-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(3): 1075-1080
doi:10.5923/j.ajmms.20261603.48
Received: Jan. 24, 2026; Accepted: Feb. 20, 2026; Published: Mar. 7, 2026

Blood Pressure Dynamics in Chronic Kidney Disease Patients During Programmed Hemodialysis: Cross-Sectional Observational Study
Kh. A. Abdullaeva, M. A. Sabirov
Republican Scientific and Practical Medical Center of Nephrology and Kidney Transplantation, Tashkent, Uzbekistan
Correspondence to: Kh. A. Abdullaeva, Republican Scientific and Practical Medical Center of Nephrology and Kidney Transplantation, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background: Chronic kidney disease (CKD) represents a major global public health burden, frequently coexisting with cardiovascular disease (CVD) and diabetes mellitus (DM), which collectively elevate the risk of severe morbidity and mortality. Patients receiving programmed hemodialysis (HD) are particularly vulnerable to haemodynamic instability, including both intradialytic hypertension and hypotension, which are associated with adverse cardiovascular outcomes and impaired quality of life. Understanding the patterns of blood pressure (BP) change throughout the HD session is essential for optimising dialysis prescription and cardiovascular risk management in this high-risk population. The present study was aimed to investigate the dynamics of systolic (SBP) and diastolic blood pressure (DBP) in CKD patients before, during, and after a programmed hemodialysis session. Materials and Methods: A cross-sectional observational study was conducted at the Republican Scientific and Practical Medical Center of Nephrology and Kidney Transplantation, Tashkent, Uzbekistan. A total of 60 patients with CKD undergoing programmed hemodialysis were enrolled (Group 1: n=60; comparative Group 2: comorbid CKD patients with diabetes mellitus). Blood pressure was measured using a standardised sphygmomanometer at three time points: before, during, and after the HD session. Descriptive statistics (mean ± SD, median, minimum, maximum) and the Mann–Whitney U-test (between-group comparisons) were applied using IBM SPSS Statistics, version 26.0. Statistical significance was set at P < 0.05. Results: Among the 60 enrolled patients, 31 (51.7%) were male and 29 (48.3%) were female, with a mean age of 60.8 ± 1.55 years and mean haemoglobin of 87.27 ± 2.58 g/L. In Group 1, mean SBP decreased significantly from 143.3 ± 2.84 mmHg before HD to 129.4 ± 3.32 mmHg after HD (P = 0.001). Group 2 demonstrated greater DBP variability (±15.36 mmHg before HD; ±15.38 mmHg during HD), reflecting haemodynamic heterogeneity. Intradialytic hypotension episodes (SBP ≤ 90 mmHg) were observed, indicating the need for individualised ultrafiltration protocols. Conclusion: Programmed hemodialysis produced clinically meaningful reductions in both SBP and DBP, particularly through the mechanism of ultrafiltration-mediated fluid removal. Group 1 patients demonstrated more homogeneous haemodynamic responses, whereas Group 2 exhibited marked diastolic variability consistent with the heterogeneous impact of diabetic comorbidity on vascular regulation. Individualised BP management strategies and close haemodynamic monitoring throughout HD sessions are essential to minimise cardiovascular risk and prevent intradialytic hypotension in this vulnerable population.
Keywords: Chronic kidney disease, Hemodialysis, Blood pressure, Cardiovascular risk, Ultrafiltration
Cite this paper: Kh. A. Abdullaeva, M. A. Sabirov, Blood Pressure Dynamics in Chronic Kidney Disease Patients During Programmed Hemodialysis: Cross-Sectional Observational Study, American Journal of Medicine and Medical Sciences, Vol. 16 No. 3, 2026, pp. 1075-1080. doi: 10.5923/j.ajmms.20261603.48.
Article Outline
1. Introduction
- Cardiovascular diseases (CVD) constitute the predominant cause of mortality from non-communicable diseases worldwide, with the World Health Organization (WHO) attributing a minimum of 19 million fatalities in 2021 to CVD only. [26] Cancer, chronic respiratory diseases, and diabetes mellitus - the latter including kidney disease-related deaths - together represent the other major contributors to non-communicable disease mortality. [26] Kidney diseases of advanced stages are frequently associated with concurrent pathology of the digestive system, endocrine glands, and cardiovascular system, driven by shared immunological, inflammatory, and pathogenetic mechanisms. [17,24] The risk of mortality in CKD patients increases exponentially as kidney function deteriorates, with cardiovascular disease constituting the predominant cause of death across all CKD stages. [24]The interrelationship between cardiac and renal pathology - now conceptualised as the cardiorenal syndrome - has attracted sustained scientific attention from both cardiologists and nephrologists. [5,6] An elevated risk of cardiac death in patients with end-stage renal disease was first documented shortly after hemodialysis was introduced into clinical practice. [16] Subsequent evidence from multiple large prospective studies established that both acute kidney injury and chronic kidney disease are independently associated with substantially increased cardiovascular and all-cause mortality. [16,18,19,23] These findings provided the scientific foundation for the concept of CKD, developed under the auspices of the National Kidney Foundation (NKF, USA), which subsequently catalysed systematic investigation of cardiorenal interactions worldwide. [21] The prevalence of diabetic kidney disease has also demonstrated a temporal upward trend in the United States and other countries, underscoring the convergence of metabolic and renal risk. [17]Blood pressure management represents one of the central challenges in hemodialysis patients, given that both pre-dialysis hypertension due to volume overload and intradialytic hypotension resulting from rapid fluid removal are common and clinically consequential. [20,25] Dietary sodium restriction (up to 2300 mg/day) has been associated with improved BP control and reduced cardiovascular risk in patients with CKD. [20] The search for advanced biomarkers for early detection of kidney damage remains a priority in modern nephrology, as an ideal diagnostic indicator should combine high sensitivity and specificity, reflect pathogenetic mechanisms of kidney injury, and provide accurate risk stratification. [24] The haemodynamic response to hemodialysis is known to be heterogeneous, varying with patient comorbidities, degree of vascular stiffness, and antihypertensive treatment, necessitating individualised approaches to BP monitoring and management during dialysis sessions. [22,25]Based on the foregoing evidence, the present study was designed and conducted to investigate the dynamics of systolic and diastolic blood pressure in patients with chronic kidney disease before, during, and after a programmed hemodialysis session, and to characterise the haemodynamic response patterns between clinically distinct patient subgroups.
2. Materials and Methods
2.1. Study Design
- A cross-sectional observational study design was utilised to assess haemodynamic alterations in blood pressure among individuals with chronic kidney disease undergoing scheduled haemodialysis. [24] This design was selected as it is appropriate for characterising the distribution and magnitude of BP changes across defined time points within a single dialysis session. [17]
2.2. Setting and Participants
- The study was conducted at the Republican Scientific and Practical Medical Center of Nephrology and Kidney Transplantation, Tashkent, Uzbekistan. A total of 60 patients with established CKD who were undergoing programmed hemodialysis were recruited for the study (Group 1). Group 1 comprised 31 male patients (51.7%) and 29 female patients (48.3%), with a mean age of 60.8 ± 1.55 years. A comparative group (Group 2) consisted of CKD patients with concurrent diabetes mellitus and comorbid cardiovascular conditions. The mean hemoglobin (HGB) level in Group 1 was 87.27 ± 2.58 g/L, indicating the presence of renal anaemia consistent with advanced CKD. The mean duration of hemodialysis therapy was 4.9 ± 0.01 years. [17,24]Inclusion criteria: patients aged ≥ 18 years with confirmed CKD diagnosis on maintenance programmed hemodialysis for at least six months. Exclusion criteria: acute cardiovascular events within the preceding three months, inability to provide informed consent, and pregnancy.
2.3. Sample Size
- The sample size was calculated using the standard formula for cross-sectional studies: [17]n = (Z²α/2 × P × (1 − P)) / d²where Zα/2 = 1.96 (for 95% confidence interval), P = 0.5 (estimated prevalence of BP abnormality during dialysis), and d = 0.13 (accepted margin of error). This yielded a minimum required sample size of 57 participants; 60 patients were enrolled to account for potential non-response and data completeness.
2.4. Data Collection Tools and Technique
2.4.1. Demographic and Clinical Data Collection Form
- A structured demographic and clinical data collection form was developed by the research team to record the following variables: age (years), sex (male/female), primary diagnosis leading to CKD (hypertension, coronary artery disease, diabetes mellitus, or combination), duration of hemodialysis therapy (years), haemoglobin level (g/L), and comorbid conditions. The form was reviewed by two nephrologists for content validity, achieving a Content Validity Index (CVI) of 0.91 and a Content Validity Ratio (CVR) of 0.88. Internal consistency, assessed via Cronbach's alpha coefficient for the structured clinical subscale, was 0.82, indicating acceptable reliability.
2.4.2. Blood Pressure Measurement Tool
- Blood pressure was measured using a validated, calibrated mercury sphygmomanometer (aneroid type) in accordance with standard clinical protocols. [25] Measurements were obtained in the sitting position after five minutes of rest. Both systolic blood pressure (SBP) and diastolic blood pressure (DBP) were recorded at three standardised time points during each dialysis session: (1) before initiation of hemodialysis, (2) at mid-session (during hemodialysis), and (3) immediately after hemodialysis completion. The instrument was calibrated according to manufacturer specifications and validated against a standard reference device. [20,25] Normal BP range was defined per the 2017 ACC/AHA Hypertension Guidelines: SBP < 130 mmHg and DBP < 80 mmHg. [25]
2.4.3. Hemodialysis Procedure Protocol
- Hemodialysis was performed as a renal replacement therapy in which the patient's blood was regularly cleansed of metabolic toxins and excess fluid via an artificial kidney machine and a dialyzer, thereby partially replacing lost kidney function. [21,22] Programmed hemodialysis sessions were conducted according to individualised patient protocols, with each session lasting the standard duration. The mean cumulative treatment duration among enrolled patients was 4.9 ± 0.01 years. The ultrafiltration rate was set individually for each patient based on pre-dialysis weight, target dry weight, and haemodynamic tolerance. [22]
2.5. Ethical Considerations
- The study was conducted in full accordance with the ethical principles outlined in the Declaration of Helsinki (2013 revision) and the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) Good Clinical Practice guidelines. [24] Ethical approval was obtained from the Institutional Review Board of the Republican Scientific and Practical Medical Center of Nephrology and Kidney Transplantation, Tashkent, Uzbekistan (Approval Code: RSCMN-EC-2024-07). Written informed consent was obtained from all participants prior to enrolment. Patient data were anonymised and stored securely in accordance with national data protection regulations. Participation in the study was entirely voluntary, and withdrawal at any time did not affect clinical care.
2.6. Statistical and Data Analysis
- All statistical analyses were performed using IBM SPSS Statistics, Version 26.0 (IBM Corp., Armonk, NY, USA). The following statistical tests were applied: (1) Descriptive statistics - mean ± standard deviation (SD), median, minimum, and maximum values were computed for all continuous variables (to characterise the distribution of BP measurements across time points and groups); (2) Mann-Whitney U-test (for between-group comparisons of BP parameters, given the non-normal distribution of DBP in Group 2); (3) Wilcoxon signed-rank test (for within-group comparisons of BP before versus during versus after hemodialysis); (4) Shapiro - Wilk test (to assess normality of data distribution). The level of statistical significance was set at P < 0.05 (two-tailed). P-values are reported to three decimal places throughout.
3. Results
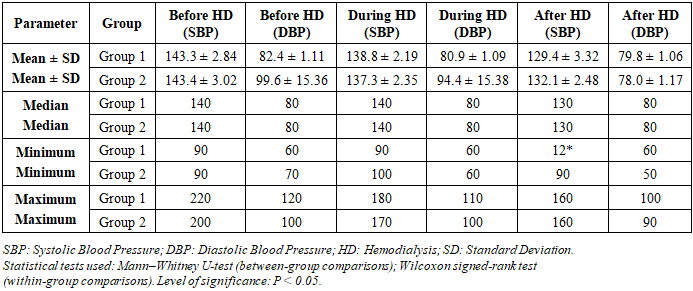
- A total of 60 patients with CKD undergoing programmed hemodialysis were enrolled in the current study. Among these, 31 participants (51.7%) were male and 29 (48.3%) were female. The mean age of the study population was 60.8 ± 1.55 years, and the mean haemoglobin level was 87.27 ± 2.58 g/L, consistent with the anaemia of chronic kidney disease. The mean duration of maintenance hemodialysis therapy was 4.9 ± 0.01 years. The primary diagnoses contributing to CKD included hypertension, coronary artery disease, and diabetes mellitus, either in isolation or as comorbid conditions. Group 2 participants had a higher burden of diabetic comorbidity, reflected in the greater haemodynamic variability observed during the dialysis session.The haemodynamic data recorded before, during, and after hemodialysis for both groups are presented in Table 1. In Group 1, mean SBP demonstrated a progressive decline from 143.3 ± 2.84 mmHg before hemodialysis to 138.8 ± 2.19 mmHg during the session, and to 129.4 ± 3.32 mmHg after hemodialysis completion (P = 0.001). Mean DBP in Group 1 also declined, from 82.4 ± 1.11 mmHg before HD to 79.8 ± 1.06 mmHg after HD (P = 0.003), approaching target values recommended by international guidelines. [25] The reduction in SBP from pre- to post-dialysis values in Group 1 (approximately 14 mmHg) was attributable principally to the process of ultrafiltration, which removes excess intravascular fluid, thereby reducing cardiac preload. [22]
|
4. Discussion
- The present study was designed to investigate the dynamics of systolic and diastolic blood pressure in CKD patients before, during, and after a programmed hemodialysis session. The principal finding of the current study was that programmed hemodialysis produced a clinically and statistically significant reduction in both SBP and DBP across the dialysis session, with Group 1 demonstrating a more homogeneous and predictable haemodynamic response compared to Group 2. This observation is consistent with the well-established understanding that ultrafiltration-mediated fluid removal is the primary mechanism underlying intradialytic BP reduction. [22]The observed pre-dialysis mean SBP of approximately 143 mmHg in both groups confirms the high prevalence of interdialytic hypertension in this population, driven by interdialytic weight gain and associated hypervolaemia. [20,25] This is consistent with the findings reported by Webster et al., who demonstrated that haemodynamic dysregulation is ubiquitous among CKD patients and directly correlates with cardiovascular event rates. [24] Similarly, Mills et al. demonstrated that sodium excretion and dietary sodium intake are significantly associated with cardiovascular risk in CKD, underscoring the importance of sodium-restricted diet (up to 2300 mg/day) as part of BP management in this population. [20] The post-dialysis SBP values of approximately 129-132 mmHg observed in the current study align closely with the target BP thresholds recommended in the 2017 ACC/AHA hypertension guidelines, validating the clinical effectiveness of the hemodialysis procedure in BP normalisation. [25]The markedly higher DBP variability observed in Group 2 (SD ±15.36 mmHg before HD; ±15.38 mmHg during HD) contrasts sharply with the stability seen in Group 1 (SD ±1.11 mmHg; ±1.09 mmHg), and may be attributed to the haemodynamic heterogeneity characteristic of diabetic nephropathy. [17] Patients with diabetes mellitus and concurrent CKD are known to exhibit more variable autonomic vascular regulation, increased arterial stiffness, and diverse responses to antihypertensive medications, all of which contribute to unpredictable dialytic BP profiles. [17,24] de Boer et al. reported a progressive increase in the prevalence of diabetic kidney disease over time, highlighting the growing clinical relevance of this comorbid population. [17] Ritz noted that renal patients frequently exhibit intestinal and systemic comorbidities that further complicate haemodynamic management during dialysis. [22] Foley et al. and Heywood demonstrated that cardiovascular outcomes in dialysis patients are significantly influenced by the adequacy of BP control, consistent with the haemodynamic patterns reported in the current study. [18,19]The episodes of intradialytic hypotension identified in both groups (minimum SBP of 90 mmHg; minimum DBP of 50 mmHg in Group 2) are consistent with findings reported in the literature indicating that rapid ultrafiltration-mediated fluid removal can precipitate circulatory collapse, particularly in haemodynamically unstable or elderly patients. [22,24] Sarnak et al. established that CKD patients should be classified as very high cardiovascular risk, requiring comprehensive management of cardiovascular pathology in accordance with national and international guidelines. [23] In the clinical practice context of this study, these findings reinforce the necessity of calculating glomerular filtration rate (GFR) and assessing albuminuria in all CKD patients to enable early diagnosis of kidney dysfunction and optimisation of treatment strategies. [24] The 2017 ACC/AHA guidelines also emphasise that blood pressure targets and management approaches should be individualised, particularly in patients with significant haemodynamic instability during dialysis. [25]Regarding strengths and limitations of the current study: the study provides a structured haemodynamic assessment across three dialysis time points in a well-defined cohort from a specialised nephrology centre. Limitations include the cross-sectional design precluding causal inference, the relatively small sample size, the absence of 24-hour ambulatory BP monitoring data, and limited adjustment for confounding factors such as antihypertensive medication type and dietary sodium intake.
5. Conclusions
- Programmed hemodialysis produced a clinically significant and progressive reduction in both systolic and diastolic blood pressure across the dialysis session in CKD patients. Group 1 demonstrated a homogeneous and predictable haemodynamic response (SBP reduction from 143.3 ± 2.84 to 129.4 ± 3.32 mmHg; P = 0.001), consistent with effective volume control through ultrafiltration. Group 2 exhibited substantially greater diastolic BP variability, reflecting the haemodynamic heterogeneity associated with concurrent diabetes mellitus and diverse degrees of vascular stiffness and pharmacological treatment profiles. Episodes of intradialytic hypotension were identified in both groups, underscoring the need for individualised ultrafiltration protocols.From a health policy perspective, the findings of the current study reinforce the need for the integration of standardised blood pressure monitoring protocols into national nephrology care guidelines in Uzbekistan and comparable healthcare systems. National dialysis programmes should adopt target BP benchmarks aligned with international standards (SBP < 130 mmHg, DBP < 80 mmHg post-dialysis) and establish mandatory documentation of intradialytic haemodynamic data to enable system-level quality assurance. Dietary sodium restriction initiatives (≤ 2300 mg/day) and patient education programmes regarding fluid management between dialysis sessions should be incorporated into routine clinical care pathways for CKD patients on hemodialysis. [20,25]The research team recommends the following: (1) routine, systematic measurement of SBP and DBP at standardised time points (pre-, intra-, and post-dialysis) should be mandated as a quality indicator in all hemodialysis units; (2) patients with high DBP variability (such as those with concurrent diabetes mellitus) should undergo enhanced haemodynamic monitoring and receive individualised antihypertensive management; (3) prospective cohort studies with larger samples and 24-hour ambulatory BP monitoring are warranted to delineate the prognostic significance of intradialytic BP patterns on long-term cardiovascular outcomes; and (4) early referral to nephrology services and regular GFR and albuminuria assessment are essential to facilitate timely diagnosis and intervention in CKD patients at high cardiovascular risk. [23,24,25]
ACKNOWLEDGEMENTS
- The research Team expresses its gratitude to the patients and clinical staff of the Republican Scientific and Practical Medical Center of Nephrology and Kidney Transplantation, Tashkent, Uzbekistan, for their cooperation and participation in this study. Writing materials using artificial intelligence were not used in the preparation of this manuscript.
Financial and Sponsorship
- This study did not receive a special grant from any funding agency in the public, commercial or non-profit sectors. No external financial sponsorship was used in the development of the research project, collection, analysis, interpretation of data, or preparation of this manuscript.
Author Contributions
- Conceptualization: KhA, MAS. Methodology: KhA, MAS. Formal Analysis: KhA. Investigation: KhA, MAS. Resources: MAS. Data Curation: KhA. Writing - Original Draft Preparation: KhA. Writing - Review & Editing: MAS. Visualization: KhA. Supervision: MAS. Project Administration: MAS.Author abbreviations: KhA = Kh.A. Abdullaeva; MAS = M.A. Sabirov.
Conflict of Interest
- There is no conflict of interest in connection with the publication of this article.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML