-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 2142-2144
doi:10.5923/j.ajmms.20261604.119
Received: Feb. 20, 2026; Accepted: Mar. 15, 2026; Published: Apr. 29, 2026

Morphological and Immunohistochemical Alterations in the Upper Respiratory Tract Epithelium and Microvasculature Under Chronic Exposure to the Pesticide Nurinol
Yuldasheva Mokhigul Turdaliyevna
Fergana Medical Institute of Public Health, Fergana, Uzbekistan
Correspondence to: Yuldasheva Mokhigul Turdaliyevna, Fergana Medical Institute of Public Health, Fergana, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Chronic exposure to pesticides represents a critical and growing public health concern, particularly affecting the respiratory system as the primary interface for inhaled toxicants. The present study provides an in-depth analysis of morphological and immunohistochemical alterations in the mucous membrane of the upper respiratory tract following prolonged exposure to the pesticide Nurinol. Using an experimental inhalation model, progressive epithelial damage, microvascular disturbances, and immune dysregulation were identified. Histological examination revealed dystrophic, necrobiotic, and inflammatory changes accompanied by impaired mucociliary clearance. Immunohistochemical profiling demonstrated dynamic modulation of CD68, CD4, CD20, VEGFR, and Caspase-3 expression, reflecting macrophage activation, lymphocytic infiltration, angiogenic responses, and apoptosis induction. The severity of tissue injury correlated with exposure duration, progressing from adaptive-compensatory changes to irreversible atrophic and sclerotic remodeling. Mitochondrial dysfunction and ATP depletion emerged as key mechanisms underlying epithelial degeneration. These findings highlight the diagnostic and prognostic significance of integrated morphologic and immunohistochemical assessment in toxic respiratory pathology.
Keywords: Pesticides, Nurinol, Respiratory mucosa, Immunohistochemistry, Apoptosis, Microcirculation, Epithelial injury
Cite this paper: Yuldasheva Mokhigul Turdaliyevna, Morphological and Immunohistochemical Alterations in the Upper Respiratory Tract Epithelium and Microvasculature Under Chronic Exposure to the Pesticide Nurinol, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 2142-2144. doi: 10.5923/j.ajmms.20261604.119.
1. Introduction
- Chronic exposure to environmental toxicants, particularly pesticides, has become a major global health issue due to increased industrialization and agricultural intensification [1,2]. Among various organ systems, the respiratory tract is especially vulnerable, as it constitutes the primary route of entry for airborne toxic substances. The mucosal surface of the upper respiratory tract serves as both a physical and immunological barrier, making it a critical target for toxic injury [3].Pesticides such as organophosphates and pyrethroids exert complex biological effects, including oxidative stress, mitochondrial dysfunction, and immune dysregulation [1]. These mechanisms lead to structural and functional impairment of epithelial cells, disruption of mucociliary transport, and alterations in microvascular dynamics [4]. Chronic exposure further promotes persistent inflammation and progressive tissue remodeling, which may culminate in fibrosis and loss of functional integrity [5].Despite extensive research on systemic toxicity of pesticides, the morphological and immunohistochemical mechanisms of respiratory mucosal injury remain insufficiently characterized, particularly in relation to combined pesticide formulations such as Nurinol. Understanding these mechanisms is essential for early diagnosis, risk assessment, and development of targeted therapeutic interventions.Aim of the study:To investigate the time-dependent morphological and immunohistochemical changes in the upper respiratory tract mucosa under chronic inhalation exposure to the pesticide Nurinol.
2. Materials and Methods
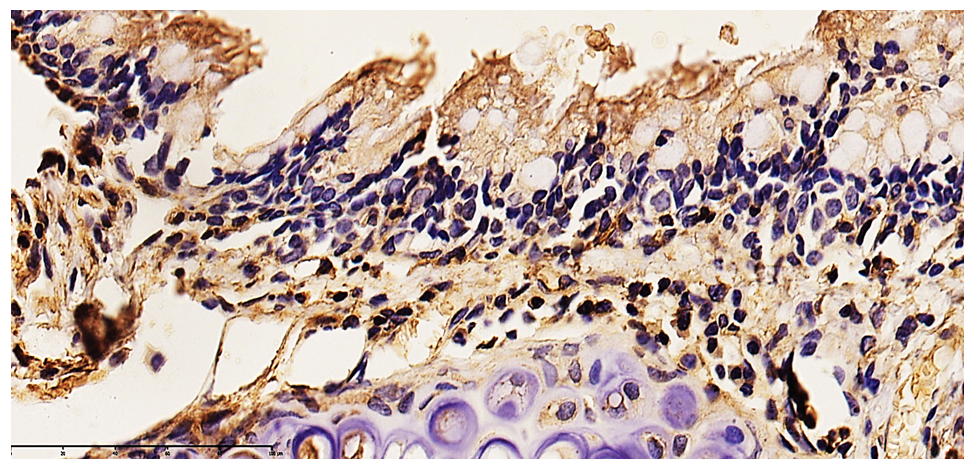
- The study was conducted on laboratory animals divided into control, experimental (30, 60, and 180 days), and treatment groups. An inhalation exposure model of pesticides was used [10].The following methods were applied:Clinical-functional methods (spirometry, body plethysmography)Imaging techniques (radiography, CT)Morphological methods (light and electron microscopy, immunohistochemistry) [8]Biochemical analysis (GDH, ALP, glucose levels)Immunohistochemical markers included CD68, CD4, CD20, Caspase-3, and VEGFR [7]. Evaluation was performed using the Remmele (IRS) scoring system. Toxicological analysis of biological media was also conducted. Statistical analysis was performed using standard methods.
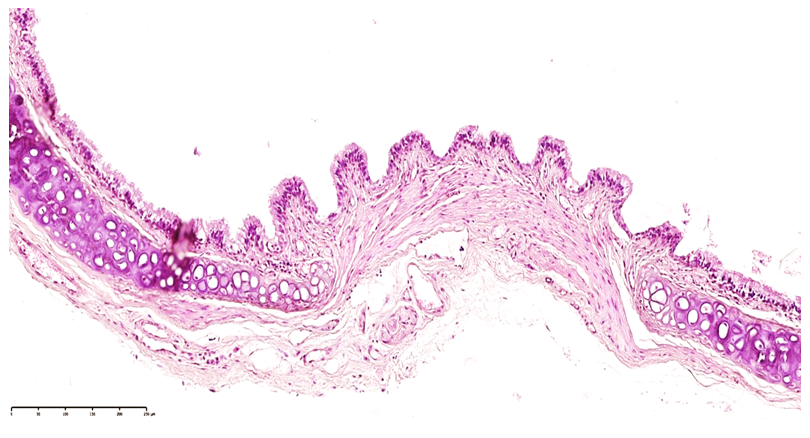
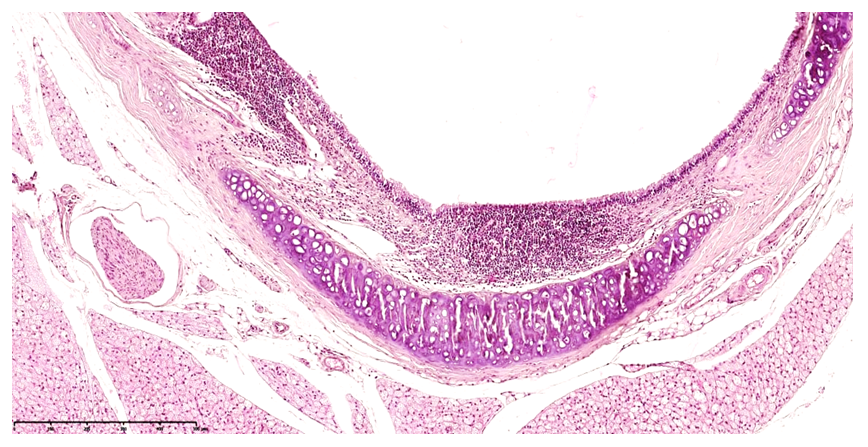
3. Results
- Under pesticide exposure, early stages are characterized by morphofunctional stress in the mucosa of the upper respiratory tract, including the nasal cavity, pharynx, trachea, and bronchi [3]. These changes largely depend on tissue sensitivity to the pesticide.Nurinol exposure leads to disturbances in neurovascular innervation. The morphological substrate includes metabolic disorders in epithelial cells, accompanied by dystrophic and necrobiotic changes [8]. Initially, vascular congestion and mild perivascular plasma edema are observed.
4. Discussion
- The findings of this study demonstrate a clear time-dependent progression of respiratory mucosal damage under chronic pesticide exposure.The early adaptive responses observed at 30 days reflect compensatory mechanisms aimed at maintaining epithelial integrity. However, prolonged exposure leads to exhaustion of these mechanisms, resulting in structural and functional failure.Activation of apoptotic pathways, as evidenced by Caspase-3 expression, plays a central role in epithelial degeneration. Simultaneously, immune activation (CD68, CD4, CD20) indicates both innate and adaptive immune involvement in tissue remodeling.A key pathogenic mechanism identified in this study is mitochondrial dysfunction, leading to ATP depletion and energy collapse. This explains the loss of ciliary activity and impaired mucociliary clearance, which are essential for respiratory defense [9].Furthermore, vascular changes mediated by VEGFR expression suggest angiogenic responses to hypoxia and tissue injury. Chronic inflammation and impaired regeneration ultimately result in fibrotic and sclerotic remodeling, consistent with previous findings in toxic respiratory pathology [5].These results align with global studies on pesticide toxicity, confirming the multifactorial nature of tissue injury involving oxidative stress, immune dysregulation, and apoptosis [1–3].
5. Conclusions
- The study demonstrated that chronic inhalation exposure to the pesticide Nurinol leads to significant morphological and immunohistochemical changes in the mucosa of the upper respiratory tract [1,2].Dystrophic, necrobiotic, and inflammatory processes develop, accompanied by impaired mucociliary transport, epithelial damage, and microcirculatory disturbances [4].Immunohistochemical analysis revealed altered expression of CD68, CD4, CD20, VEGFR, and Caspase-3, indicating activation of apoptosis, inflammation, and immune remodeling [6,7].The progression of morphological changes depends on exposure duration, ranging from adaptive responses to atrophic and sclerotic transformations [5].Disruption of energy metabolism in epithelial cells plays a key role, leading to functional failure and desquamation [9].These findings confirm that combined morphological and immunohistochemical analysis is an effective method for assessing toxic damage to the respiratory tract and may be useful for early diagnosis and prevention of pesticide-related pathologies.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML