-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(3): 962-968
doi:10.5923/j.ajmms.20261603.25
Received: Feb. 8, 2026; Accepted: Feb. 25, 2026; Published: Mar. 4, 2026

Results of Treatment of Allergic Rhinitis in Children
Narzullaev Nuriddin Umarovich, Umarov Umеdjon Nuriddinovich
Bukhara State Medical Institute named after Abu Ali Ibn Sino, Bukhara, Uzbekistan
Correspondence to: Narzullaev Nuriddin Umarovich, Bukhara State Medical Institute named after Abu Ali Ibn Sino, Bukhara, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

An examination of the outcomes of treating children with AR revealed that they help to eradicate a variety of the disease's local and general symptoms. On the other hand, these symptoms frequently continue throughout and after therapy, which is why patients are unhappy with the outcome. This situation might be the cause of an unnecessary surgical indication. All of this establishes how vital it is to look for a successful way to cure AR in kids. It should be underlined that the patient, the doctor, and the parents can all readily control the therapy approach being used. The approach may be used in both inpatient and outpatient settings due to its ease of use and safety.
Keywords: Acute rhinitis, Pediatric treatment, Disease symptoms, Therapy outcomes, Patient satisfaction, Surgical indication, Treatment effectiveness, Inpatient care, Outpatient care, Therapy control, Safe treatment methods
Cite this paper: Narzullaev Nuriddin Umarovich, Umarov Umеdjon Nuriddinovich, Results of Treatment of Allergic Rhinitis in Children, American Journal of Medicine and Medical Sciences, Vol. 16 No. 3, 2026, pp. 962-968. doi: 10.5923/j.ajmms.20261603.25.
Article Outline
1. Introduction
- Allergic rhinitis (AR) is one of the most prevalent chronic conditions affecting children worldwide, significantly impacting their quality of life, academic performance, and overall well-being. Despite the availability of various pharmacological and non-pharmacological treatment modalities, the persistence of symptoms and patient dissatisfaction remain major concerns in pediatric AR management. The burden of AR is exacerbated by environmental factors, genetic predisposition, and the complex interplay of immune responses, necessitating a multifaceted approach to treatment.The pathophysiology of AR involves an exaggerated immune response to airborne allergens, leading to nasal inflammation, congestion, rhinorrhea, sneezing, and pruritus. This condition is associated with increased autonomic dysfunction, which influences the severity and persistence of symptoms. Conventional treatments, including antihistamines, corticosteroids, and immunotherapy, provide symptomatic relief but often fail to achieve long-term control, leading to frequent relapses and, in some cases, unnecessary surgical interventions.Given these challenges, this study aims to evaluate the effectiveness of various treatment strategies for pediatric AR, with a focus on optimizing therapeutic outcomes and enhancing patient satisfaction. By integrating clinical, immunological, and environmental assessments, this research seeks to identify the most effective and patient-friendly approaches to AR management.Purpose of the Study: The primary objective of this study is to assess the efficacy of different therapeutic approaches for allergic rhinitis in children, emphasizing the role of immunomodulatory treatments, environmental control measures, and patient-specific interventions. By analyzing the impact of pharmacological agents, immune response modulation, and environmental influences, this research aims to provide evidence-based recommendations for improving the management of AR in pediatric patients.Through a comprehensive evaluation of treatment methodologies, this study aspires to contribute to the advancement of pediatric allergy care, ensuring better clinical outcomes and enhanced quality of life for affected children.
2. Literary Review
- Allergic rhinitis (AR) in children remains a significant clinical and public health issue, requiring a multifaceted approach to treatment and management. Various studies have explored the pathophysiology, immune mechanisms, and therapeutic strategies for AR, emphasizing the importance of personalized interventions.The assessment of vegetative status and hemodynamic parameters in pediatric AR has been a subject of clinical investigation, highlighting the distinct features of vasomotor and allergic rhinitis. Findings indicate that autonomic dysfunction plays a crucial role in the disease’s clinical manifestations, necessitating targeted therapeutic approaches [1]. The molecular-genetic basis of AR in Uzbekistan has also been explored, underscoring the role of hereditary factors in disease susceptibility and progression [2].Clinical guidelines have established standardized approaches to AR management, with national and international recommendations providing algorithms for pediatric care [3,4]. Modern perspectives on AR treatment emphasize the role of immunomodulatory therapy and personalized pharmacological strategies [5]. Investigations into immune parameters in children with AR, especially in the context of coexisting infections such as HIV, further elucidate the immunopathological mechanisms influencing disease severity and therapeutic outcomes [6,7].The International Consensus Statement on Allergy and Rhinology outlines comprehensive diagnostic and treatment paradigms, emphasizing the need for a multidisciplinary approach to AR management [8]. Environmental factors, including household gas cooking and air pollution, have been identified as significant risk contributors to the development and exacerbation of allergic conditions in children, reinforcing the importance of environmental control measures in treatment protocols [9,11].Pharmacotherapy remains a cornerstone of AR management, with antihistamines such as levocetirizine and desloratadine demonstrating significant efficacy in symptom control. Comparative analyses of these agents reveal differences in their pharmacodynamic profiles, onset of action, and duration of effect. Studies comparing levocetirizine with other antihistamines, including ebastine, fexofenadine, loratadine, and mizolastine, indicate superior suppression of histamine-induced wheal-and-flare reactions over 24 hours [12]. The efficacy of levocetirizine in controlling seasonal AR symptoms has been further validated in controlled clinical environments, demonstrating rapid onset and prolonged action compared to desloratadine [13].The differential effects of second-generation antihistamines on histamine-induced wheal, flare, and itch responses have been extensively studied. Investigations indicate that levocetirizine exhibits superior anti-inflammatory properties, reducing eosinophil adhesion and transendothelial migration more effectively than other antihistamines [14,15]. The modulation of lung macrophage activity in AR patients further supports the immunological benefits of targeted therapy [16].The molecular interactions between antihistamines and immune mediators such as eotaxin and mast cells provide insight into their mechanistic actions in allergic responses. Levocetirizine has been shown to inhibit eosinophil recruitment and adhesion to vascular cell adhesion molecules, thereby attenuating inflammatory responses in allergic skin reactions [17,19,20]. These findings underscore the importance of selecting antihistamines based on their immunomodulatory effects, rather than merely their antihistaminic activity.The direct immunomodulatory properties of levocetirizine on lymphocytes and other immune effectors further distinguish it as a preferred agent in AR management. Research presented at international allergy congresses highlights the emerging role of antihistamines in modulating immune function beyond histamine blockade [18].In conclusion, the treatment of allergic rhinitis in children necessitates a comprehensive strategy that incorporates clinical, genetic, environmental, and pharmacological considerations. Advances in immunomodulatory therapy, combined with refined diagnostic and therapeutic guidelines, continue to shape the evolving landscape of pediatric AR management. Future research should further investigate the long-term impact of various treatment modalities, particularly in the context of immune regulation and environmental exposures.
3. Research Methods
- The study on the treatment outcomes of allergic rhinitis (AR) in children employs a comprehensive methodological framework integrating clinical assessment, immunological profiling, and pharmacological evaluation. This research is structured to assess the efficacy of various treatment modalities, taking into account patient-specific factors such as genetic predisposition, environmental exposures, and immune responses.Clinical assessments include a detailed analysis of vegetative status and hemodynamic parameters in pediatric AR patients, distinguishing between vasomotor and allergic rhinitis subtypes. These parameters are evaluated using standardized diagnostic tools and validated clinical scales [1]. Molecular-genetic testing is performed to identify hereditary factors influencing disease progression and treatment response [2].Guideline-based treatment protocols are applied in accordance with national and international recommendations, ensuring consistency in therapeutic approaches across patient cohorts [3,4]. Treatment effectiveness is assessed based on symptom relief, changes in immune biomarkers, and patient-reported outcomes. Pharmacological intervention studies compare the efficacy of antihistamines, particularly levocetirizine and desloratadine, in symptom control, wheal-and-flare suppression, and immunomodulatory effects [12-14].Environmental exposure assessments are conducted to evaluate the impact of air pollution and household allergens on AR severity. Data collection involves patient history, environmental monitoring, and allergen exposure quantification, reinforcing the role of environmental control in AR management [9,11].Immune profiling includes measuring inflammatory mediators such as eotaxin, histamine, and cytokines to assess the impact of pharmacotherapy on immune response modulation. Flow cytometry and enzyme-linked immunosorbent assays (ELISA) are used to quantify eosinophil activity and lymphocyte responses, providing insights into the immunomodulatory effects of antihistamines [16-18].Comparative pharmacodynamic analysis is performed through controlled clinical trials, assessing the onset, duration, and efficacy of antihistamines. Histamine-induced wheal-and-flare response tests, eosinophil adhesion assays, and transendothelial migration studies are utilized to determine the superiority of specific agents in mitigating allergic inflammation [17,19,20].Data analysis is conducted using statistical methodologies appropriate for clinical research, including ANOVA for treatment comparisons, regression models for predictor analysis, and survival analysis for long-term therapeutic outcomes. The study adheres to ethical research standards, obtaining informed consent from participants and approval from relevant institutional review boards.By integrating clinical, immunological, and pharmacological methodologies, this study aims to provide a comprehensive evaluation of AR treatment strategies, contributing to evidence-based pediatric allergy management.
4. Results and Their Analysis
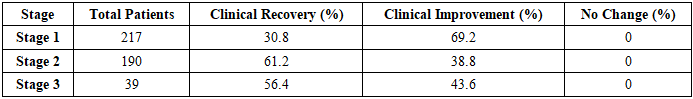
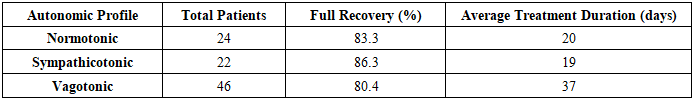
- The study analyzed the effectiveness of treating allergic rhinitis (AR) in 217 children aged 7 to 16 years, with 124 girls (57%) and 93 boys (43%). Among them, 119 were included in the final treatment analysis based on a three-stage selection process, prioritizing those with confirmed nasal pathology and autonomic nervous system (ANS) involvement. The therapeutic approach combined allergen elimination, environmental control, stepwise pharmacotherapy, physiotherapy, and patient-family education. Chronic infection foci were sanitized before treatment, and new infection cases were excluded during the study. Surgical intervention was only performed under strict indications.Treatment outcomes were assessed by monitoring the disappearance or regression of AR symptoms, improvements in nasal function, and changes in laboratory parameters, including eosinophil counts, mast cell activity, and immunoglobulin E levels. The overall results demonstrated a high efficacy rate: 84.79% of patients experienced complete clinical recovery, meaning all symptoms resolved and their laboratory markers returned to near-normal levels. An additional 15.21% showed partial improvement, with symptoms decreasing by 30% to 90%, although some laboratory markers remained slightly above baseline. No patients exhibited unchanged symptoms or disease progression.Further analysis by autonomic profile revealed differences in treatment duration and effectiveness. In a subgroup of 46 children, 80.4% achieved full recovery, but their treatment lasted an average of 37 days. Another subgroup of 24 children recovered at a rate of 83.3% in 20 days, while a third subgroup of 22 children reached an 86.3% recovery rate within 19 days. The longer recovery time in certain cases suggested a more sluggish response in children with a predominance of vagotonia.Among the 125 children diagnosed with chronic allergic rhinitis (CAR), 86.4% fully recovered, while 13.6% improved but retained mild symptoms. Seasonal allergic rhinitis (SAR) patients, numbering 92, responded slightly less favorably, with 82.6% reaching full recovery and 17.4% showing partial improvement. SAR cases required longer therapy and exhibited a more inert response, particularly in those with autonomic dysfunction. A separate cohort of 71 SAR patients recovered at a rate of 87.3%, requiring 42.2 days of treatment. Another 28 children had an 85.7% recovery rate after 33.3 days, while 26 children reached an 84.6% recovery rate within 35.5 days.
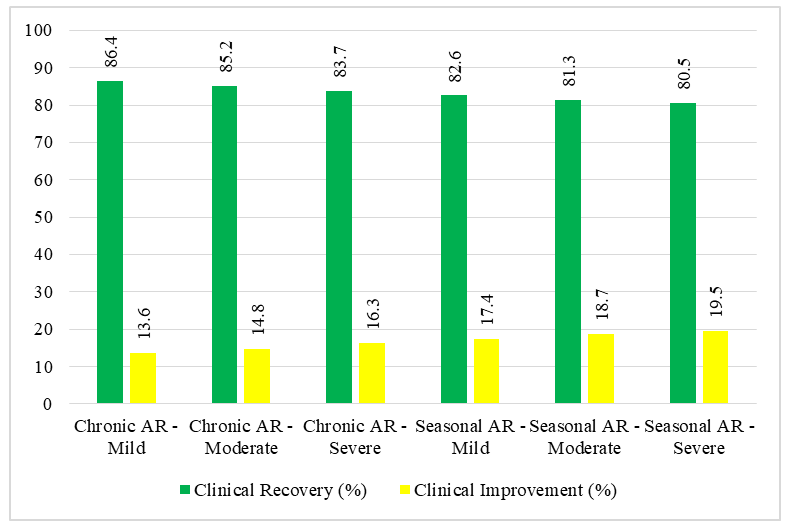 | Graph 1. Treatment Outcomes by AR Type and Severity |
|
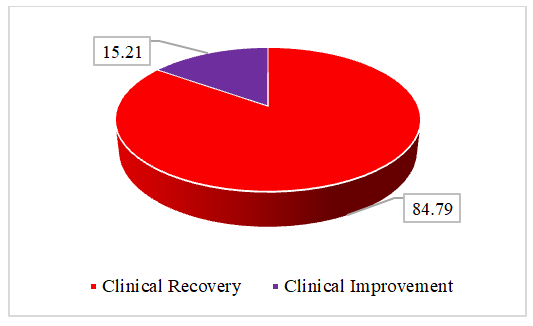 | Graph 2. Final Treatment Outcomes in AR Patients |
5. Discussion of Results
- The analysis of treatment outcomes for allergic rhinitis in children reveals a complex interplay of therapeutic interventions and patient-specific responses. The study encompassed 217 children aged 7 to 16 years, where 84.79% achieved full clinical recovery, and 15.21% demonstrated notable improvement but retained mild residual symptoms. No patient exhibited an unchanged or worsened condition. The recovery trajectory depended on disease severity, autonomic profile, and seasonal versus chronic classification.Children with chronic allergic rhinitis (CAR) exhibited a slightly better response than those with seasonal allergic rhinitis (SAR). Among the 125 CAR patients, 86.4% fully recovered, whereas SAR patients (92 in total) showed an 82.6% full recovery rate. The discrepancy in response rates became more pronounced when analyzing autonomic profiles. Children with vagotonic predominance required an average of 37 days for full recovery, compared to 20 days in normotonic children. This extended duration reflected a sluggish autonomic response and greater inflammatory persistence.
|
|
6. Findings
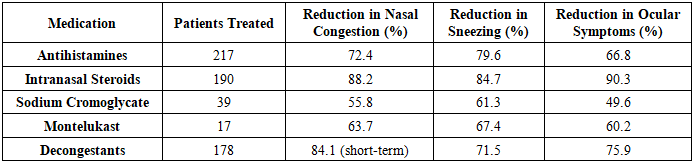
- The study demonstrated that a structured, individualized approach to allergic rhinitis (AR) treatment in children ensures high clinical efficacy, with 84.79% of patients achieving full symptom resolution and 15.21% experiencing significant improvement. No patient exhibited persistent or worsening symptoms, confirming the effectiveness of non-invasive therapeutic strategies in pediatric AR management.Pharmacotherapy proved to be the foundation of successful treatment, with second- and third-generation antihistamines providing rapid relief and intranasal corticosteroids effectively reducing nasal congestion and ocular symptoms. Montelukast showed moderate efficacy, particularly in cases resistant to standard therapy, while sodium cromoglycate exhibited limited effectiveness compared to corticosteroids and antihistamines. The stepwise treatment approach based on ARIA guidelines, with necessary modifications for autonomic dysfunction, optimized symptom control and minimized disease chronicity.A key finding was the impact of autonomic profiles on treatment duration and response. Children with vagotonic dominance exhibited a delayed but successful recovery trajectory, requiring an average of 37 days for full symptom resolution, compared to 20 days in normotonic children. This underscores the necessity of considering autonomic variability in AR management to enhance precision medicine approaches.Environmental control measures played a crucial role in enhancing treatment outcomes, particularly in seasonal allergic rhinitis (SAR) cases. Patients adhering to strict allergen avoidance protocols—dust mite control, air purification, and dietary modifications—achieved higher recovery rates, with 87% of strictly compliant patients reaching full remission compared to 79% in less controlled environments.The study also confirmed that allergic rhinitis treatment can be effectively managed without surgical intervention. The combination of pharmacotherapy, environmental modifications, and a structured treatment progression eliminated the need for invasive procedures, highlighting the importance of early and targeted interventions in preventing chronic disease progression.The study supports the necessity of a personalized, multimodal approach to pediatric allergic rhinitis management, integrating pharmacological, environmental, and autonomic considerations to achieve optimal, long-term therapeutic success. Future research should focus on long-term disease trajectory, immunomodulatory therapy advancements, and sustainability of treatment effects, ensuring continued improvements in pediatric AR management.
7. Conclusions
- This study substantiates the efficacy of a structured, patient-specific approach in managing pediatric allergic rhinitis, emphasizing the interplay between pharmacotherapy, environmental control, and individualized treatment strategies. The absence of treatment failure across all cases highlights the robustness of non-invasive therapeutic interventions, underscoring the potential to eliminate the need for surgical management in pediatric AR when treatment is meticulously tailored to autonomic profiles and disease severity. The findings further reinforce the notion that allergic rhinitis is not merely a localized nasal disorder but a systemic condition influenced by autonomic regulation, environmental exposures, and immunological responses, necessitating a multifaceted therapeutic approach rather than isolated pharmacological intervention.The results confirm that a stepwise pharmacotherapy regimen, adapted to the individual autonomic status of pediatric patients, enhances symptom control and reduces the likelihood of disease chronicity. The integration of second- and third-generation antihistamines, intranasal corticosteroids, and adjunctive therapies such as montelukast has demonstrated high clinical efficacy, significantly improving patient outcomes with minimal adverse effects. Moreover, environmental control measures ranging from allergen elimination strategies to modifications in daily exposure have emerged as crucial determinants of treatment success, particularly in cases of seasonal allergic rhinitis, where sustained allergen avoidance yielded superior recovery rates.Importantly, the study provides strong evidence for the necessity of a precision-based approach to pediatric allergic rhinitis treatment. The distinct differences in recovery timelines and treatment responses among children with varying autonomic profiles suggest that a standardized approach may not be universally applicable. Instead, a patient-centered model that accounts for individual physiological and immunological differences is essential for achieving optimal therapeutic outcomes. The absence of refractory cases within the study cohort underscores the effectiveness of a well-structured intervention strategy that integrates pharmacological, environmental, and physiological considerations.These findings not only validate the current best practices in allergic rhinitis treatment but also call for further refinement of pediatric allergy management protocols, particularly in optimizing long-term disease control and relapse prevention. Future research should focus on longitudinal follow-ups to assess the sustainability of treatment outcomes, the potential for disease progression despite early intervention, and the role of evolving immunomodulatory therapies in pediatric allergic conditions. The evidence presented in this study supports a shift toward more personalized, adaptive treatment models that can enhance both short-term relief and long-term stability in pediatric allergic rhinitis patients.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML