-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 2138-2141
doi:10.5923/j.ajmms.20261604.118
Received: Mar. 29, 2026; Accepted: Apr. 23, 2026; Published: Apr. 29, 2026

Prevalence and Regional Patterns of Anemia Among Women of Early Reproductive Age in the Aral Sea Region: A Cross-Sectional Study from Khorezm, Uzbekistan
Guli Utkirovna Sultanmuratova1, Salomat Aleksandrovna Matyakubova2
1MD, PhD, Tashkent State Medical University, Tashkent, Uzbekistan
2MD, DSc, Khorezm Regional Branch of the Republican Specialized Scientific and Practical Medical Center for Mother and Child Health, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

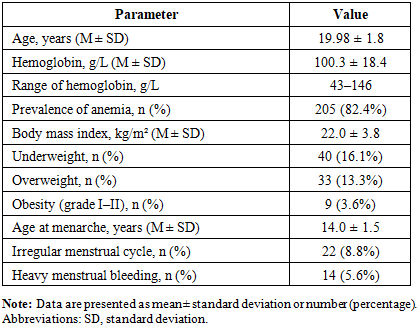
Objective: To assess the prevalence, severity, and regional patterns of anemia among women of early reproductive age in the ecologically vulnerable Aral Sea region of Uzbekistan, with a focus on iron and vitamin deficiencies. Methods: A cross-sectional study was conducted among 249 women aged 19–25 years across 11 districts of the Khorezm region. Laboratory assessment included complete blood count, serum ferritin, vitamin B12, and folate levels. In districts with the lowest hemoglobin levels, a structured questionnaire was used to evaluate dietary patterns and clinical risk factors. Results: Anemia was identified in 82.4% of participants, with a mean hemoglobin level of 100.3 ± 18.4 g/L. Marked regional heterogeneity was observed, with the lowest hemoglobin levels in Gurlen and Khazarasp districts. Anemia was predominantly microcytic and hypochromic and was strongly associated with depleted iron stores and a high prevalence of folate deficiency. The findings indicate a pattern of combined micronutrient deficiency in nutritionally vulnerable populations. Conclusion: Anemia among young women in the Aral Sea region is alarmingly prevalent and demonstrates significant regional variability. The coexistence of iron and folate deficiencies highlights the need for region-specific screening strategies and targeted nutritional interventions in environmentally disadvantaged settings.
Keywords: Anemia, Aral Sea region, Early reproductive age, Iron deficiency, Uzbekistan, Folic acid, Vitamin B12
Cite this paper: Guli Utkirovna Sultanmuratova, Salomat Aleksandrovna Matyakubova, Prevalence and Regional Patterns of Anemia Among Women of Early Reproductive Age in the Aral Sea Region: A Cross-Sectional Study from Khorezm, Uzbekistan, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 2138-2141. doi: 10.5923/j.ajmms.20261604.118.
Article Outline
1. Introduction
- Anemia remains one of the most common nutritional and hematological disorders among women of reproductive age worldwide and represents a major public health and clinical concern [1–3]. Iron deficiency is considered the leading cause of anemia; however, deficiencies of folic acid and vitamin B12 also contribute to impaired erythropoiesis, particularly in young women [4–7]. In reproductive-age women, anemia is associated with menstrual disorders, reduced physical and cognitive performance, impaired quality of life, and adverse pregnancy outcomes later in life [3,8].Regions exposed to environmental stress and socioeconomic disadvantage are characterized by a higher burden of micronutrient deficiencies [2,9]. The Khorezm region of Uzbekistan is located in the Aral Sea basin, an area affected by long-term ecological degradation, reduced agricultural diversity, and limited access to nutritionally adequate diets, which may contribute to the development of anemia and combined micronutrient deficiencies in young women [10–12].Despite the clinical relevance of anemia in reproductive-age women, data on its regional distribution, severity, and underlying micronutrient deficiencies in environmentally vulnerable areas remain limited. The present study aimed to assess the prevalence and structure of anemia among women of early reproductive age in the Khorezm region and to identify regional patterns associated with iron and vitamin deficiencies.
2. Materials and Methods
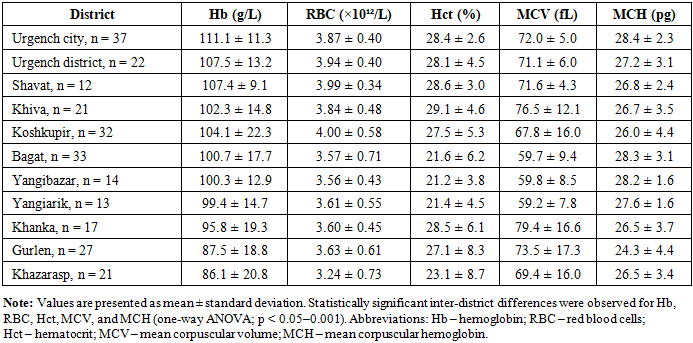
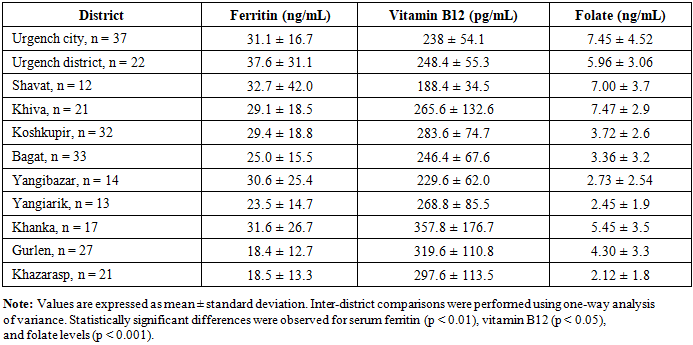
- Study design and participantsA cross-sectional study was conducted in the Khorezm region of Uzbekistan. A total of 249 women aged 19–25 years were included. Participants were permanent residents of 11 districts of the region. Participants were distributed across 11 districts as follows: Urgench city (n = 37), Urgench district (n = 22), Shavat (n = 12), Khiva (n = 21), Koshkupir (n = 32), Bagat (n = 33), Yangibazar (n = 14), Yangiarik (n = 13), Khanka (n = 17), Gurlen (n = 27), and Khazarasp (n = 21). Inclusion criteria were female sex, early reproductive age (19–25 years), permanent residence in the study area, and written informed consent. Women with acute inflammatory conditions, chronic systemic diseases, or diagnosed hematological disorders of non-nutritional origin and pregnant women were excluded from the study. The study protocol was reviewed and approved by the Local Ethics Committee of Tashkent State Medical University (Protocol No. 2, dated 28 January 2024). The study was conducted in accordance with the principles of the Declaration of Helsinki. Written informed consent was obtained from all participants prior to enrollment.Laboratory assessmentAll participants underwent venous blood sampling. Complete blood count was performed with determination of hemoglobin, erythrocyte count, hematocrit, leukocytes, platelets, and erythrocyte indices (MCV, MCH). Serum ferritin and vitamin B12 levels were measured using immunochemiluminescent assays, and serum folic acid was assessed by enzyme-linked immunosorbent assay. Laboratory values were interpreted according to standard reference ranges. Anemia was defined according to World Health Organization criteria as a hemoglobin concentration <120 g/L in non-pregnant women of reproductive age.Questionnaire surveyIn districts with the lowest mean hemoglobin levels (Gurlen, Khazarasp, and Khanka), a structured questionnaire was administered. The questionnaire assessed dietary habits, consumption of iron-rich foods, intake of tea and coffee, dietary diversity, previous diagnosis of anemia, and adherence to iron and vitamin supplementation.Statistical analysisStatistical analyses were performed using SPSS software (version 26.0; IBM Corp., Armonk, NY, USA). Quantitative variables are presented as mean ± standard deviation (SD). Inter-district comparisons were conducted using one-way analysis of variance (ANOVA) followed by Tukey’s post hoc test. A two-sided p-value < 0.05 was considered statistically significant.
3. Results
- General characteristics of the study populationThe mean age of participants was 19.98 ± 1.8 years. All women were students. The mean hemoglobin level in the overall cohort was 100.3 ± 18.4 g/L (range 43–146 g/L), and anemia was diagnosed in 82.4% of participants. Detailed demographic and clinical characteristics are presented in Table 1.
|
|
|
4. Discussion
- The present study demonstrates a markedly high prevalence of anemia (82.4%) among women of early reproductive age in the Khorezm region. This prevalence substantially exceeds the global estimate for non-pregnant women of reproductive age (approximately 29–33%) reported by the World Health Organization [1,2]. This discrepancy highlights the disproportionate burden of micronutrient deficiency in environmentally and socioeconomically vulnerable regions. The predominance of microcytic and hypochromic anemia across districts suggests iron deficiency as the principal underlying mechanism, consistent with findings from other low- and middle-income countries [4,10,11].Importantly, in several districts anemia was characterized not only by iron depletion but also by a high prevalence of folate deficiency, suggesting combined micronutrient deficiencies [6,7]. Similar patterns have been reported in nutritionally vulnerable populations, where dietary diversity is limited and access to supplementation is inadequate [8,9].The association between unfavorable nutritional habits, low adherence to iron and vitamin supplementation, and the severity of anemia observed in the present study aligns with FIGO recommendations emphasizing the importance of micronutrient sufficiency before pregnancy [9]. These findings highlight the need for region-specific screening and preventive strategies targeting women of reproductive age, particularly in environmentally disadvantaged areas such as the Aral Sea region [10–12].The markedly high prevalence of anemia identified in this study has important public health implications for the Aral Sea region. Early reproductive age represents a critical window for optimizing micronutrient status prior to future pregnancy. Failure to address iron and folate deficiencies at this stage may contribute to adverse maternal and perinatal outcomes. Region-specific screening programs, nutritional education, and targeted supplementation strategies should therefore be integrated into primary health care services for young women living in environmentally vulnerable areas.This study has several limitations. First, its cross-sectional design does not allow causal inference between nutritional factors and anemia. Second, the study population consisted exclusively of students, which may limit the generalizability of the findings to all women of reproductive age in the region. Third, inflammatory markers were not assessed; therefore, serum ferritin levels may have been influenced by subclinical inflammation. Finally, dietary assessment was based on self-reported data and may be subject to recall bias.Overall, anemia among women of early reproductive age in the Khorezm region is highly prevalent and exhibits marked regional heterogeneity. In environmentally vulnerable districts, anemia is frequently associated with combined iron and folate deficiency. These findings underscore the urgent need for region-specific screening and preventive strategies targeting women prior to pregnancy.
Disclosure
- This article has not been previously presented at any conference and has not been published elsewhere in abstract or full-text form.
Ethics Statement
- The study protocol was reviewed and approved by the Ethics Committee of the Khorezm Regional Branch of the Republican Specialized Scientific and Practical Medical Center for Mother and Child Health (Uzbekistan).The study was conducted in accordance with the principles of the Declaration of Helsinki.
Consent
- Written informed consent was obtained from all participants prior to enrollment in the study. No patient-identifiable data are included in this manuscript.
Conflicts of Interest
- The authors declare no conflicts of interest.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML