-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(3): 1065-1069
doi:10.5923/j.ajmms.20261603.45
Received: Jan. 15, 2026; Accepted: Feb. 6, 2026; Published: Mar. 7, 2026

Association of COMT Genetic Variants with the Development of Cardiovascular Complications During Anthracycline-Containing Therapy
Abdurakhman A. Kayumov1, Muborakkhon B. Zokirova1, 2, Nargiza M. Nurillaeva2
1Department of Hematology, Republican Specialized Scientific and Practical Medical Center of Hematology, Tashkent, Uzbekistan
2Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: Abdurakhman A. Kayumov, Department of Hematology, Republican Specialized Scientific and Practical Medical Center of Hematology, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Background. Acute leukemias are among the most severe oncohematological diseases requiring intensive chemotherapy. Cardiotoxicity remains one of the most significant treatment-related complications, limiting therapeutic options and affecting survival. Genetic factors, including polymorphisms in the COMT (catechol-O-methyltransferase) gene, may influence individual susceptibility to anthracycline-induced cardiac damage. Materials and Methods. The study included 102 patients with acute leukemia and 97 healthy controls matched by age and sex. Among the patients, 64 developed cardiological complications during therapy, while 38 had no signs of cardiotoxicity. The COMTVal158Met (rs4680) polymorphism was analyzed using Real-Time PCR. Statistical evaluation included comparison of allele and genotype frequencies (χ² test), and calculation of odds ratios (OR) and 95% confidence intervals (CI). Results. The COMT Val158Met polymorphism was not associated with the presence of acute leukemia (p > 0.2). However, a significant relationship was found between the Met allele and the risk of cardiotoxicity. Carriers of the Met allele demonstrated a higher risk of cardiac complications (OR = 3.8; 95% CI: 2.11–6.95), and the Met/Met genotype further increased this risk (OR = 4.5; 95% CI: 1.49–13.31). Conversely, the Val allele and Val/Val genotype exhibited protective effects (OR = 0.3; 95% CI: 0.14–0.47). Conclusion. The COMT Val158Met polymorphism does not predispose to the development of acute leukemia itself but has a strong influence on the occurrence of cardiotoxicity during therapy. The Met allele and Met/Met genotype are markers of increased risk, while Val/Val provides protection. Incorporation of COMT genotyping into clinical practice may enhance personalized risk prediction and improve treatment outcomes for patients undergoing anthracycline-containing chemotherapy.
Keywords: Acute leukemia, Cardiotoxicity, COMT Val158Met polymorphism, Genetic predisposition, Anthracycline chemotherapy, Personalized medicine
Cite this paper: Abdurakhman A. Kayumov, Muborakkhon B. Zokirova, Nargiza M. Nurillaeva, Association of COMT Genetic Variants with the Development of Cardiovascular Complications During Anthracycline-Containing Therapy, American Journal of Medicine and Medical Sciences, Vol. 16 No. 3, 2026, pp. 1065-1069. doi: 10.5923/j.ajmms.20261603.45.
Article Outline
1. Introduction
- Acute leukemias are among the most severe and life-threatening oncohematological diseases that require intensive chemotherapy. Despite substantial progress in diagnostics and treatment, the prognosis in a subset of patients remains unfavorable due to the high incidence of complications associated not only with the disease itself but also with the toxic effects of antineoplastic agents. One of the most significant adverse effects is cardiotoxicity, which limits the use of certain chemotherapeutic drugs and significantly reduces both the quality of life and overall survival of patients [1,2,3,4,5,6].Cardiological complications in the treatment of acute leukemias include myocardial dysfunction, the development of chronic heart failure, arrhythmias, and other cardiovascular disorders. Their pathogenesis is multifactorial and involves the dose-dependent effects of cytostatic agents (primarily anthracyclines), individual variations in drug metabolism, as well as the patient’s genetic predisposition. In recent years, particular attention has been paid to genetic markers that can be used to predict the risk of cardiotoxicity and to personalize therapeutic approaches [7,8,9].One of the most promising candidate genes is COMT (catechol-O-methyltransferase), which encodes the enzyme catechol-O-methyltransferase involved in the metabolism of catecholamines (adrenaline, noradrenaline, and dopamine). The Val158Met polymorphism (rs4680) results in a substitution of valine by methionine at position 158, leading to reduced enzymatic activity and altered catecholamine levels in the body. It is known that carriers of the Met allele exhibit lower enzyme activity and higher catecholamine concentrations, which may, in turn, contribute to impaired cardiovascular regulation and increased myocardial sensitivity to damaging factors [10].Several studies have demonstrated that this polymorphism is associated with the risk of various cardiovascular diseases, including arterial hypertension, coronary artery disease, and heart failure. However, its role in the development of cardiotoxicity in patients with acute leukemia remains insufficiently explored. Considering the importance of this issue, the investigation of the COMT Val158Met polymorphism in patients with acute leukemia undergoing chemotherapy is of considerable scientific and practical significance [11,12,13].
2. Aim of the Study
- The present study aimed to investigate the distribution and frequency of the Val158Met alleles and genotypes of the COMT gene among patients with acute leukemia, as well as in a control group of healthy individuals. Additionally, the study sought to assess the association between the COMT Val158Met polymorphism and the occurrence of cardiological complications during anthracycline-containing chemotherapy.
3. Materials and Methods
3.1. Study Population
- The main study group comprised 102 patients diagnosed with acute leukemia who underwent treatment at the Republican Specialized Scientific and Practical Medical Center of Hematology. Among them, 64 patients developed cardiological complications during therapy, whereas 38 patients completed treatment without signs of cardiotoxicity.The control group consisted of 97 apparently healthy controls matched for age and sex, with no evidence of oncohematological or cardiovascular disorders.The mean age of patients in the main group was 46 years, and the sex distribution was 68% male and 32% female.Exclusion criteria included:• pre-existing severe heart failure (NYHA class III–IV);• severe concomitant liver or kidney diseases.
3.2. Study Design and Subgroup Division
- The study included participants from both the main and control groups. In addition, two subgroups were identified within the main cohort:1. Patients who developed cardiological complications during treatment;2. Patients without cardiological complications.This design allowed for the comparative assessment of allele and genotype frequencies in relation to the presence or absence of cardiotoxic manifestations.
3.3. Genetic Analysis
- The Val158Met polymorphism (rs4680) of the COMT gene was analyzed using molecular genetic testing. Genotyping was performed by the real-time polymerase chain reaction (Real-Time PCR) method. DNA isolated from venous blood via the phenol–chloroform extraction method served as the template for amplification of the specific COMT gene fragment.The reaction mixture contained: PCR buffer, deoxynucleotide triphosphates (dNTPs), specific primers, thermostable DNA polymerase, and fluorescent TaqMan probes, which enabled differentiation of allelic variants.Amplification was carried out in an automatic thermocycler (Applied Biosystems StepOnePlus) following the standard protocol, including sequential stages of DNA denaturation, primer annealing, and elongation.The fluorescence signal was recorded at each amplification cycle, allowing real-time tracking of amplicon accumulation and identification of specific alleles. Allele-specific probes labeled with distinct fluorophores were used to discriminate between Val and Met alleles. The presence of a characteristic fluorescent signal indicated the corresponding genotype (Val/Val, Val/Met, or Met/Met).Quality control procedures included both negative controls (no template DNA) and positive controls (samples with known genotypes).
3.4. Statistical Analysis
- Statistical evaluation included the comparison of allele and genotype frequencies using the chi-square (χ²) test, as well as calculation of the relative risk (RR), odds ratio (OR), and 95% confidence intervals (CI). All analyses were performed to determine potential associations between the COMT Val158Met polymorphism and the development of cardiotoxic complications.
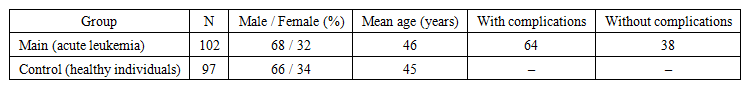 | Table 1. Characteristics of the Study Groups |
4. Results
4.1. Distribution of Alleles and Genotypes in the Main and Control Groups
- The analysis of the distribution of Val and Met alleles in the main and control groups revealed comparable frequencies (Val: 51.0% vs. 43.8%; Met: 49.0% vs. 56.2%). The differences did not reach statistical significance (χ² = 2.0; p = 0.29). Similarly, the distribution of genotypes (Val/Val, Val/Met, Met/Met) showed no significant differences between the groups (p > 0.2).Thus, the COMT Val158Met polymorphism was not associated with the presence of acute leukemia itself.
4.2. Comparison between Patients with and without Cardiological Complications
- A comparison of subgroups demonstrated pronounced differences in allele and genotype distribution. The Met allele was significantly more frequent among patients with complications (60.9% vs. 28.9%; χ² = 19.5; p = 0.01). The calculated odds ratio (OR) confirmed a high risk of complications for carriers of this allele (OR = 3.8; 95% CI: 2.11–6.95).Conversely, the Val allele predominated among patients without complications (71.1% vs. 39.1%), demonstrating a protective effect (OR = 0.3; 95% CI: 0.14–0.47).The Val/Val genotype was associated with a reduced risk of complications (12.5% vs. 52.6%; OR = 0.1; 95% CI: 0.05–0.32), whereas the Met/Met genotype markedly increased the likelihood of their development (34.4% vs. 10.5%; OR = 4.5; 95% CI: 1.49–13.31).These findings indicate that the Met allele and Met/Met genotype act as predictors of adverse outcomes with the development of cardiotoxicity, while the Val/Val genotype exerts a strong protective effect.
4.3. Comparison with the Control Group
- When comparing patients with complications to the control group, no significant differences were found in allele or genotype distribution (p > 0.3), suggesting that the COMT Val158Met polymorphism is not a risk factor for the occurrence of leukemia itself and does not represent a specific marker of oncohematological disease. Its relevance is observed primarily in the predisposition to cardiological complications.In contrast, patients without complications differed significantly from the control group.The Val allele (71.1% vs. 43.8%; OR = 3.1; 95% CI: 1.8–5.5) and the Val/Val genotype (52.6% vs. 21.6%; OR = 4.0; 95% CI: 1.85–8.73) were more frequent among patients without complications. Conversely, the Met allele and Met/Met genotype occurred less frequently compared to the control group, confirming the protective role of the Val allele associated with a lower risk of cardiotoxicity and a more favorable clinical course.
4.4. Summary of Genetic Associations
- Overall, the COMT Val158Met polymorphism was not associated with the risk of developing acute leukemia.However, the Met allele and Met/Met genotype were identified as markers of an increased likelihood of cardiotoxicity, whereas the Val allele and Val/Val genotype demonstrated protective effects and were associated with a stable treatment course without complications.These findings support the potential use of COMT Val158Met genotyping as a reliable tool for personalized prediction of complications and optimization of chemotherapy strategies in patients with acute leukemia.
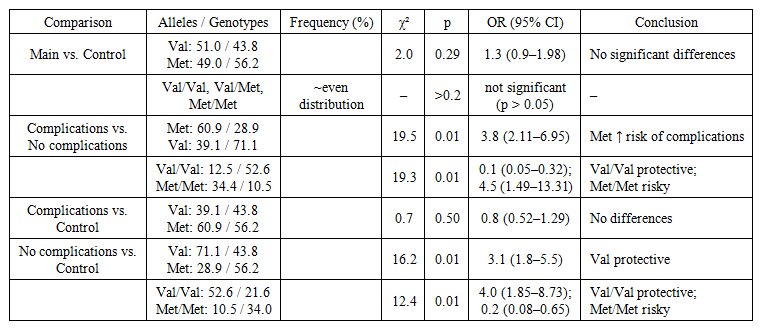 | Table 2. Associations of the COMT Val158Met Polymorphism |
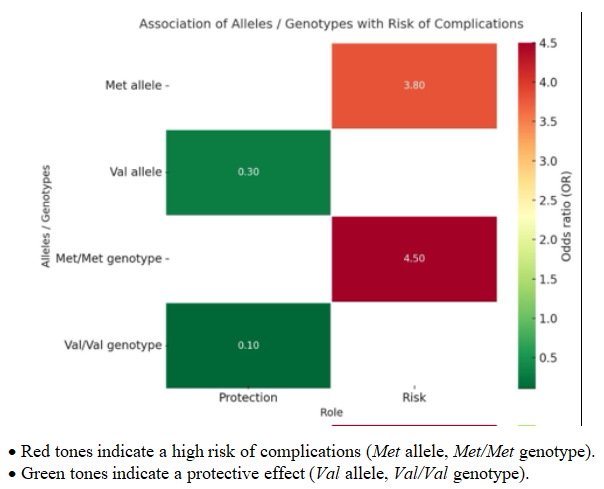 | Figure 1. Association of Alleles and Genotypes with the Risk of Complications |
5. Conclusions
- The analysis demonstrated that the COMT Val158Met polymorphism is not associated with the risk of developing acute leukemia itself, yet it plays a significant role in the development of cardiotoxic complications arising during therapy. This finding suggests that the polymorphism reflects not a predisposition to the disease per se, but rather individual characteristics of metabolic and neurohumoral regulation that determine the cardiovascular response to pharmacological stress.The presence of the Met allele, particularly the homozygous Met/Met genotype, was associated with a significantly higher risk of cardiological complications (OR up to 4.5). This observation is consistent with established evidence indicating reduced COMT enzyme activity in carriers of this variant, leading to altered catecholamine metabolism, which may cause autonomic imbalance and increased myocardial susceptibility to the toxic effects of chemotherapeutic agents.In contrast, the Val allele and the Val/Val genotype exhibited a strong protective effect, being associated with a more favorable clinical course and a lower incidence of cardiotoxicity. These results have practical clinical relevance, highlighting the potential use of the COMT Val158Met polymorphism as a prognostic genetic marker of complication risk. Such an approach opens new perspectives for personalized therapy selection and enhanced cardiac monitoring in high-risk patients.Overall, the findings contribute to a deeper understanding of the role of genetic factors in the development of therapy-related complications among oncohematological patients.Incorporating COMT Val158Met genotyping into the standard diagnostic algorithm could improve treatment efficacy, reduce the frequency of complications, and advance clinical practice toward the principles of personalized medicine.
ACKNOWLEDGEMENTS
- The authors express their sincere gratitude to the staff of the Republican Specialized Scientific and Practical Medical Center of Hematology (Tashkent, Uzbekistan) for their valuable assistance in patient recruitment, data collection, and laboratory analyses. The authors also acknowledge the contribution of all study participants for their cooperation and commitment to the research process.
DISCLOSURE
- The author declares no conflicts of interest relevant to the content of this article. No specific funding was received for this work.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML