisms in the control group was consistent with Hardy–Weinberg equilibrium, confirming the reliability of the genetic data.5. The combined genetic, c clinical, and laboratory findings indicate that NOS3 gene polymorphisms play a crucial role in the development of femoral head osteonecrosis via endothelial disfunction.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML