-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(4): 2052-2057
doi:10.5923/j.ajmms.20261604.100
Received: Apr. 6, 2026; Accepted: Apr. 23, 2026; Published: Apr. 25, 2026

Personalized Diagnostic and Treatment Algorithm for X-Ray Endovascular Interventions in Patients with Diabetic Foot Syndrome and Concomitant Ischemic Heart Disease: Comparative Evaluation of Clinical Efficacy
Kamalov S. T.
PhD Candidate, Department of Vascular Surgery, Republican Specialized Center of Surgery named after Academician V.V. Vakhidov, Tashkent, Uzbekistan
Correspondence to: Kamalov S. T., PhD Candidate, Department of Vascular Surgery, Republican Specialized Center of Surgery named after Academician V.V. Vakhidov, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

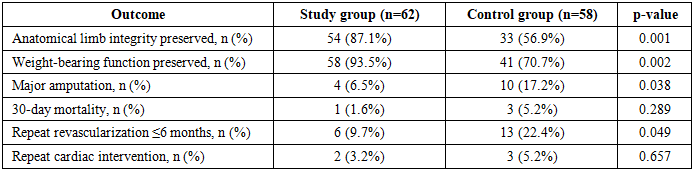
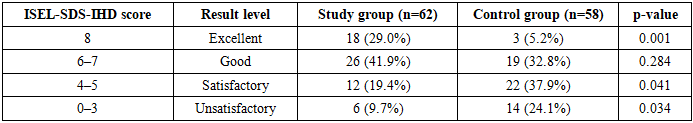
(1) Background: The coexistence of diabetic foot syndrome (DFS) and ischemic heart disease (IHD) substantially increases the risk of major amputation and mortality when endovascular interventions are planned without systematic cardiac risk stratification. A personalized treatment algorithm integrating cardiac functional assessment into the revascularization decision framework may improve outcomes in this high-risk population. (2) Methods: A prospective comparative study included 120 patients with DFS and IHD treated at the Republican Specialized Center of Surgery named after V.V. Vakhidov between 2015 and 2024. The control group (n=58, 2015–2019) received standard X-ray endovascular interventions (XEI) without preliminary cardiac risk stratification. The study group (n=62, 2020–2024) was managed according to a personalized algorithm incorporating risk-stratified sequencing of revascularization based on a validated logistic regression model. Outcomes were evaluated using the ISEL-SDS-IHD scale. (3) Results: Limb anatomical integrity was preserved in 87.1% vs. 56.9% (p=0.001); weight-bearing function in 93.5% vs. 70.7% (p=0.002); major amputation rate was 6.5% vs. 17.2% (p=0.038); repeat revascularization rate was 9.7% vs. 22.4% (p=0.049). Hospital stay was reduced by 37% (11.4 vs. 18.1 bed-days, p=0.001). Excellent ISEL-SDS-IHD outcomes (score 8) were achieved in 29.0% vs. 5.2% (p=0.001). (4) Conclusions: The personalized algorithm significantly reduces amputation rates, repeat interventions, and hospitalization duration while improving cardiac and limb functional outcomes in patients with DFS and IHD.
Keywords: Diabetic foot syndrome, Ischemic heart disease, Personalized algorithm, Endovascular intervention, Risk stratification, Limb salvage, Heart-first strategy, Limb-first strategy, ISEL-SDS-IHD
Cite this paper: Kamalov S. T., Personalized Diagnostic and Treatment Algorithm for X-Ray Endovascular Interventions in Patients with Diabetic Foot Syndrome and Concomitant Ischemic Heart Disease: Comparative Evaluation of Clinical Efficacy, American Journal of Medicine and Medical Sciences, Vol. 16 No. 4, 2026, pp. 2052-2057. doi: 10.5923/j.ajmms.20261604.100.
1. Introduction
- The combination of diabetic foot syndrome (DFS) and ischemic heart disease (IHD) constitutes one of the most clinically demanding comorbid configurations in vascular surgery. More than 70% of patients presenting with DFS have concurrent IHD - frequently subclinical or inadequately characterized - while verified systolic dysfunction (LVEF <50%) is present in one-third, and prior myocardial infarction in one-fifth of this population [1,2]. This systemic vascular burden is not merely additive: IHD directly impairs myocardial output and peripheral tissue reperfusion, thereby limiting the functional efficacy of technically successful lower-extremity revascularization, even when angiographic flow restoration is achieved [3].Under the traditional treatment paradigm, X-ray endovascular interventions (XEI) on lower-limb arteries are planned primarily on the basis of angiographic anatomy and limb ischemia severity, without systematic assessment of coronary functional reserve or myocardial status [4]. This fragmented approach leaves a critical therapeutic gap: in patients with decompensated IHD, even technically successful revascularization may fail to restore adequate tissue perfusion, resulting in progressive necrosis, wound infection, and ultimately major amputation. Prior analysis of the control cohort at our center demonstrated that this scenario materialized in 37.9% of cases managed under the standard protocol, with major amputation rates reaching 45.4% and 30-day mortality 13.6% among those with unfavorable outcomes [5].Contemporary vascular guidelines from both the International Working Group on the Diabetic Foot (IWGDF) and the Global Vascular Guidelines (GVG) on chronic limb-threatening ischemia emphasize the necessity of multidisciplinary assessment and cardiac functional evaluation before revascularization in high-risk diabetic patients [6,7]. However, practical implementation of risk-stratified, sequenced intervention algorithms - defining which organ system (heart or limb) should be prioritized and in what order - remains inconsistently adopted, particularly in resource-limited settings where multidisciplinary coordination is suboptimal [8].The debate between "limb-first" and "heart-first" intervention strategies has been growing in the vascular literature without definitive resolution by prospective randomized evidence [9]. The choice of strategy depends on a range of clinical parameters - including LVEF, angina functional class, ulcer staging, and angioarchitectural complexity - none of which individually captures the composite risk. A model-driven, individualized algorithm that objectively quantifies risk and allocates patients to risk-stratified therapeutic pathways represents a logical advancement in the clinical management of this population [10].The present study was undertaken to develop and prospectively evaluate the clinical efficacy of a personalized diagnostic and treatment algorithm for XEI in patients with DFS and IHD, comparing outcomes against the historical control cohort managed under the traditional approach at the same institution.
2. Materials and Methods
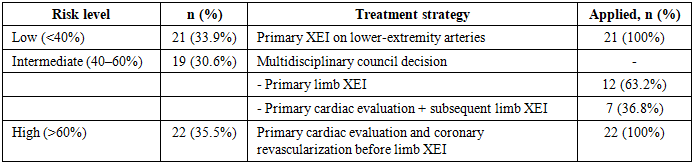
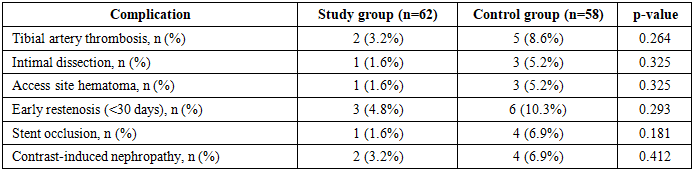
- Study design and patient population. This prospective comparative study was conducted at the Republican Specialized Center of Surgery named after Academician V.V. Vakhidov (Tashkent, Republic of Uzbekistan) over a ten-year period from 2015 to 2024. A total of 120 patients with DFS complicated by lower-limb ischemia and documented IHD were included. Patients were distributed into two groups by chronological and tactical principle.The control group (n=58) comprised patients treated between 2015 and 2019 under the standard institutional protocol, in which XEI on lower-extremity arteries were performed without preliminary cardiac risk stratification. IHD was not incorporated as a determinant of intervention sequencing or extent.The study group (n=62) comprised patients treated between 2020 and 2024 using a newly developed personalized treatment algorithm, which integrated preoperative echocardiographic evaluation, stress testing where feasible, coronary angiography, and - when indicated - coronary stenting prior to peripheral XEI.Inclusion criteria: confirmed type 2 diabetes mellitus; clinically and instrumentally verified lower-limb ischemia (Fontaine stage IIb–IV); ulcerative or necrotic foot lesions (Wagner stage I–V); documented IHD; absence of active sepsis or terminal organ failure. Patients with decompensated NYHA class IV heart failure, acute coronary syndrome within 30 days prior to enrollment, terminal renal failure requiring dialysis, or actively progressing oncological disease were excluded. Both groups were comparable in sex distribution (male 53.4% vs. 54.8%), mean age (68.3±9.5 vs. 66.4±8.9 years), diabetes duration (10.3±5.4 vs. 10.5±5.2 years), mean HbA1c (9.0±1.5%), and proportions of advanced Fontaine and Wagner stages.The personalized algorithm. The algorithm was grounded in a validated logistic regression–based prognostic model developed from the control cohort, identifying six independent predictors of major amputation or 30-day mortality: LVEF <50% (OR=8.26), angina CCS class III–IV (OR=7.24), Wagner stage IV–V (OR=6.47), heel or total foot involvement (OR=5.59), HbA1c ≥10% (OR=4.90), and requirement for contralateral crossover access (OR=4.34), with AUC=0.915 [5].Based on the calculated individual probability of unfavorable outcome (P), patients in the study group were stratified into three risk strata with corresponding therapeutic strategies:• Low risk (P<0.40; n=21, 33.9%): Primary XEI on lower-extremity arteries without mandatory coronary pre-assessment. Patients had preserved LVEF, compensated DM, and localized foot lesions.• Intermediate risk (P=0.40–0.60; n=19, 30.6%): Decision delegated to a multidisciplinary council comprising vascular surgeon, cardiologist, and functional diagnostician. Twelve patients (63.2%) underwent primary limb XEI; seven (36.8%) underwent preliminary cardiac evaluation with subsequent limb revascularization.• High risk (P>0.60; n=22, 35.5%): Priority cardiac evaluation with coronary angiography and coronary stenting where indicated, followed by XEI on lower-extremity arteries. Seventeen patients underwent sequential staged revascularization; five underwent simultaneous procedures.Outcome assessment. Primary outcome measures were: preservation of anatomical limb integrity (no major amputation above ankle level); preservation of weight-bearing function; major amputation rate; 30-day all-cause mortality; and repeat revascularization within 6 months. Secondary outcomes included specific procedural complications, length of hospitalization, number of repeat hospitalizations and necrotomies within 6 months, and resource utilization.Integrated outcome quality was assessed using the ISEL-SDS-IHD scale (Integrated System for Evaluation of Limb-saving in patients with Diabetic foot Syndrome and Ischemic Heart Disease), an eight-domain instrument developed as part of the study. Each domain is scored 0–1: (1) no major amputation; (2) preserved weight-bearing function; (3) NYHA regression ≥1 class or LVEF increase ≥5%; (4) Wagner stage regression or wound healing; (5) HbA1c reduction ≥1% at 3 months; (6) no repeat revascularization within 6 months; (7) no specific procedural complications; (8) patient-reported treatment satisfaction. Total score: 0–8; interpretation: 0–3 unsatisfactory, 4–5 satisfactory, 6–7 good, 8 excellent.Statistical analysis. IBM SPSS Statistics v.26.0 (USA) and Microsoft Excel 2019 were used. Continuous variables are presented as M±SD; between-group comparisons used Student's t-test or Mann-Whitney U test. Categorical variables were compared by χ² or Fisher's exact test. Significance was set at p<0.05. Intragroup dynamics were assessed by paired t-test for continuous variables.
3. Results and Discussion
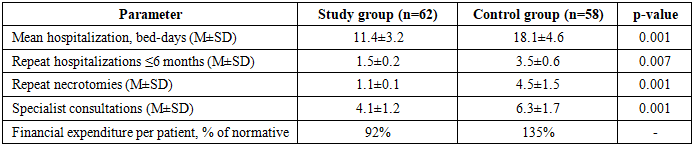
- Risk distribution in the study group. Of 62 patients in the study group, 33.9% (n=21) were classified as low risk, 30.6% (n=19) as intermediate, and 35.5% (n=22) as high risk according to the prognostic model (Table 1). Among high-risk patients, all 22 underwent primary cardiac evaluation; coronary stenting was performed in 17 (77.3%) before peripheral XEI. This distribution confirms that over one-third of patients presenting with DFS and IHD carry high cardiac risk that warrants priority coronary intervention before limb revascularization - a proportion that would have been missed without systematic pre-procedural stratification.
|
|
|
|
|
|
4. Conclusions
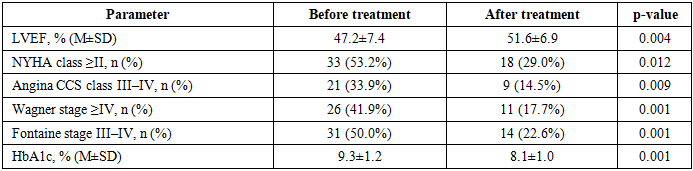
- The personalized diagnostic and treatment algorithm for X-ray endovascular interventions, based on individual risk stratification using a validated logistic regression model, demonstrated statistically significant superiority over the traditional non-stratified approach across all primary clinical outcome measures in patients with DFS and IHD.Key findings are: anatomical limb integrity was preserved in 87.1% vs. 56.9% (p=0.001); weight-bearing function in 93.5% vs. 70.7% (p=0.002); major amputation was reduced 2.6-fold (6.5% vs. 17.2%, p=0.038); repeat revascularization was reduced 2.3-fold (9.7% vs. 22.4%, p=0.049). Mean LVEF improved by 4.4 percentage points (p=0.004), NYHA class ≥II prevalence fell by 24.2 points (p=0.012), and HbA1c decreased by 1.2% (p=0.001) within the study group. Hospitalization was shortened by 37% (p=0.001), and per-patient financial expenditure was 46.7% lower than in the control group. Excellent ISEL-SDS-IHD outcomes were achieved 5.6 times more frequently (p=0.001).The three-tier risk stratification - directing low-risk patients to primary limb XEI, intermediate-risk patients to multidisciplinary evaluation, and high-risk patients to priority coronary revascularization - provides a practical, objective framework that resolves the "limb-first vs. heart-first" dilemma on an evidence-based individual basis. The ISEL-SDS-IHD scale offers a validated composite instrument for integrated outcome monitoring in this comorbid population.Prospective multicenter validation is warranted to confirm generalizability. Pending such evidence, the algorithm may be recommended for implementation in vascular surgery centers managing patients with combined DFS and IHD, particularly where multidisciplinary cardiac-vascular coordination is available.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML