-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(3): 1173-1176
doi:10.5923/j.ajmms.20261603.67
Received: Mar. 2, 2026; Accepted: Mar. 15, 2026; Published: Mar. 17, 2026

Clinical Characteristics and Treatment Strategies for Encrusted Cystitis
Dadakhanov N. E.
Republican Specialized Scientific and Practical Medical Center of Urology, Uzbekistan
Correspondence to: Dadakhanov N. E., Republican Specialized Scientific and Practical Medical Center of Urology, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background. Encrusted cystitis (EC) is a rare but severe inflammatory disease of the urinary bladder characterized by deposition of crystalline salts on the mucosal surface. The condition is often associated with urease-producing microorganisms and an alkaline urinary environment, which promotes persistent infection and formation of encrustations. Objective. To evaluate the clinical effectiveness of different treatment strategies for encrusted cystitis and determine optimal management approaches depending on the extent of bladder mucosal involvement. Methods. A retrospective study was conducted at the Republican Specialized Scientific and Practical Medical Center of Urology between 2010 and 2024. Fifty-nine patients aged 34–78 years with confirmed encrusted cystitis were included. Diagnostic evaluation consisted of laboratory testing, urine culture, determination of urine pH, ultrasound examination, cystoscopy and computed tomography when indicated. Treatment strategies included transurethral resection (TUR) of encrusted lesions, antibacterial therapy based on culture sensitivity, urine acidification, bladder instillation, and urinary diversion procedures in selected cases. Results. Complete recovery after initial treatment was observed in 36 patients. Recurrence occurred in 23 patients, of whom 18 required percutaneous cystostomy combined with basic therapy, which resulted in stable remission. In 5 patients with extensive disease, bilateral nephrostomy and repeated surgical interventions were required. Treatment outcomes were closely associated with the extent of mucosal involvement. Conclusion. Effective management of encrusted cystitis requires a combined approach including surgical removal of encrustations, antimicrobial therapy, correction of urinary pH and bladder instillation. In cases of extensive disease or recurrence, urinary diversion techniques may significantly improve treatment outcomes.
Keywords: Encrusted cystitis, Urinary tract infection, Transurethral resection, Urinary diversion, Biofilm
Cite this paper: Dadakhanov N. E., Clinical Characteristics and Treatment Strategies for Encrusted Cystitis, American Journal of Medicine and Medical Sciences, Vol. 16 No. 3, 2026, pp. 1173-1176. doi: 10.5923/j.ajmms.20261603.67.
1. Introduction
- Encrusted cystitis (EC) is considered one of the rare and most severe forms of chronic inflammation of the urinary bladder. It is characterized not only by a persistent inflammatory process but also by the formation of salt deposits on the bladder mucosa, which leads to the development of dense plaques and biofilms [2,4]. The disease was first described in the mid-twentieth century; however, it remains insufficiently studied to this day due to its relatively low prevalence and the complexity of correct diagnosis and recognition [6,7].The pathogenesis of EC is associated with the activity of urease-producing microorganisms that hydrolyze urea and shift the acidity of urine toward the alkaline side. Increased pH promotes precipitation of calcium, magnesium and ammonium phosphate crystals, the formation of struvites and the maintenance of a chronic inflammatory process [9,11]. At the same time, microbial biofilms create resistance to systemic antibacterial therapy, which makes treatment particularly difficult.The main risk factors include bladder catheterization, sometimes even a single episode, interventions on the lower urinary tract, the presence of diabetes mellitus, immunodeficiency conditions and chronic urinary tract infections [1,5]. In recent years, the detection rate of EC has increased, which can be explained not only by a real increase in the number of patients but also by the development of diagnostic capabilities, including endoscopy, computed tomography and molecular research methods [10,12].Despite its rarity, encrusted cystitis represents a serious clinical problem, since it often leads to severe pain symptoms, decreased quality of life, the need for repeated surgical interventions and prolonged hospitalization [8].Aim of the Study. To assess the clinical effectiveness of various treatment approaches for encrusted cystitis and to develop practical recommendations for choosing patient management tactics depending on the extent of bladder involvement.
2. Materials and Methods
- The study was conducted at the Republican Specialized Scientific and Practical Medical Center of Urology from 2010 to 2024. A total of 59 patients aged 34 to 78 years with a confirmed diagnosis of encrusted cystitis were observed.The inclusion criteria were the presence of endoscopic signs of EC, morphological confirmation of encrustation, and a persistent course of the disease despite standard antibacterial therapy. Patients complained of dysuria of varying severity, pain in the suprapubic region, episodes of macroscopic hematuria, and the absence of a stable effect from antibacterial therapy.
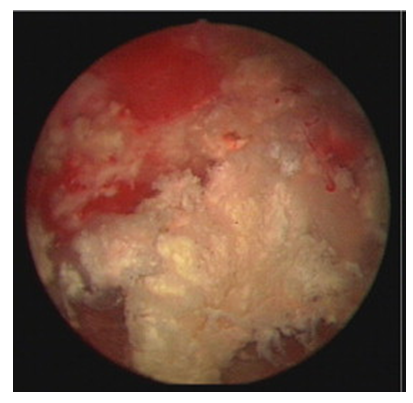 | Figure 1. Endoscopic view of encrustations on the urinary bladder mucosa |
3. Results
- All patients underwent TUR of the encrusted areas of the bladder mucosa and received the so-called basic therapy, which included a course of antibiotic therapy depending on urine culture results, drugs acidifying the urine and bladder instillation.In 36 patients, complete recovery without recurrence was recorded after the first surgical intervention. Recurrences were observed in 23 patients. In 18 of them, percutaneous cystostomy was performed to correct the condition, followed by a course of basic therapy, which made it possible to achieve stable remission. In 5 patients, symptoms persisted despite TUR and local therapy. These patients underwent bilateral nephrostomies, repeated resections and prolonged treatment courses. As a result, clinical improvement and reduction in the extent of lesions were achieved in these patients, with eventual recovery. The average drainage period was 37 days.Histological analysis showed inflammatory-infiltrative changes, cicatricial-sclerotic transformation of the mucosa and pronounced crystallization of salts.The obtained results indicate that surgical removal of the affected mucosal areas alone without additional therapy does not ensure complete recovery. The reason is the persistence of an unfavorable microbial background, a non-healing wound surface and an a006Ckaline bladder environment, which support chronic inflammation.The effectiveness of treatment directly depended on the area of the lesion:• in limited processes up to 10 cm², transurethral resection and basic therapy were sufficient• in lesions 10–40 cm², combined treatment (resection + cystostomy + basic therapy) was required• in extensive processes >40 cm², the optimal solution was a multistage approach including repeated interventions and prolonged courses of basic therapyInternational experience also confirms the need for a combined approach. According to European studies in recent years, the use of antibiotics alone or resection alone leads to recurrences in more than half of cases, whereas the combination of surgical sanitation and local therapy provides remission in 70–80% of patients [13,14].
4. Discussion
- The obtained results demonstrate that the effectiveness of treatment for encrusted cystitis largely depends on the extent of bladder mucosal involvement. In patients with limited lesions, transurethral resection combined with basic therapy was sufficient to achieve stable remission. However, in cases with more extensive involvement, additional urinary diversion procedures such as cystostomy or nephrostomy were required to ensure adequate drainage and create conditions for mucosal healing.These findings are consistent with previously published studies, which emphasize that surgical removal of encrustations alone rarely provides long-term recovery. According to international reports, the combination of surgical resection of the encrusted area with antimicrobial therapy and urine acidification significantly improves treatment outcomes and reduces recurrence rates.In cases of limited mucosal encrustation accompanied by frequent recurrences, cystostomy may be recommended to provide functional rest for the bladder. Urinary diversion reduces the contact of infected alkaline urine with the damaged mucosa and creates more favorable conditions for tissue recovery.In the most severe cases, when the area of encrustation exceeds 40 cm² and the disease demonstrates persistent recurrence, bilateral percutaneous nephrostomy may represent the most effective therapeutic option. Complete diversion of urine directly from the kidneys prevents its contact with the affected bladder mucosa, providing the organ with functional rest and allowing gradual regeneration of the bladder tissues over several months.An important factor influencing recurrence is the persistence of an alkaline urinary environment and bacterial biofilms. These conditions promote continued salt crystallization and support chronic inflammation of the bladder mucosa. Therefore, correction of urinary pH and targeted antimicrobial therapy remain essential components of treatment.The limitations of the present study include its retrospective design and the relatively limited number of patients due to the rarity of the disease. However, the study covers a long observation period and provides valuable clinical data on treatment strategies for encrusted cystitis.
5. Conclusions
- Encrusted cystitis remains one of the most difficult forms of chronic cystitis to treat. The obtained data confirm the need for a combined approach, which often depends on the extent of bladder mucosal encrustation and the frequency of recurrences.Thus, only comprehensive management including surgical, pharmacological and metabolic control with urinary diversion in cases of frequent recurrences allows achieving complete recovery in patients.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML