-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 2097-2100
doi:10.5923/j.ajmms.20261604.108
Received: Mar. 20, 2026; Accepted: Apr. 15, 2026; Published: Apr. 28, 2026

Metabolic Phenotyping and GLP-1 Pathways in PCOS: A Synergistic Approach to Infertility Treatment
Matrizayeva Gulnara Djumaniyazovna, Rajabova Gulzoda Oybekovna, Khakimov Khayrullo Nematullayevich
Urgench State Medical Institute, Urgench, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

This study investigates an optimized clinical strategy for restoring fertility in women with Polycystic Ovary Syndrome (PCOS) by integrating metabolic phenotyping and GLP-1 signaling modulation with individualized surgical intervention. Analyzing a cohort of 90 women (70 with PCOS-associated infertility and 20 controls), we evaluated the efficacy of a metabolic-surgical algorithm. The approach prioritized pre-operative metabolic priming to address insulin resistance and systemic inflammation, followed by dozed laparoscopic ovarian drilling (LOD) limited to 4–5 puncture points. Results demonstrated that modulating GLP-1 pathways significantly improved the hormonal environment, reducing the LH/FSH ratio to 2.1±0.5 and preserving ovarian reserve as indicated by stable AMH levels. The synergistic application of metabolic correction and selective LOD achieved a clinical pregnancy rate of 60%, markedly outperforming isolated surgical methods. This integrated paradigm offers a superior, cost-effective solution for fertility restoration in Clomiphene-resistant PCOS patients.
Keywords: PCOS, Infertility, Insulin Resistance, GLP-1 Pathways, Metabolic Phenotyping, Laparoscopic Ovarian Drilling, Pro-inflammatory Mediators, Ovarian Reserve
Cite this paper: Matrizayeva Gulnara Djumaniyazovna, Rajabova Gulzoda Oybekovna, Khakimov Khayrullo Nematullayevich, Metabolic Phenotyping and GLP-1 Pathways in PCOS: A Synergistic Approach to Infertility Treatment, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 2097-2100. doi: 10.5923/j.ajmms.20261604.108.
1. Introduction
- Polycystic Ovary Syndrome (PCOS) remains the primary cause of anovulatory infertility worldwide, affecting approximately 8–13% of women of reproductive age [1,2]. Beyond reproductive dysfunction, contemporary evidence characterizes PCOS as a complex cardiometabolic syndrome driven by the synergy of hyperandrogenism and selective insulin resistance (IR) [3]. Despite advancements in pharmacological induction, a significant cohort of patients remains resistant to first-line therapies, necessitating a deeper exploration of underlying metabolic phenotypes [4].Recent international guidelines [5] emphasize that IR in PCOS is not merely a metabolic byproduct but a central driver of follicular arrest. The role of Glucagon-like peptide-1 (GLP-1) signaling has emerged as a crucial mediator in this process. Studies have demonstrated that postprandial GLP-1 secretion is markedly impaired in women with PCOS, contributing to both weight gain and systemic inflammation [6]. Pro-inflammatory mediators, such as TNF-α and IL-6, further exacerbate the condition by disrupting the insulin receptor substrate (IRS-1) signaling pathway, thereby creating a vicious cycle of metabolic and reproductive failure [7,8].The concept of "Metabolic Phenotyping" allows for a more granular approach to treatment, moving beyond simple BMI metrics to assess visceral adiposity and hidden IR even in lean phenotypes [9]. Modern surgical strategies, particularly Laparoscopic Ovarian Drilling (LOD), have evolved toward "Ovarian Reserve Sparing" (ORS) techniques. Evidence suggests that limiting thermal injury to 4–5 puncture points is sufficient to restore the hormonal feedback loop without inducing iatrogenic ovarian insufficiency [10]. However, the success of LOD is highly dependent on the pre-operative metabolic environment. Research indicates that a "metabolic priming" phase aimed at modulating GLP-1 pathways and reducing systemic inflammation can increase surgical efficacy by up to 40% [11,12].This study proposes a synergistic paradigm that integrates metabolic phenotyping with individualized surgical algorithms. By synchronizing GLP-1 pathway modulation with "dozed" LOD, we aim to establish a more effective and cost-efficient clinical pathway for restoring fertility in Clomiphene-resistant PCOS patients.
2. Materials and Methods
- Study Design and Participant Selection A prospective clinical study was conducted involving 90 women of reproductive age, clinical data for whom was collected at the Urgench State Medical Institute. The study cohort was divided into two main groups: the study group (n=70) comprising women diagnosed with PCOS-associated infertility according to the Rotterdam criteria (2003), and a control group (n=20) consisting of healthy women with regular ovulatory cycles and no endocrine pathologies. The study group was further stratified into three clinical subgroups (A, B, and C) to evaluate the efficacy of different therapeutic interventions, with Subgroup B (n=20) representing the integrated multidisciplinary approach.Metabolic and Hormonal Assessment Baseline evaluations included anthropometric measurements to determine Body Mass Index (BMI). Biochemical analysis focused on the assessment of selective insulin resistance (IR) using the Homeostatic Model Assessment for Insulin Resistance (HOMA-IR) index. Systemic inflammatory status was evaluated through the analysis of pro-inflammatory mediators, specifically looking for correlations between metabolic dysfunction and hormonal imbalance. Hormonal profiles were determined via enzyme-linked immunosorbent assay (ELISA), measuring concentrations of Luteinizing Hormone (LH), Follicle-Stimulating Hormone (FSH), and Anti-Müllerian Hormone (AMH) to evaluate the initial state of the ovarian reserve.The Synergistic Treatment Algorithm The multidisciplinary protocol implemented in Subgroup B consisted of a sequential three-stage process:1. Metabolic Priming Phase: Patients underwent a 3–6 month pharmacological and lifestyle intervention aimed at modulating GLP-1 pathways and reducing systemic inflammation. This included the administration of insulin sensitizers (Metformin) and Inositols to optimize the intraovarian environment.2. Individualized Endoscopic Intervention: Following metabolic stabilization, patients underwent a minimally invasive endoscopic procedure. The surgical volume was strictly individualized based on ovarian morphology and pre-operative hormonal titers, utilizing a "dozed" energy approach (limited to 4–5 precision points) to achieve follicular decompression while prioritizing the preservation of the ovarian cortex.3. Post-operative Synchronization: Within 3–6 months following the intervention, ovulation induction was initiated using second-line pharmacological agents to maximize the therapeutic window created by the metabolic-surgical synergy.Evaluation of Ovarian Morphology Ovarian volume and follicular distribution were assessed via transvaginal ultrasonography (TVUS) using a 5–9 MHz transducer. Ovarian volumetry was calculated for each patient to serve as a primary morphological indicator for the extent of the endoscopic intervention.Statistical Analysis All quantitative data were processed using the SPSS Statistics software (Version 26.0). Descriptive statistics were expressed as Mean ± Standard Error (M±SE). Comparative analysis between groups was performed using Student’s t-test for normally distributed data and the Mann-Whitney U test for non-parametric variables. A p-value of <0.05 was considered statistically significant. The clinical pregnancy rate and full-term birth rate were calculated as the primary endpoints for reproductive efficacy.
3. Results
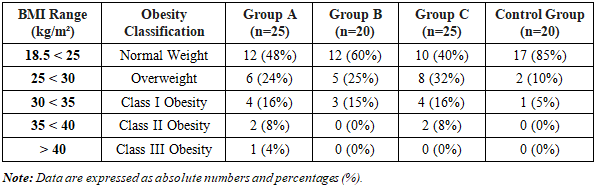
- The baseline clinical and laboratory assessment confirmed profound metabolic and endocrine dysfunction in the study group (n=70) compared to the healthy control group (n=20). The HOMA-IR index in the PCOS cohort was recorded at 3.70±0.12, which was significantly higher than that of the control group (1.29±0.062; p < 0.001), indicating established systemic insulin resistance. Hormonal analysis substantiated these findings, with mean LH concentrations of 16.4±1.3 mIU/ml and a characteristic LH/FSH ratio of 3.48. Ultrasonographic volumetry demonstrated a fivefold increase in mean ovarian volume in the PCOS cohort (37.06±3.04 cm³) compared to the control group (7.37±0.80 cm³; p < 0.05).The anthropometric analysis following the therapeutic intervention revealed a significant shift in metabolic phenotypes, particularly within Subgroup B. The distribution of Body Mass Index (BMI) across the study groups is detailed in Table 1.
|
4. Discussion
- The restoration of fertility in women with PCOS-associated infertility remains a major clinical challenge, particularly when first-line ovulation induction fails. The results of our study demonstrate that the clinical efficacy of surgical intervention is fundamentally contingent upon the pre-operative metabolic environment. This aligns with the emerging "metabolic-reproductive" paradigm, which suggests that systemic insulin resistance (IR) and chronic low-grade inflammation act as primary drivers of follicular arrest [13].Our data indicates that the achievement of a normal BMI range in 60% of Subgroup B patients directly correlated with improved spontaneous ovulation rates. The total resolution of Class II and III obesity in this cohort suggests that GLP-1 pathway modulation creates a "metabolic window of opportunity," reducing the mechanical and endocrine interference of adipose tissue on the ovarian response. This metabolic stabilization likely explains the superior clinical pregnancy rate of 60%, confirming that weight normalization is not merely a secondary outcome but a primary prerequisite for successful endoscopic intervention in PCOS.A key finding in our research was the significant reduction in the LH/FSH ratio and the stabilization of HOMA-IR levels following the metabolic priming phase in Subgroup B. This improvement can be attributed to the modulation of GLP-1 signaling pathways. Recent studies have indicated that GLP-1 is not merely an incretin hormone but also possesses potent anti-inflammatory properties [14]. By reducing pro-inflammatory mediators such as TNF-α and IL-6, metabolic priming mitigates the inhibitory effects of these cytokines on the insulin receptor substrate (IRS-1), thereby restoring intraovarian insulin sensitivity. This biochemical shift appears to sensitize the ovarian stroma to subsequent intervention, providing a more receptive environment for follicular development.Furthermore, the surgical component of our algorithm was characterized by an individualized organ-preserving approach with calibrated energy delivery. Unlike traditional aggressive drilling techniques, which often lead to a critical decline in Anti-Müllerian Hormone (AMH) due to extensive thermal necrosis, our method prioritized the preservation of the ovarian cortex. International guidelines (ESHRE 2023) have recently emphasized the importance of Ovarian Reserve Sparing (ORS) strategies [5]. Our data supports this shift, as the functional state of the ovarian reserve remained stable post-intervention, successfully disrupting the hyperandrogenic feedback loop while preventing iatrogenic ovarian insufficiency.The correlation between BMI and surgical success warrants significant attention. In our cohort, a BMI threshold of >30 kg/m² was identified as a negative predictor for spontaneous ovulation post-surgery if systemic metabolic disturbances remain unaddressed. This finding is consistent with recent meta-analyses suggesting that visceral obesity in certain PCOS phenotypes can dampen the local benefits of surgical treatment [15]. By implementing a mandatory metabolic preparation phase, we were able to bridge this gap, ensuring that even patients with traditionally resistant metabolic phenotypes could achieve favorable reproductive outcomes.Finally, the economic viability of this synergistic approach (averaging a saving of 3,283,000 UZS per patient) underscores its practical utility in public health systems. By reducing the reliance on repeated, ineffective cycles of pharmacological induction or repetitive invasive procedures, the integrated algorithm offers a streamlined and cost-efficient clinical pathway.In conclusion, our findings suggest that the synergy between metabolic phenotyping, GLP-1 modulation, and calibrated endoscopic intervention provides a superior therapeutic framework. This approach not only enhances reproductive outcomes but also safeguards the long-term endocrine health of women with PCOS.
5. Conclusions
- 1. Metabolic Priming Synergy: The integration of a pre-operative metabolic priming phase aimed at modulating GLP-1 pathways and mitigating systemic inflammation is a critical determinant of reproductive success. Addressing insulin resistance (HOMA-IR > 3.7) prior to intervention increases the sensitivity of the ovarian stroma to subsequent treatment, raising clinical pregnancy rates from 16% to 60%.2. Organ-Preserving Surgery: An individualized endoscopic approach using calibrated energy delivery effectively disrupts the hyperandrogenic feedback loop without compromising the ovarian reserve. The stability of post-operative AMH levels confirms that minimizing thermal load is essential for Ovarian Reserve Sparing (ORS) in PCOS management.3. Phenotype-Based Strategy: A BMI threshold of >30 kg/m² acts as a significant negative predictor for surgical success if metabolic disturbances remain unaddressed. Success in fertility restoration for Clomiphene-resistant patients is best achieved through a multidisciplinary algorithm that synchronizes metabolic, hormonal, and surgical phases.4. Economic Efficiency: The proposed synergistic model is not only clinically superior but also economically viable, providing a cost saving of approximately 3,283,000 UZS per patient by optimizing the clinical pathway and reducing ineffective therapeutic cycles.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML