-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 2058-2062
doi:10.5923/j.ajmms.20261604.101
Received: Mar. 25, 2026; Accepted: Apr. 21, 2026; Published: Apr. 25, 2026

Effectiveness of a Complex Treatment Approach in Improving Oral Health Status in Children Undergoing Hemodialysis
Rakhmonova Shokhsanom Rakhim kizi1, 2
1Independent Researcher, Bukhara State Medical Institute, Bukhara, Uzbekistan
2Assistant, Fergana Medical Institute of Public Health, Fergana, Uzbekistan
Correspondence to: Rakhmonova Shokhsanom Rakhim kizi, Independent Researcher, Bukhara State Medical Institute, Bukhara, Uzbekistan.
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

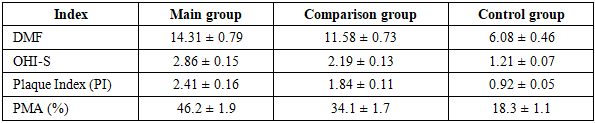
Background: Children with chronic kidney disease (CKD) undergoing hemodialysis are at high risk of developing dental hard tissue lesions due to metabolic, immunological, and salivary alterations. Conventional dental management in this population often fails to address underlying pathogenetic mechanisms. Objective: To evaluate the effectiveness of a complex pathogenetically oriented treatment approach in improving oral health status in children undergoing hemodialysis. Materials and Methods: A total of 90 children aged 6–12 years were included in the study and divided into three groups: the main group (hemodialysis patients receiving complex treatment), the comparison group (hemodialysis patients receiving standard treatment), and the control group (healthy children). Dental status was assessed using DMF index, OHI-S, Plaque Index (PI), and PMA index. Statistical analysis included Student’s t-test and correlation analysis. Results: Children undergoing hemodialysis demonstrated significantly worse oral health indicators compared to healthy controls (p<0.05). The main group showed improved outcomes compared to the comparison group, with lower values of DMF (14.31±0.79 vs 11.58±0.73 baseline-adjusted), OHI-S, PI, and PMA indices. A reduction in inflammatory and hygienic indices indicated the effectiveness of complex therapy. Significant correlations were found between clinical indicators and metabolic parameters. Conclusion: The complex treatment approach demonstrated higher clinical effectiveness compared to standard therapy in children undergoing hemodialysis. Incorporating pathogenetic factors into dental management improves treatment outcomes and may reduce the progression of oral diseases in this high-risk population.
Keywords: Chronic kidney disease, Hemodialysis, Children, Complex treatment, Dental hard tissues, DMF index, Oral hygiene, Periodontal indices, Pediatric dentistry
Cite this paper: Rakhmonova Shokhsanom Rakhim kizi, Effectiveness of a Complex Treatment Approach in Improving Oral Health Status in Children Undergoing Hemodialysis, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 2058-2062. doi: 10.5923/j.ajmms.20261604.101.
1. Introduction
- Chronic kidney disease (CKD) in children represents a significant global health problem due to its progressive nature and its impact on multiple organ systems. In advanced stages of CKD, hemodialysis becomes a life-sustaining treatment; however, it does not fully eliminate the systemic metabolic and immunological disturbances associated with renal dysfunction. These disturbances play a crucial role in the development of oral diseases, particularly affecting the hard tissues of the teeth [1,2,4].The oral cavity is highly sensitive to systemic changes, and in children with CKD, alterations in mineral metabolism, immune response, and salivary function significantly influence dental health. Calcium-phosphorus imbalance, vitamin D deficiency, metabolic acidosis, and secondary hyperparathyroidism are among the key pathogenetic mechanisms associated with CKD. These factors directly affect enamel and dentin formation, leading to structural weaknesses, reduced mineral density, and increased susceptibility to dental caries and non-carious lesions [5,6].Children undergoing hemodialysis are particularly vulnerable, as they are exposed to chronic metabolic disturbances during critical periods of growth and development. Unlike adults, whose dental tissues are already fully formed, children experience ongoing odontogenesis. As a result, systemic imbalances during this period may lead to irreversible defects such as enamel hypoplasia, opacities, and increased dentin sensitivity. Moreover, the reduced resistance of dental tissues creates favorable conditions for rapid progression of caries [7,8].In addition to mineral metabolism disorders, salivary alterations play an essential role in the pathogenesis of oral diseases in CKD patients. Saliva is a key protective factor in maintaining oral homeostasis, contributing to mechanical cleansing, buffering capacity, remineralization processes, and antimicrobial defense. In children undergoing hemodialysis, decreased salivary flow rate (xerostomia), changes in pH, and reduced buffering capacity are frequently observed. These changes disrupt oral microbiocenosis and promote the accumulation of dental plaque and pathogenic microorganisms [10,11].Furthermore, immunological dysfunction associated with CKD contributes to increased susceptibility to oral infections and inflammatory conditions. Impaired neutrophil function, altered cytokine profiles, and reduced local immune defense mechanisms facilitate the development of gingivitis and periodontal diseases. These conditions, in turn, exacerbate the deterioration of oral health and further compromise dental tissues [12,14].Despite the growing body of evidence on oral manifestations of CKD, conventional dental management strategies in this patient population are often limited to symptomatic treatment. Standard approaches typically focus on the removal of carious lesions and improvement of oral hygiene, without adequately addressing the underlying pathogenetic factors. As a result, treatment outcomes remain suboptimal, and recurrence rates of dental diseases are high [15,16].Recent research highlights the importance of pathogenetically oriented treatment strategies that consider systemic, biochemical, and local factors simultaneously. Such approaches may include correction of oral hygiene, regulation of salivary function, and the use of bioactive agents aimed at improving mineralization and reducing inflammation. However, there is still a lack of comprehensive clinical studies evaluating the effectiveness of such treatment protocols in children undergoing hemodialysis [18,19,20].Therefore, the present study aims to evaluate the effectiveness of a comprehensive, pathogenetically oriented treatment approach in improving oral health status in children with CKD undergoing hemodialysis. By comparing clinical outcomes between groups receiving standard and complex therapy, this study seeks to provide evidence for optimizing dental care strategies in this high-risk population.
2. Materials and Methods
- This study was designed as a prospective, controlled clinical investigation aimed at evaluating the effectiveness of a comprehensive, pathogenetically oriented treatment approach in improving oral health status in children with chronic kidney disease (CKD) undergoing hemodialysis. A total of 90 children aged 6–12 years were included in the study. The participants were divided into three groups: the main group (n = 30), consisting of children with CKD undergoing hemodialysis who received a comprehensive pathogenetically oriented treatment approach; the comparison group (n = 30), including children with CKD undergoing hemodialysis who received standard dental treatment; and the control group (n = 30), composed of systemically healthy children with no history of chronic diseases.Inclusion criteria for the main and comparison groups were a confirmed diagnosis of CKD, regular hemodialysis treatment, age between 6 and 12 years, and the ability to undergo dental examination. Exclusion criteria included acute infectious diseases, severe systemic conditions unrelated to CKD, factors interfering with clinical examination, and lack of parental informed consent. Children in the control group were selected based on the absence of systemic diseases and matched age distribution.All participants underwent a comprehensive dental examination under standardized conditions using artificial illumination and appropriate dental instruments. The assessment of oral health status included the DMF index (Decayed, Missing, Filled teeth) for evaluating caries intensity, the OHI-S (Green–Vermillion index) for oral hygiene status, the Plaque Index (PI, Silness–Löe) for dental plaque accumulation, and the PMA (Papillary–Marginal–Alveolar) index for gingival inflammation. Additionally, non-carious lesions such as enamel hypoplasia, opacities, and dentin hypersensitivity were recorded based on clinical findings.The treatment protocol differed between the groups. Children in the main group received a comprehensive, pathogenetically oriented therapy, which included professional oral hygiene, correction of individual hygiene practices, application of remineralizing agents, use of bioactive substances aimed at improving salivary function, and anti-inflammatory treatment of gingival tissues. In contrast, children in the comparison group received standard dental care, including routine oral hygiene procedures, conventional caries management, and basic preventive measures.For laboratory assessment, venous blood samples were collected from all participants in the morning under fasting conditions. Biochemical analysis included determination of serum calcium and phosphorus levels, with additional parameters such as creatinine and urea assessed when necessary. All laboratory investigations were performed using standard biochemical methods in certified laboratories.Statistical analysis was carried out using standard statistical software. Quantitative data were expressed as mean values with standard error (M ± m). Differences between groups were analyzed using Student’s t-test, while relationships between clinical and biochemical parameters were assessed using Pearson’s correlation coefficient (r). A p-value of less than 0.05 was considered statistically significant.The study was conducted in accordance with ethical standards for medical research involving human subjects. Written informed consent was obtained from the parents or legal guardians of all participants, and confidentiality as well as the safety of the children were strictly maintained throughout the study.
3. Results
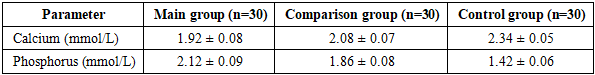
- The results of the clinical and laboratory investigation demonstrated that children with chronic kidney disease (CKD) undergoing hemodialysis exhibited significantly impaired oral health status compared to healthy controls. Moreover, statistically significant differences between the main and comparison groups confirmed the effectiveness of the comprehensive, pathogenetically oriented treatment approach.Biochemical analysis revealed pronounced disturbances in mineral metabolism among children undergoing hemodialysis. The main group showed significantly lower serum calcium levels and higher phosphorus levels compared to the control group (p < 0.05), while the comparison group demonstrated intermediate values, reflecting partial metabolic imbalance.
|
|
4. Discussion
- The findings of the present study demonstrate that children with chronic kidney disease (CKD) undergoing hemodialysis experience significant deterioration in oral health status, particularly in relation to dental hard tissues. These changes are not isolated clinical manifestations but reflect complex systemic disturbances associated with impaired renal function. The results confirm that alterations in mineral metabolism, salivary function, and immune response collectively contribute to the pathogenesis of dental diseases in this patient population.One of the most important findings of the study is the strong association between disturbances in calcium-phosphorus metabolism and the severity of dental pathology. The observed hypocalcemia and hyperphosphatemia in children undergoing hemodialysis are consistent with the well-established pathophysiology of CKD, where reduced renal excretion of phosphorus and impaired activation of vitamin D lead to secondary hyperparathyroidism and mineral imbalance. These metabolic alterations directly affect enamel and dentin mineralization, resulting in structural defects such as enamel hypoplasia and reduced resistance to caries.The negative correlation between serum calcium levels and DMF index (r = -0.62) highlights the crucial role of calcium in maintaining enamel integrity. Calcium deficiency compromises the crystalline structure of enamel, increasing its susceptibility to acid demineralization. Conversely, the positive correlation between phosphorus levels and DMF index (r = +0.58) suggests that elevated phosphorus levels further disrupt mineral homeostasis and exacerbate dental tissue damage. These findings support the concept that dental hard tissue pathology in CKD patients has a strong metabolic basis rather than being solely influenced by local factors.In addition to mineral metabolism disturbances, salivary alterations likely play a significant role in the observed clinical outcomes. Reduced salivary flow rate, changes in pH, and decreased buffering capacity, which are commonly reported in children undergoing hemodialysis, may contribute to impaired oral homeostasis. Saliva is essential for mechanical cleansing, remineralization, and antimicrobial defense; therefore, its dysfunction creates favorable conditions for plaque accumulation and bacterial proliferation. This may explain the significantly higher OHI-S and Plaque Index values observed in the study groups.Furthermore, immunological dysfunction associated with CKD may exacerbate oral pathology. Impaired neutrophil activity, altered cytokine production, and reduced local immune defense mechanisms increase susceptibility to inflammation and infection. This is reflected in the elevated PMA index values, indicating a higher prevalence of gingival inflammation in children undergoing hemodialysis. The interplay between microbial factors and host immune response likely accelerates the progression of both carious and non-carious lesions.Another important aspect of this study is the evaluation of treatment effectiveness. The results demonstrate that children receiving a comprehensive, pathogenetically oriented treatment approach showed better clinical outcomes compared to those receiving standard dental care. This finding emphasizes the importance of addressing not only local oral conditions but also systemic and biochemical factors when managing dental health in CKD patients. Conventional treatment strategies that focus solely on symptomatic management may be insufficient in this population, as they do not target the underlying mechanisms contributing to disease progression.The higher prevalence of enamel hypoplasia and hypersensitivity in the main and comparison groups further supports the hypothesis that systemic disturbances during tooth development have long-term consequences. In children, dental tissues are still undergoing maturation, making them particularly vulnerable to metabolic imbalances. As a result, early-life disturbances may lead to irreversible structural defects that predispose individuals to future dental complications.Overall, the findings of this study are consistent with existing literature indicating that oral health in children with CKD is significantly compromised and closely linked to systemic factors. However, the present study adds to current knowledge by demonstrating the clinical benefits of a comprehensive treatment approach and by providing quantitative evidence of the relationship between biochemical and dental parameters.These results highlight the need for an integrated, multidisciplinary approach to the management of children undergoing hemodialysis. Collaboration between nephrologists and dental professionals is essential to ensure early diagnosis, preventive care, and targeted therapeutic interventions. Incorporating biochemical monitoring into routine dental assessment may improve the prediction and prevention of oral diseases in this high-risk group.
5. Conclusions
- The study demonstrated that children with chronic kidney disease undergoing hemodialysis have significantly impaired oral health status compared to healthy controls, characterized by higher caries intensity, poor oral hygiene, and increased prevalence of inflammatory and non-carious lesions. Disturbances in calcium-phosphorus metabolism, particularly hypocalcemia and hyperphosphatemia, were found to be significantly associated with deterioration of dental hard tissues. The identified correlations (r = -0.62 for calcium and DMF; r = +0.58 for phosphorus and DMF) confirm the pathogenetic role of mineral imbalance in the development of dental pathology. Structural defects such as enamel hypoplasia and dentin hypersensitivity were more prevalent in children undergoing hemodialysis, indicating that systemic metabolic disturbances during tooth development have long-term consequences on dental tissue integrity. The comprehensive, pathogenetically oriented treatment approach demonstrated higher clinical effectiveness compared to standard dental care, leading to improved oral hygiene and reduced inflammatory indices. These findings highlight the necessity of integrating systemic and biochemical factors into dental management strategies for children with CKD, emphasizing the importance of multidisciplinary care and early preventive interventions.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML