-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 2101-2104
doi:10.5923/j.ajmms.20261604.109
Received: Mar. 26, 2026; Accepted: Apr. 20, 2026; Published: Apr. 28, 2026

Comprehensive Morphometric and Morphological Assessment of the Juxtaglomerular Apparatus in Experimental Obstructive Hydronephrosis
Tursunbaev Jasur Anvarovich1, Ermatov Nizom Jumakulovich2, Rasulov Hamidulla Abdullaevich2
1Tashkent State Medical University Termez Branch, Termez, Uzbekistan
2Tashkent State Medical University, Tashkent, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

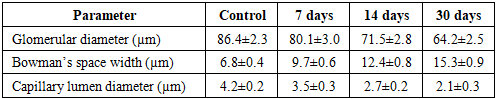
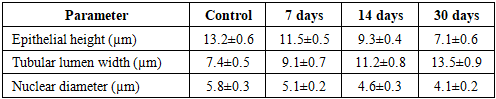
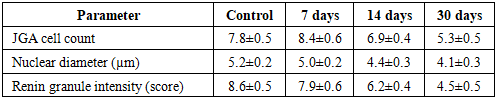
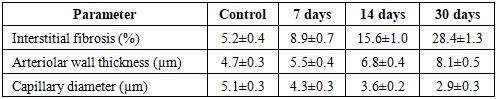
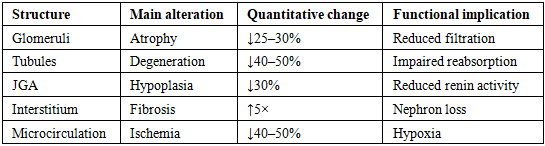
Hydronephrosis is a progressive pathological condition characterized by urinary outflow obstruction, leading to increased intrarenal pressure, impaired renal hemodynamics, and gradual structural damage of kidney tissue. According to recent epidemiological data, obstructive uropathies account for a significant proportion of chronic kidney disease cases worldwide, affecting approximately 10–13% of the population. Among the most sensitive renal structures involved in early pathological changes is the juxtaglomerular apparatus (JGA), which plays a crucial role in regulating the renin–angiotensin system and maintaining glomerular filtration. The aim of this study was to investigate the morphological and morphometric changes of the juxtaglomerular apparatus in experimental hydronephrosis and to evaluate their progression over time. The study was conducted on 60 adult male laboratory rats divided into control, experimental, and recovery groups. Hydronephrosis was induced by partial ureteral ligation, and renal tissue was examined at 7, 14, and 30 days. Histological and morphometric analyses were performed using standard staining techniques and digital image processing. The results demonstrated progressive structural alterations, including glomerular atrophy, tubular degeneration, interstitial fibrosis, and significant changes in JGA parameters. Morphometric analysis revealed a 25–30% decrease in glomerular diameter, a twofold increase in Bowman’s space, and a 40–50% reduction in tubular epithelial height. The JGA showed an initial compensatory increase in cell number, followed by a marked decline and reduced renin granule density (up to 47%) in later stages. Interstitial fibrosis increased more than fivefold, indicating the development of nephrosclerosis.
Keywords: Hydronephrosis, Kidney, Juxtaglomerular apparatus, Morphometry, Renal ischemia, Nephrosclerosis, Tubular degeneration, Interstitial fibrosis
Cite this paper: Tursunbaev Jasur Anvarovich, Ermatov Nizom Jumakulovich, Rasulov Hamidulla Abdullaevich, Comprehensive Morphometric and Morphological Assessment of the Juxtaglomerular Apparatus in Experimental Obstructive Hydronephrosis, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 2101-2104. doi: 10.5923/j.ajmms.20261604.109.
1. Introduction
- Chronic kidney diseases represent a significant global health burden, affecting approximately 10–13% of the worldwide population and contributing substantially to morbidity and mortality [1,3,5]. Among these conditions, obstructive uropathies, particularly hydronephrosis, play a critical role due to their progressive and often irreversible impact on renal structure and function [2,7]. Hydronephrosis arises from partial or complete obstruction of urinary outflow, leading to increased intrapelvic pressure, impaired renal hemodynamics, tissue hypoxia, and eventual nephron loss [9,10].Recent experimental and clinical studies emphasize that early pathological alterations in hydronephrosis primarily affect microstructural components of the kidney, especially the juxtaglomerular apparatus (JGA), which serves as a key regulator of the renin–angiotensin system and maintains glomerular filtration homeostasis [4,9,12]. The JGA comprises specialized juxtaglomerular cells, macula densa, and extraglomerular mesangial cells, functioning as a sensor-effector system responding to changes in perfusion pressure and sodium concentration [13].Dolgov et al. (2018) demonstrated that experimental hydronephrosis initially induces hypertrophy and hyperactivity of JGA cells, followed by progressive degeneration and reduced renin secretion in later stages [15]. Similarly, Kang et al. (2021) reported that prolonged obstruction results in microvascular remodeling, interstitial fibrosis, and functional suppression of the JGA [8]. Nakamura et al. (2020) further confirmed that ischemic injury leads to structural disorganization and decreased cellular density within the JGA, impairing autoregulatory mechanisms [11].Despite these advances, the dynamic morphometric alterations of the JGA during different stages of hydronephrosis remain insufficiently characterized. A comprehensive quantitative evaluation of these changes is essential for understanding disease progression and identifying early diagnostic markers.Aim of the study: To investigate morphological and morphometric changes of the juxtaglomerular apparatus in experimental hydronephrosis and to perform a comparative analysis across different time stages.
2. Materials and Methods
- This experimental study was conducted on 60 adult male white laboratory rats weighing 200–250 g. The animals were randomly divided into three groups: control group (n=20), experimental hydronephrosis group (n=20), and recovery group (n=20).Experimental hydronephrosis was induced by partial ligation of the ureter under general anesthesia using ketamine (50 mg/kg). The obstruction was created in a sterile surgical environment, ensuring partial urinary flow restriction. Animals were observed over 7, 14, and 30 days.At designated time points, animals were euthanized following AVMA (2020) guidelines. Kidney tissues were collected from cortical and juxtamedullary regions and fixed in 10% neutral buffered formalin for 24 hours. Samples were processed through graded ethanol dehydration and embedded in paraffin blocks. Sections of 4–5 µm thickness were prepared.Histological staining included hematoxylin–eosin for general morphology, PAS staining for basement membranes, and Van Gieson staining for collagen fibers. Microscopy was performed using Leica and Zeiss microscopes at magnifications of 40× and 100×.Morphometric analysis was carried out using ImageJ software. Parameters measured included glomerular diameter, Bowman’s space width, tubular epithelial height, number of JGA cells, nuclear diameter, interstitial fibrosis percentage, and arteriolar wall thickness. Data were expressed as mean ± standard error (M±m), and statistical analysis was performed using Student’s t-test with significance set at p<0.05.
3. Results
- The experimental study revealed a consistent and progressive pattern of morphological and morphometric alterations in renal tissue under conditions of obstructive hydronephrosis. The obtained data clearly demonstrate that the pathological process evolves through sequential stages, beginning with adaptive-compensatory responses and progressing toward irreversible degenerative and fibrotic transformations.During the early phase (7 days), structural alterations were relatively mild and primarily involved microcirculatory disturbances. Histological examination revealed endothelial cell swelling, vacuolization of cytoplasm, and mild mesangial expansion. The juxtaglomerular apparatus (JGA) exhibited signs of functional activation, characterized by a slight increase in cell number and preserved renin granule density.At 14 days, the pathological changes became more pronounced. Glomerular architecture appeared disorganized, Bowman’s space widened significantly, and tubular epithelial degeneration intensified. At this stage, compensatory mechanisms began to decline, and early signs of functional impairment became evident.By day 30, advanced degenerative changes dominated the renal tissue. Extensive interstitial fibrosis, tubular atrophy, glomerulosclerosis, and significant depletion of JGA cellular elements were observed. These findings indicate the transition from reversible injury to irreversible structural damage.
|
|
|
|
|
4. Discussion
- The present study provides a comprehensive morphometric and morphological analysis of renal alterations in experimental hydronephrosis, with a particular focus on the juxtaglomerular apparatus. The findings clearly demonstrate that hydronephrosis induces a cascade of structural and functional changes that evolve progressively over time, beginning with compensatory mechanisms and culminating in irreversible tissue damage.At the early stage (7 days), the observed increase in JGA cell number and relative preservation of renin granules suggest a compensatory activation of the renin–angiotensin system. This response likely represents an adaptive mechanism aimed at maintaining glomerular perfusion and filtration pressure under conditions of partial urinary obstruction. Similar findings have been reported by Dolgov et al. (2018), who described early hypertrophic changes in JGA cells as a protective response to reduced renal blood flow.However, as the obstruction persists, this compensatory capacity becomes exhausted. By day 14 and especially day 30, there is a significant decline in JGA cellularity, nuclear size, and renin granule density. These changes indicate functional deactivation of the JGA and disruption of renal autoregulatory mechanisms. Nakamura et al. (2020) similarly observed that prolonged ischemia leads to structural disorganization of the JGA, impairing its regulatory function.Glomerular alterations observed in this study, including reduced diameter and expanded Bowman’s space, reflect progressive ischemic injury and decreased filtration efficiency. Kang et al. (2021) emphasized that chronic obstruction results in glomerulosclerosis driven by sustained hypoperfusion and endothelial dysfunction. The narrowing of capillary lumens further supports the presence of severe microcirculatory compromise.Tubular changes are equally significant, with marked epithelial atrophy, lumen dilation, and nuclear shrinkage. These findings indicate impaired tubular transport and reabsorptive function. Anderson and O’Connor (2019) attributed such changes to mitochondrial dysfunction and energy depletion under ischemic conditions, leading to apoptosis and tubular collapse.Interstitial fibrosis emerges as one of the most critical pathological features in later stages. The dramatic increase in fibrotic tissue observed in this study confirms that hydronephrosis ultimately leads to nephrosclerosis. Chevalier (2019) highlighted interstitial fibrosis as the final common pathway of chronic kidney injury, resulting in irreversible loss of functional nephrons.Importantly, the integrated morphometric analysis reveals that all renal compartments are affected simultaneously and interdependently. The decline in JGA function appears closely linked to vascular remodeling and glomerular hypoperfusion, suggesting that disruption of the renin–angiotensin system plays a central role in disease progression.Thus, the results of this study not only confirm previous findings but also provide new quantitative insights into the dynamic structural changes occurring in hydronephrosis. The use of morphometric parameters offers a sensitive and objective method for evaluating disease progression and may serve as a valuable tool for early diagnosis and therapeutic monitoring.
5. Conclusions
- The study demonstrates that experimental hydronephrosis induces progressive and interconnected morphological and morphometric alterations in the kidney, affecting glomerular, tubular, interstitial, and juxtaglomerular structures. Initially, adaptive responses such as hypertrophy of JGA cells and activation of the renin–angiotensin system are observed; however, prolonged obstruction leads to functional exhaustion, cellular degeneration, and structural disintegration. The significant reduction in glomerular size, tubular epithelial height, and JGA activity, along with a marked increase in interstitial fibrosis, confirms the development of nephrosclerosis. Morphometric analysis proves to be a highly sensitive method for detecting early pathological changes and evaluating disease dynamics. These findings provide important insights into the pathogenesis of hydronephrosis and may contribute to the development of early diagnostic strategies and targeted therapeutic interventions.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML