-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(3): 1085-1090
doi:10.5923/j.ajmms.20261603.50
Received: Feb. 3, 2026; Accepted: Feb. 25, 2026; Published: Mar. 7, 2026

Analysis of Microrna Expression Levels in Patients with Submucous Uterine Myoma
Akhmedova Nilufar Makhmudjanovna1, Mukhitdinova Tukhtakhon Kadyrovna2
1PhD, Associate Professor, Department of Obstetrics and Gynecology, Andijan State Medical Institute (ASMI), Andijan, Uzbekistan
2DSc, Professor, Department of Obstetrics and Gynecology, Andijan State Medical Institute (ASMI), Andijan, Uzbekistan
Correspondence to: Akhmedova Nilufar Makhmudjanovna, PhD, Associate Professor, Department of Obstetrics and Gynecology, Andijan State Medical Institute (ASMI), Andijan, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Submucous uterine myoma represents a significant clinical problem in gynecology due to its high prevalence and impact on reproductive health. Background: Submucous uterine myoma is a common benign tumor in women of reproductive age. Epigenetic mechanisms, particularly microRNAs regulating proliferation, apoptosis, and angiogenesis, may contribute to fibroid development and serve as potential biomarkers. Methods: The study included 200 women with submucous uterine myoma and 30 healthy controls. Expression levels of microRNA-29, miR-5096, miR-5585, miR-21, and miR-221 were analyzed and compared between groups. Associations between microRNA expression and clinical characteristics, hormonal status, lipid metabolism, and hemostatic parameters were evaluated. Results: Patients with submucous uterine myoma demonstrated significantly reduced expression of the studied microRNAs compared with controls. More pronounced decreases were observed in women with larger myomatous nodes. MicroRNA levels were associated with endocrine indicators, lipid profile parameters, and coagulation markers. Conclusion: Dysregulation of the studied microRNAs appears to play a pathogenetic role and may support their use as biomarkers for early diagnosis and personalized management.
Keywords: MicroRNA, Submucosal uterine myoma, Expression, Biomarkers
Cite this paper: Akhmedova Nilufar Makhmudjanovna, Mukhitdinova Tukhtakhon Kadyrovna, Analysis of Microrna Expression Levels in Patients with Submucous Uterine Myoma, American Journal of Medicine and Medical Sciences, Vol. 16 No. 3, 2026, pp. 1085-1090. doi: 10.5923/j.ajmms.20261603.50.
Article Outline
1. Introduction
- In recent decades, significant advances in molecular biology and bioinformatics have enabled the development of highly accurate and efficient methods for quantitative analysis of microRNAs (miRNAs) in various biological tissues [1,4]. Among these, extracellular circulating microRNAs, particularly those encapsulated in exosomes, have attracted considerable attention due to their high stability, tissue-specific expression patterns, and regulatory roles in gene expression, making them promising candidates as biomarkers for a wide range of pathological conditions [2,3,6]. Uterine fibroids, particularly submucosal types, are benign smooth muscle tumors that can lead to abnormal uterine bleeding, infertility, and other complications. Although the exact mechanisms of fibroid development are not fully understood, recent evidence suggests that dysregulation of specific miRNAs may influence key pathological processes, including abnormal cell proliferation, extracellular matrix deposition, angiogenesis, and apoptosis [5]. Exosomes, vesicular structures containing miRNAs, can cross intercellular barriers and mediate intercellular communication, which may play a crucial role in the initiation and progression of submucosal uterine fibroids [3,4]. Several studies have reported distinct miRNA expression profiles in patients with uterine fibroids, indicating their potential as non-invasive diagnostic biomarkers and as molecular targets for therapeutic intervention. However, there remains a lack of comprehensive analysis specifically focusing on submucosal fibroids and the relationship between circulating miRNA levels and disease characteristics. Therefore, the aim of this study is to analyze the expression levels of specific circulating microRNAs in patients with submucosal uterine fibroids and to investigate their potential roles in the pathogenesis and clinical presentation of the disease. This research may provide novel insights into the molecular mechanisms underlying fibroid development and contribute to improved non-invasive diagnostic strategies.Purpose of the study. Study of the expression level of submucous uterine fibroids in women of reproductive age.
2. Material and Methods of Research
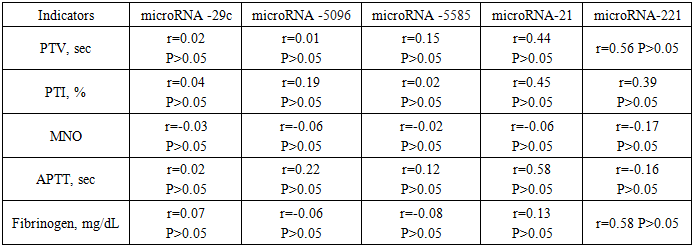
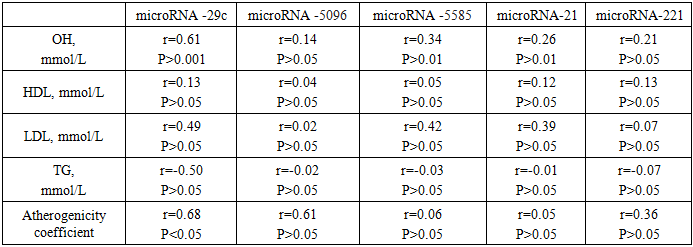
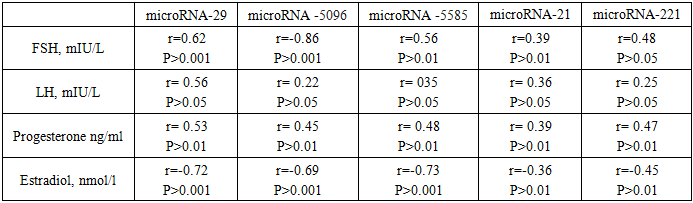
- Study design and participants. This prospective clinical-laboratory study was conducted at the Gynecology Department, Andijan Regional Perinatal Center, Andijan, Uzbekistan, from 2019 to 2024. A total of 200 women diagnosed with submucosal uterine fibroids were included. Patients were divided into two groups according to the size, location, and number of myomatous nodes:• Group 1 (n = 160, 69.6%) – patients with nodes ≤ 5 cm, treated by resectoscopy.• Group 2 (n = 40, 17.4%) – patients with nodes > 5 cm, treated by laparoscopic myomectomy.A control group of 30 apparently healthy women was included to evaluate laboratory parameters. Inclusion criteria: Women of reproductive age diagnosed with submucosal uterine fibroids confirmed by imaging and planned for surgical treatment. Exclusion criteria: Women with other uterine pathologies (e.g., adenomyosis), malignant tumors, pregnancy, acute inflammatory diseases, or chronic systemic illnesses (e.g., diabetes, hypertension). Sample size calculation: The sample included all eligible patients admitted to the gynecology department during the study period (convenience sampling).Data collection instruments. microRNA expression analysis was performed using quantitative RT-PCR. The following targets were analyzed: miR-29, miR-5096, miR-5585, miR-21, and miR-221. • PCR kits / reagents:• RNA extraction: miRNeasy Mini Kit (Qiagen, Germany)• Lysis reagent: TRIzol® LS Reagent (Invitrogen, USA)• Reverse transcription: TaqMan® microRNA Reverse Transcription Kit (Applied Biosystems, USA) and miScript Reverse Transcription Kit (Qiagen, Germany)• qPCR amplification: miScript SYBR Green PCR Kit (Qiagen, Germany)• miRNA panels: Custom primers for the specific microRNAs analyzed in this study.• Quantification method: Relative quantification using the 2^(-ΔCt) method.• Normalization: Expression levels were normalized against U6 small nuclear RNA as an internal control.Procedures1. Sample collection: 5 mL of venous blood was collected into Vacutainer® tubes. Samples were centrifuged at 5000 g for 10 minutes using a Sigma 4-16KS centrifuge (Sigma Laborzentrifugen, Germany).2. RNA extraction: 100 µL of plasma was mixed with 750 µL TRIzol® LS, incubated for 5 min at room temperature, and 200 µL chloroform was added. The mixture was vortexed for 15 seconds (Microspin FV-2400, BioSan, Latvia) and incubated for 2–3 minutes. Centrifugation was performed at 14000 g for 15 min at 4°C. RNA was purified using miRNeasy Mini Kit buffers (RWT, RPE) according to the manufacturer’s protocol.3. RNA quantification and storage: RNA concentration was measured using NanoDrop 1000 NP spectrophotometer (Thermo Scientific, USA). Yield ranged from 30 to 800 ng/µL (A260/280 = 1.99). Samples were stored at –40 °C until further use.4. Reverse transcription: Reverse transcription was performed using TaqMan® microRNA Reverse Transcription Kit and miScript Reverse Transcription Kit on a Veriti 4-channel amplifier (Applied Biosystems, USA). The thermal profile was: 30 min at 16°C, 30 min at 42°C, 5 min at 85°C, then hold at 4°C.5. qPCR amplification: Quantitative PCR was performed using Rotor Gene 6000 and Rotor Gene Q instruments (Qiagen, Germany) with miScript SYBR Green PCR Kit, following manufacturer instructions. Relative expression was calculated as 2^(-ΔCt) × 100, normalized to U6 snRNA, and expressed in arbitrary units.Ethical considerations. The study was approved by the Ethics Committee of Andijan State Medical Institute, Andijan, Uzbekistan, approval number ASMI-2026-01. All participants provided written informed consent before inclusion in the study. Participation was voluntary, and confidentiality of all patient data was strictly maintained. All procedures were conducted in accordance with the Declaration of Helsinki and local regulations for research involving human participants.Data Analysis• Statistical analysis was performed using SPSS v.25.• Group comparisons: Differences in miRNA expression between the control group and patient groups were analyzed using t-test or Mann–Whitney U test, depending on data distribution.• Correlation analyses: Pearson’s correlation coefficient (r) was used to evaluate relationships between microRNA expression levels and clinical, hormonal, lipid, and coagulation parameters (PT, PTI, MNO, APTT, Fibrinogen, FSH, LH, Progesterone, Estradiol, HDL, LDL, TG).• Significance: A p-value < 0.05 was considered statistically significant.• Tests were selected based on the type of variable and distribution of the data to ensure statistical validity.
3. Results
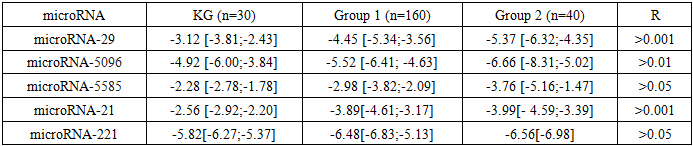
- A study of microRNA expression levels revealed its direction and the pathogenetic significance of submucosal uterine fibroids. Specifically, patients with submucosal uterine fibroids in the first group showed a significant decrease in miR-29 expression by 42.6% relative to the control group, which is confirmed by published data. In the second group of patients, this decrease reached 72.1%, indicating a more pronounced dysregulation of this microRNA in the pathogenesis of the disease. Expression of miR-5096 also showed a downward trend in patients with submucosal uterine fibroids, albeit to a lesser extent. In the first group, the decrease was 12.2%, and in the second, 13.4% (P> 0.01), indicating the maintenance of molecular mechanisms that contribute to the development of uterine fibroids. Notably, the transfer of miR-5096 from glioma cells to astrocytes enhanced their invasive properties, which may indicate a potential role for this microRNA in the processes of metastasis and tumor progression. Reducing miR-5585 expression to 3.99 relative units significantly enhanced the proliferation, migration, and invasive properties of microvascular endothelial cells in submucosal uterine fibroids. This effect was achieved by suppressing cell apoptosis and reducing the production of reactive oxygen species, indicating a key role for this microRNA in regulating angiogenesis and neovascularization of tumor tissue. (Table 1).
|
|
|
|
4. Discussion
- This study shows that patients with submucosal uterine myoma have significantly reduced expression of circulating miR-29, miR-5096, miR-5585, miR-21, and miR-221, with the most pronounced decreases in women with larger fibroids. The findings suggest that dysregulated miRNAs contribute to fibroid pathogenesis and may serve as non-invasive biomarkers of disease severity. Reduced miR-29 and miR-21 levels are consistent with previous studies indicating their roles in extracellular matrix remodeling, cell proliferation, and fibrosis in uterine leiomyomas [1,2,8]. Decreased miR-29 may enhance collagen deposition, while lower miR-21 promotes myoma cell survival [6]. miR-5096 and miR-5585 also decreased, suggesting involvement in endothelial proliferation and angiogenesis, processes critical for fibroid vascularization [7]. miR-221 reductions correlated with coagulation parameters, implying systemic effects beyond the uterus [5]. Hormonal influence is evident: miRNA expression correlated with FSH, LH, progesterone, and estradiol levels. miR-29 showed a strong inverse relationship with estradiol (r=–0.72, P <0.001), indicating estrogen-driven suppression. Progesterone-dominated phases were associated with enhanced myoma cell proliferation and mitotic activity, reflecting the known sensitivity of fibroids to hormonal signals. These results support the role of miRNAs as mediators of hormone-dependent fibroid growth. Comparisons with other research show similar patterns of miR-29 and miR-21 dysregulation in leiomyomas. This study extends these findings to submucosal fibroids and links miRNA changes to lipid and coagulation parameters, suggesting a broader systemic impact. Limitations include the small number of patients with large fibroids, cross-sectional design, and focus on only five miRNAs. Circulating miRNA levels may not fully reflect tissue-specific expression, further studies with paired tissue and plasma samples are needed. The results highlight the potential of miR-29 and miR-21 as biomarkers for fibroid progression and suggest that monitoring these miRNAs could inform personalized management strategies. Associations with coagulation and metabolic markers also point to potential links between reproductive and systemic health, warranting further investigation.
5. Conclusions
- Abnormal microRNA expression is a promising biomarker for the diagnosis and monitoring of submucous uterine myomas. Unlike mRNA, microRNAs are highly stable, expressed at lower levels, and have simpler expression profiles, making them particularly suitable for high-throughput analysis and clinical applications. Dysregulation of specific microRNAs, including miR-29, miR-21, miR-5096, miR-5585, and miR-221, is closely associated with fibroid pathogenesis by influencing key cellular processes such as proliferation, apoptosis, extracellular matrix remodeling, and angiogenesis. Their expression also correlates with hormonal status, lipid metabolism, and coagulation parameters, suggesting systemic effects beyond the uterus. These findings highlight the potential of microRNAs as non-invasive biomarkers for early detection, disease monitoring, and prognosis. Furthermore, understanding microRNA-mediated molecular mechanisms provides new avenues for targeted therapeutic interventions aimed at modulating their expression. Integrating comprehensive microRNA profiling into clinical practice could support personalized management strategies, optimize treatment selection, and ultimately improve reproductive and overall health outcomes in women affected by submucous uterine myomas.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML