Ulugbek Bakhodirovich Akhmedov1, Firuz Khusenovich Kenzhaev2
1DSc., Head of the Cardiology Unit, State Institution “Republican Specialized Scientific and Practical Medical Center of Surgery named after Academician V. Vakhidov”, Tashkent, Uzbekistan
2MD, Surgeon, Department of Surgery of Coronary Artery Disease and Its Complications, State Institution “Republican Specialized Scientific and Practical Medical Center of Surgery named after Academician V. Vakhidov”, Tashkent, Uzbekistan
Correspondence to: Firuz Khusenovich Kenzhaev, MD, Surgeon, Department of Surgery of Coronary Artery Disease and Its Complications, State Institution “Republican Specialized Scientific and Practical Medical Center of Surgery named after Academician V. Vakhidov”, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
Morphological assessment of coronary artery disease does not always reflect the functional significance of coronary stenoses or myocardial ischemia. Myocardial multislice computed tomography (MSCT) perfusion may provide additional information for optimizing surgical decision-making in patients undergoing coronary artery bypass grafting (CABG). Methods: this single-center study included 80 patients with coronary artery disease who underwent CABG between 2019 and 2024. Patients were divided into two groups: the main group (n=35) underwent preoperative myocardial MSCT perfusion with assessment of myocardial viability, while the control group (n=45) did not undergo perfusion imaging. Results: patients in the main group demonstrated more pronounced improvement in left ventricular systolic function after CABG, with a significant increase in ejection fraction (p < 0.05), shorter hospital stay, and lower in-hospital mortality. Conclusions: myocardial MSCT perfusion is a valuable tool for preoperative assessment of myocardial viability and perfusion status, facilitating individualized surgical planning and improved clinical outcomes.
Keywords:
Coronary artery disease, Myocardial perfusion, Multislice computed tomography, Coronary artery bypass grafting, Myocardial viability
Cite this paper: Ulugbek Bakhodirovich Akhmedov, Firuz Khusenovich Kenzhaev, Practical Aspects of Myocardial MSCT Perfusion in the Preoperative Assessment of Patients with Coronary Artery Disease, American Journal of Medicine and Medical Sciences, Vol. 16 No. 3, 2026, pp. 941-947. doi: 10.5923/j.ajmms.20261603.20.
1. Introduction
Coronary artery disease (CAD) remains one of the leading causes of mortality and disability worldwide. According to the World Health Organization (WHO), based on the analysis of global mortality for 2021 [13], patients with multivessel coronary artery disease and/or reduced left ventricular ejection fraction (LVEF) represent the most complex category in terms of therapeutic decision-making. The clinical relevance of this problem is largely determined by the fact that morphological assessment of coronary stenoses based on invasive coronary angiography and/or computed tomography coronary angiography has a limited ability to accurately reflect their functional significance, the severity of myocardial ischemia, and, consequently, the true prognostic and clinical benefit expected from coronary revascularization [1]. Contemporary guidelines for chronic coronary syndromes emphasize the need for a rational use of imaging modalities for ischemia verification and risk stratification, since the functional significance of coronary lesions largely determines prognosis and the appropriateness of an invasive treatment strategy [7,8].Over the past decade, diagnostic algorithms for the management of patients with suspected or established chronic coronary disease have undergone substantial evolution, characterized by a pronounced shift toward an integrated approach based on combined anatomical and functional assessment of the coronary arteries and myocardial ischemia. American guidelines for the evaluation of chest pain, as well as current recommendations of the European Society of Cardiology for the management of chronic coronary syndromes, support the use of computed tomography coronary angiography (CTCA) as one of the key noninvasive first-line imaging modalities and highlight the role of functional testing when clarification of the hemodynamic significance of stenoses and prognostic assessment is required [5,14]. In this context, CT perfusion technologies have assumed a distinct role, as they are capable of bridging the gap between “anatomy” and “function.”Multislice computed tomography (MSCT) with myocardial perfusion assessment enables visualization of coronary blood flow disturbances directly at the level of myocardial tissue, allowing identification of which coronary stenoses are functionally associated with ischemia and which represent predominantly anatomical changes not accompanied by clinically significant perfusion deficits. Contemporary studies and meta-analyses demonstrate that the addition of CT perfusion to coronary CT angiography significantly increases the diagnostic value of the comprehensive approach compared with anatomical assessment alone, particularly when validated against invasive reference standards, including fractional flow reserve (FFR) [2].Myocardial MSCT perfusion is of particular practical importance in patients scheduled for surgical myocardial revascularization, namely coronary artery bypass grafting (CABG). In this patient population, it is crucial to determine the presence of ischemia within the target vascular territories, the volume of myocardium potentially capable of functional recovery, and the presence of irreversible myocardial injury (scar tissue), in which expansion of the revascularization volume does not result in clinical benefit and may be associated with increased operative risk [6]. In this regard, the use of myocardial MSCT perfusion as a method for comprehensive assessment of myocardial perfusion status and viability in patients with CAD is of special interest. The information obtained is of fundamental importance for patient stratification and surgical decision-making, as it allows substantiation of the appropriateness and extent of coronary artery bypass grafting depending on the severity of reversible and irreversible myocardial perfusion abnormalities [2,6].Classical concepts of ischemic phenomena - myocardial stunning, myocardial hibernation, and ischemic preconditioning underscore the heterogeneity of ischemic myocardial injury in coronary artery disease. A proportion of functional impairment is reversible provided that timely revascularization is performed, whereas scar formation lacks recovery potential [11]. In this context, assessment of myocardial viability and perfusion status is regarded as a key element in the individualization of surgical strategy, ranging from the choice between “complete” and “limited” revascularization to justification of alternative treatment approaches in high-risk patients [2].From the perspective of clinical implementation, myocardial MSCT perfusion is attractive due to its combination of high spatial resolution, the relative availability of modern CT platforms, and the ability to integrate perfusion findings into a unified preoperative decision-making framework alongside data from invasive coronary angiography, coronary CT angiography, and echocardiography. Current guidelines and contemporary review articles emphasize the importance of multimodal imaging and an integrated anatomical–functional approach in diagnosis and treatment planning for patients with complex coronary anatomy and ambiguous clinical presentation [3,7,8].Thus, evidence accumulated in recent years allows myocardial MSCT perfusion to be considered a promising and clinically meaningful tool that facilitates more accurate risk stratification, refinement of the potential benefit of revascularization, and selection of the optimal extent of surgical intervention with due consideration of myocardial viability.Objective. To determine the value of myocardial MSCT perfusion in selecting the extent and appropriateness of surgical revascularization in patients with coronary artery disease based on the assessment of myocardial perfusion status and viability.
2. Materials and Methods
The study was conducted at the Department of Surgery of Coronary Artery Disease and Its Complications of the State Institution “Republican Specialized Scientific and Practical Medical Center of Surgery named after Academician V. Vakhidov.” The analysis included 80 patients with coronary artery disease who underwent diagnostic evaluation and surgical treatment (coronary artery bypass grafting, CABG) between 2019 and 2024. The patients were divided into two groups. The main group comprised 35 patients with reversible perfusion abnormalities and preserved myocardial viability, as determined by myocardial MSCT perfusion imaging. The control group consisted of 45 patients in whom myocardial perfusion assessment by MSCT was not performed.
3. Results
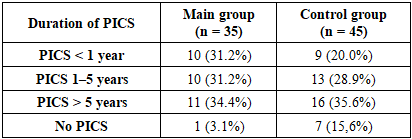
The mean age of patients in the main group was 58.5 ± 13.3 years, while in the control group it was 59.6 ± 5.5 years. No statistically significant difference in age was observed between the groups (p = 0.66), indicating their comparability with respect to this parameter.Assessment of body mass index (BMI) was included in the study to evaluate the anthropometric comparability of the groups, as overweight and obesity are recognized risk factors for coronary artery disease and may influence the clinical course of the disease, myocardial functional status, and outcomes of surgical revascularization [10].During the analysis of medical history data, the time elapsed since previous myocardial infarction with the development of postinfarction cardiosclerosis was evaluated. According to the literature, the duration of postinfarction cardiosclerosis is an important factor determining the extent of left ventricular remodeling and the severity of impaired contractile function, which is relevant for assessing the clinical status of patients with coronary artery disease [10].As shown in Table 1, the structure of the examined cohort in both groups was dominated by patients with a history of ischemic heart disease manifested as postinfarction cardiosclerosis (PICS) of more than 1 year in duration, with the largest proportion represented by patients with PICS lasting more than 5 years. In the main group, the proportion of patients with PICS of less than 1 year reached 31.2%, indicating the presence of individuals with a relatively recent myocardial infarction. In the control group, the proportion of patients with PICS of less than 1 year was 20.0%. The absence of postinfarction cardiosclerosis was observed in 3.1% of patients in the main group and in 15.6% of patients in the control group, reflecting the heterogeneity of the clinical composition of the studied population and varying degrees of chronic ischemic myocardial involvement.Table 1. Distribution of patients according to the duration of postinfarction cardiosclerosis (PICS), %
 |
| |
|
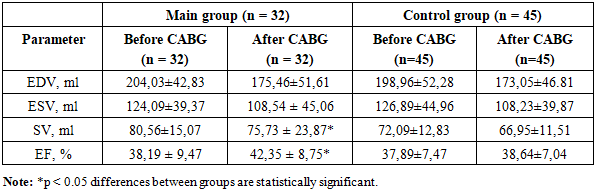
Thus, the distribution of patients according to the duration of postinfarction cardiosclerosis indicates a predominance of individuals with a long-standing history of ischemic myocardial injury, reflecting the chronic nature of the disease in the majority of the examined patients. The concomitant presence of patients with varying durations of PICS, as well as the absence of postinfarction changes in a subset of individuals, highlights the clinical heterogeneity of the study population and justifies the need for a differentiated approach to the assessment of left ventricular structural and functional status and the subsequent selection of an appropriate treatment strategy.It should be emphasized that in three patients included in the main group, a justified decision to refrain from direct surgical revascularization in the form of coronary artery bypass grafting was made based on the results of comprehensive clinical and instrumental evaluation. This strategy was driven by an unfavorable prognostic profile associated with the presence of extensive areas of nonviable myocardium identified both at rest and during stress testing by myocardial MSCT perfusion imaging. As an alternative approach, these patients underwent percutaneous coronary intervention with stenting of coronary arteries amenable to endovascular revascularization. However, follow-up examination performed one month after the intervention did not demonstrate a significant improvement in myocardial perfusion on MSCT, nor was there a clinically meaningful positive change in chronic heart failure symptoms or echocardiographic parameters of myocardial structural and functional status. Given that an alternative revascularization strategy inconsistent with the objectives and design of the present study was applied, these clinical cases were reasonably excluded from further outcome analysis, thereby ensuring methodological homogeneity of the study cohort and the validity of data interpretation.Analysis of left ventricular (LV) volumetric parameters based on echocardiographic data revealed that the LV end-diastolic volume (EDV) prior to CABG was increased in both groups, amounting to 204.03 ± 42.83 mL in the main group and 198.96 ± 52.28 mL in the control group, reflecting LV dilatation in the setting of chronic myocardial ischemia. Following CABG, a reduction in EDV was observed in both groups: to 175.46 ± 51.61 mL in the main group and to 173.05 ± 46.81 mL in the control group. Accordingly, the decrease in EDV was 28.57 mL (14.0%) in the main group and 25.91 mL (13.0%) in the control group. The favorable dynamics of LV volumetric parameters observed on echocardiography—manifested by reductions in EDV and end-systolic volume (ESV) and increases in stroke volume and ejection fraction—among patients in the main group indicate a greater potential for reversible left ventricular remodeling in the presence of viable myocardium (Table 2).Table 2. Dynamics of echocardiographic parameters with assessment of statistical significance (M ± SD)
 |
| |
|
Baseline end-systolic volume (ESV) values were elevated in both groups -124.09 ± 39.37 mL in the main group and 126.89 ± 44.96 mL in the control group -indicating a pronounced reduction in left ventricular contractile function. Following surgical revascularization, a decrease in ESV was observed in both groups, to 108.54 ± 45.06 mL in the main group and 108.23 ± 39.87 mL in the control group. The absolute reduction in ESV amounted to 15.55 mL (12.5%) in the main group and 18.66 mL (14.7%) in the control group, reflecting an improvement in systolic function and more effective emptying of the left ventricular cavity after CABG.Before surgery, stroke volume (SV) was reduced in patients of both groups, measuring 80.56 ± 15.07 mL in the main group and 72.09 ± 12.83 mL in the control group, which was consistent with the presence of systolic dysfunction. After CABG, SV values were 75.73 ± 23.87 mL in the main group and 66.95 ± 11.51 mL in the control group. Changes in stroke volume were moderate and reflected a redistribution of intracardiac hemodynamics in the context of reduced left ventricular end-volumes. More favorable SV values in the main group may be associated with recovery of contractility in viable myocardial segments.Left ventricular ejection fraction (LVEF) before coronary artery bypass grafting was reduced in all patients and amounted to 38.19 ± 9.47% in the main group and 37.89 ± 7.47% in the control group, meeting the study inclusion criteria and indicating significant left ventricular systolic dysfunction. After surgical revascularization, a positive trend in ejection fraction was observed in both groups. In the main group, LVEF increased to 42.35 ± 8.75%, which was statistically significant (p < 0.05) and reflected an improvement in global left ventricular contractile function. In the control group, post-CABG LVEF was 38.64 ± 7.04%, with a less pronounced increase.The more substantial increase in ejection fraction observed in the main group underscores the clinical relevance of preoperative myocardial viability assessment using multislice computed tomography with perfusion analysis when determining the surgical strategy.Thus, analysis of the dynamics of left ventricular contractile echocardiographic parameters demonstrated that coronary artery bypass grafting is associated with improvement in left ventricular functional status. More pronounced favorable changes, particularly in ejection fraction, were observed in patients with preoperatively confirmed myocardial viability, supporting the rationale for a comprehensive diagnostic approach in selecting candidates for surgical revascularization.Analysis of myocardial perfusion parameters at rest in the main group of patients revealed pronounced disturbances of coronary blood flow prior to surgical intervention (Fig. 1).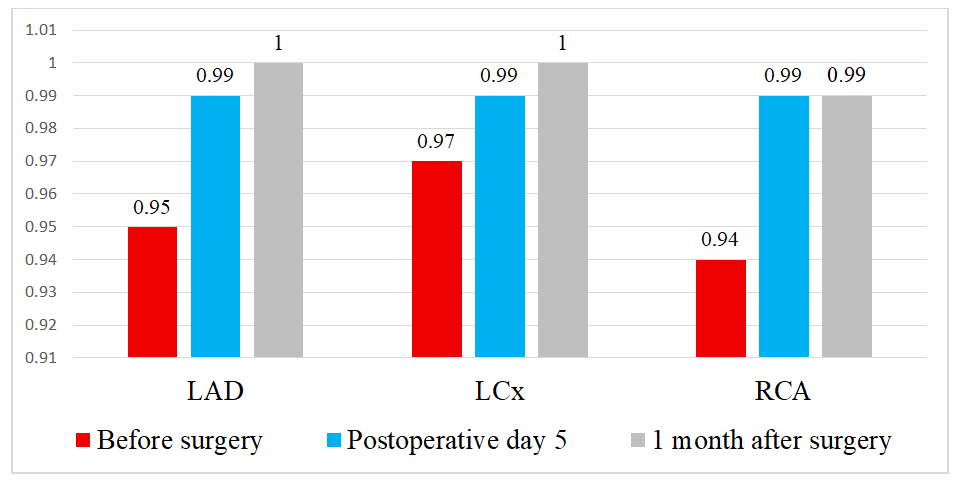 | Figure 1. Myocardial perfusion parameters at rest |
At baseline, a reduction in the TPR index was observed in the territory of the left anterior descending artery (LAD), as well as decreased TPR values in the territories of the left circumflex artery (LCx) and the right coronary artery (RCA). The most pronounced reduction in perfusion was registered in the RCA territory, which may indicate predominant involvement of this coronary basin and a greater severity of ischemic myocardial changes. In the early postoperative period, on day 5 after surgical revascularization, a clear positive trend in perfusion parameters was observed. Postoperative changes in TPR values in the LAD, LCx, and RCA territories demonstrated a tendency toward increase, approaching physiological levels, which reflects restoration of coronary microcirculation and improvement of myocardial perfusion already in the early postoperative period. In the late postoperative period, further improvement and stabilization of perfusion indices were noted, with achievement of near-normal values of both the integral parameter in the LAD territory and regional blood flow indices in the LCx and RCA territories (Fig. 1). These changes indicate sustained restoration of coronary myocardial blood supply and confirm the effectiveness of the performed surgical revascularization.Thus, dynamic myocardial MSCT perfusion assessment at rest allows objective evaluation of the severity of baseline coronary blood flow disturbances, as well as monitoring the stepwise recovery of myocardial perfusion in the early and late postoperative periods, highlighting the high diagnostic and prognostic value of this method.Analysis of myocardial MSCT perfusion parameters during pharmacological stress demonstrated that, prior to surgical intervention, patients in the main group exhibited a more pronounced reduction in perfusion indices compared with rest imaging (Fig. 2). Decreased TPR values in the LAD territory, as well as reduced perfusion in the LCx and RCA territories, reflect the presence of hemodynamically significant ischemic abnormalities that become evident predominantly under conditions of increased myocardial oxygen demand.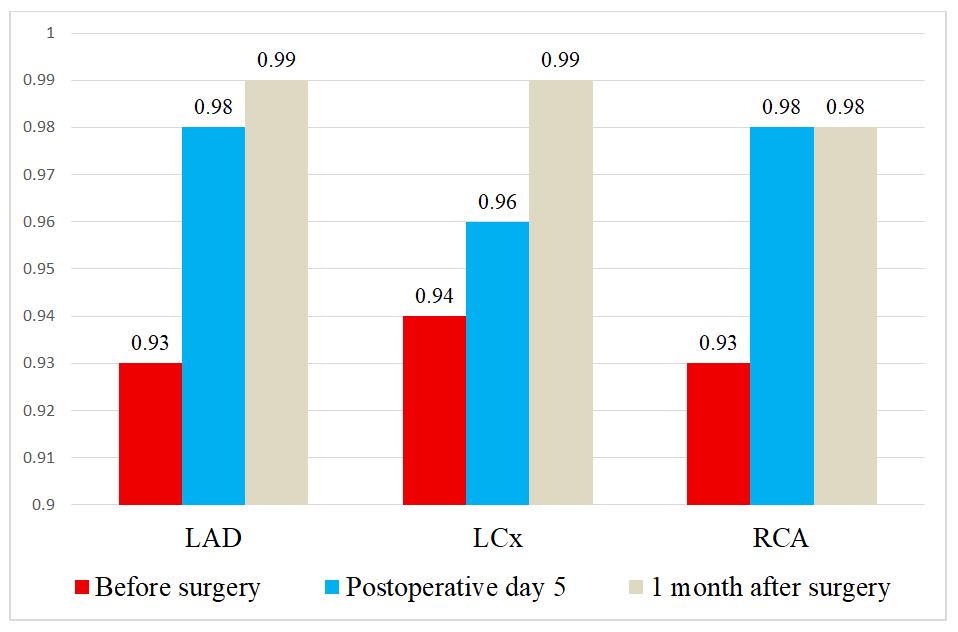 | Figure 2. Myocardial perfusion parameters during stress |
In the early postoperative period (postoperative day 5), a positive trend in TPR values was observed; however, recovery of stress-induced perfusion was less pronounced compared with rest conditions. This finding indicates persistence of a functional ischemic reserve in the early period after revascularization, which is expected and pathophysiologically justified.In the later postoperative period, a substantial improvement and stabilization of stress perfusion parameters were observed, with TPR values in the left anterior descending artery (LAD) territory as well as in the left circumflex artery (LCx) and right coronary artery (RCA) territories approaching normal levels. This dynamic pattern reflects restoration of coronary flow reserve and adequate myocardial perfusion under conditions of increased functional demand following surgical revascularization.Thus, myocardial MSCT perfusion imaging during stress enables detection of latent ischemic abnormalities prior to surgery and provides an objective assessment of the effectiveness of surgical revascularization and recovery of coronary flow reserve over time, underscoring its high diagnostic and prognostic value.The key intraoperative parameters of coronary artery bypass grafting obtained in the present study were comparable to those reported in the international literature. According to clinical observations by Van den Eynde J. et al. (2021), the mean duration of CABG ranges from 260 to 300 minutes [12]. In our study, the duration of surgical intervention was 263.5 ± 61.6 minutes in the main group and 283.3 ± 77.7 minutes in the comparison group, which is consistent with international clinical practice (Table 3).Table 3. Comparative characteristics of intraoperative parameters
 |
| |
|
According to data from international studies, the duration of cardiopulmonary bypass (CPB) during isolated CABG typically ranges from 70 to 120 minutes, increasing in the presence of multivessel coronary disease and greater technical complexity of the procedure [4,9,14]. In the present study, CPB times (93.7 ± 61.2 minutes and 113.7 ± 56.9 minutes) fall within this reported range.Aortic cross-clamp time, according to international reports, most commonly lies within 40–60 minutes [4,14]. Comparable values were obtained in our study, amounting to 52.6 ± 41.1 minutes and 44.2 ± 41.3 minutes, respectively.Thus, the intraoperative parameters observed in this study do not exceed the ranges described in the international literature, confirming the comparability of the surgical technique and operative conditions of CABG with international standards and allowing for a valid interpretation of differences in postoperative and functional outcomes.The mean length of hospital stay was shorter in the main group compared with the control group and amounted to 16.1 ± 9.0 bed-days versus 19.8 ± 5.6 bed-days, respectively. The difference between the groups was statistically significant (p = 0.039) (Table 4).Table 4. Length of hospital stay (bed-days)
 |
| |
|
The reduction in the length of hospital stay observed in the main group may indicate a more favorable postoperative course and faster clinical stabilization of patients. The obtained results suggest potential advantages of the applied diagnostic and therapeutic strategy, contributing to a shorter duration of inpatient treatment and accelerated patient rehabilitation.In the main group, the in-hospital mortality rate was 3.1%, whereas in the control group this indicator reached 8.9%, reflecting more favorable treatment outcomes among patients in the main group (Table 5).Table 5. In-hospital mortality in the study groups (n, %)
 |
| |
|
The reduction in in-hospital mortality in the main group compared with the control group (3.1% vs 8.9%) underscores the value of myocardial perfusion assessment in determining indications for surgical treatment and the extent of bypass grafting, and overall indicates the effectiveness of the applied diagnostic and therapeutic strategy. In patients of this group, the volume of irreversible myocardial injury was smaller, allowing prediction of clinical improvement following coronary artery bypass grafting. The obtained data are consistent with the concept of improving clinical outcomes through optimization of surgical and perfusion strategies.
4. Conclusions
Multislice computed tomography with myocardial perfusion analysis is an effective method for the preoperative assessment of patients with coronary artery disease. The use of MSCT perfusion enables objective evaluation of myocardial perfusion status and viability, which is of critical importance when selecting the strategy and extent of surgical revascularization.Implementation of this method facilitates differentiated selection of patients for coronary artery bypass grafting, individualization of the extent of intervention, and enhancement of surgical safety, particularly in patients with multivessel coronary artery disease and reduced left ventricular ejection fraction.Thus, myocardial MSCT perfusion may be recommended for integration into routine clinical practice of cardiology and cardiac surgery centers as an essential component of comprehensive preoperative evaluation of patients with coronary artery disease.
References
| [1] | Biancari F., Ruggieri V.G., Perrotti A., Svenarud P., Dalén M., Onorati F., et al. European Multicenter Study on Coronary Artery Bypass Grafting (E-CABG registry): study protocol for a prospective clinical registry and proposal of classification of postoperative complications. // Journal of Cardiothoracic Surgery. 2015; 10(1): 90. DOI: 10.1186/s13019-015-0292-z. |
| [2] | Danad I., Raijmakers P.G., Appelman Y., et al. Diagnostic performance of cardiac imaging methods to detect and localize myocardial ischemia. // Journal of the American College of Cardiology. 2017; 69(10): 123–134. DOI: 10.1016/j.jacc.2016.11.062. |
| [3] | Doenst T., Diab M., Sponholz C. et al. The impact of aortic cross-clamp time on outcome after coronary artery bypass grafting. European Journal of Cardio-Thoracic Surgery. 2019. |
| [4] | Gaudino M., Benedetto U., Fremes S., et al. Effect of on-pump vs off-pump coronary artery bypass grafting on long-term outcomes. // Journal of the American College of Cardiology. 2018; 71(22): 2606–2616.DOI: 10.1016/j.jacc.2018.03.506. |
| [5] | Gulati M., Levy P.D., Mukherjee D., et al.2021 AHA/ACC Guideline for the Evaluation and Diagnosis of Chest Pain. Circulation. 2021; 144(22): e368–e454. DOI: 10.1161/CIR.0000000000000950. |
| [6] | Head S.J., Milojevic M., Daemen J., et al. Myocardial viability testing and revascularization outcomes. // Annals of Thoracic Surgery. 2022; 113(1): 102–110. DOI: 10.1016/j.athoracsur.2021.05.068. |
| [7] | Knuuti J., et al. 2024 ESC Guidelines for the management of chronic coronary syndromes. // European Heart Journal. 2024; 45(10): 946–1026. DOI: 10.1093/eurheartj/ehad139. |
| [8] | Knuuti J., Wijns W., Saraste A., et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes. // European Heart Journal. 2020; 41(3): 407–477. DOI: 10.1093/eurheartj/ehz425. |
| [9] | Lamy A., Devereaux P.J., Prabhakaran D., et al. Effects of off-pump and on-pump coronary-artery bypass grafting at 5 years. // New England Journal of Medicine. 2016; 375(24): 2359–2368. DOI: 10.1056/NEJMoa1602001. |
| [10] | Pontone G., Rossi A., Guglielmo M., et al. Myocardial CT perfusion imaging: State of the art and clinical applications. // European Radiology. 2021; 31(8): 5612–5628. DOI: 10.1007/s00330-021-07777-6. |
| [11] | Ranucci M., Baryshnikova E., Ciotti E., Ranucci M., Silvetti S. Hemodilution on cardiopulmonary bypass: Thromboelastography patterns and coagulation-related outcomes. // Journal of Cardiothoracic and Vascular Anesthesia. 2017; 31(5): 1588–1594. DOI: 10.1053/j.jvca.2017.04.014. |
| [12] | Van den Eynde J, Vaesen Bentein H, Decaluwé T, De Praetere H, Wertan MC, Sutter FP, Balkhy HH, Oosterlinck W. Safe implementation of robotic-assisted minimally invasive direct coronary artery bypass: application of learning curves and cumulative sum analysis. // J Thorac Dis. 2021; 13(7): 4260–4270. doi: 10.21037/jtd-21-775. |
| [13] | World Health Organization. The top 10 causes of death [Electronic resource]. Geneva: World Health Organization; 2024. Available at: https: // www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death. |
| [14] | Writing Committee Members. 2023 AHA/ACC Guideline for the Management of Chronic Coronary Disease. // Journal of the American College of Cardiology. 2023; 82(13): 1247–1413. DOI: 10.1016/j.jacc.2023.08.1036. |




 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML



