-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(3): 938-940
doi:10.5923/j.ajmms.20261603.19
Received: Feb. 6, 2026; Accepted: Feb. 23, 2026; Published: Mar. 4, 2026

Safety and Effectiveness of Antiepileptics in Focal Epilepsy in Practice
Usmanova Durdona Djurabaevna1, Latipova Nilufar Kenjaevna2, 3
1Doctor of Medical Sciences, Professor, Department of Neurology and Child Neurology, Medical Genetics, Tashkent State Medical University, Tashkent, Uzbekistan
2PhD Candidate, Department of Neurology, Andijan State Medical Institute, Andijan, Uzbekistan
3Medical Faculty, University of Business and Science, Namangan, Uzbekistan
Correspondence to: Latipova Nilufar Kenjaevna, PhD Candidate, Department of Neurology, Andijan State Medical Institute, Andijan, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Selecting an antiepileptic drug (AED) is a complex clinical and pharmacological task requiring comprehensive consideration of the patient's age, clinical type of epilepsy, severity and frequency of epileptic seizures, as well as the drug's efficacy and tolerability profile. Despite the availability of a wide range of AEDs, questions of their comparative efficacy and safety, particularly in terms of assessing long-term clinical outcomes, remain of high scientific and practical importance. Therefore, studying the frequency and nature of adverse drug reactions (ADRs) that occur in the central nervous system with AEDs is particularly relevant. The aim of this study was to compare the frequency of adverse drug reactions (ADRs) with zonisamide and carbamazepine in patients with focal epilepsy. Material and methods: A retrospective analysis of the frequency and nature of adverse drug reactions (ADRs) recorded in the medical records of 87 patients observed in the epilepsy and paroxysmal conditions diagnostic and treatment office of an epileptologist in 2024–2025 was conducted. To assess the safety of AEDs in this study, the indicator reflecting the number of patients with registered ADRs was used as an adverse outcome. Results: Analysis of therapy showed that the use of carbamazepine was significantly more often accompanied by the development of adverse events from the central nervous system than zonisamide. Conclusions: The results of the present study showed that ADRs associated with central nervous system damage were recorded predominantly with the use of carbamazepine.
Keywords: Focal epilepsy, Antiepileptic drugs, Adverse drug reactions, Zonisamide, Carbamazepine
Cite this paper: Usmanova Durdona Djurabaevna, Latipova Nilufar Kenjaevna, Safety and Effectiveness of Antiepileptics in Focal Epilepsy in Practice, American Journal of Medicine and Medical Sciences, Vol. 16 No. 3, 2026, pp. 938-940. doi: 10.5923/j.ajmms.20261603.19.
1. Introduction
- Selecting an AED requires a comprehensive assessment of the patient's age, clinical type of epilepsy, seizure severity, and the drug's efficacy and tolerability profile. Studying the comparative efficacy and safety of AEDs, particularly with regard to long-term outcomes, remains a pressing issue. Of particular relevance is determining the incidence of adverse drug reactions affecting the central nervous system during AED use, including cognitive-behavioral impairment.These studies are characterized by certain limitations associated with the use of strict and narrow inclusion criteria related to age, gender, comorbidities, and concomitant administration of other medications. Evaluation of the frequency of adverse drug reactions (ADRs) arising from the central nervous system (CNS) during the use of antiepileptic drugs, including cognitive-behavioral changes, is relevant [3].Drug safety is a key requirement in modern pharmacotherapy. However, it should be noted that safety assessments in pre-registration clinical trials are often limited, typically due to small sample sizes and short observation periods. This necessitates continued monitoring of drug safety and tolerability after their introduction into clinical practice. Proper selection of antipsychotic drugs, taking into account the drug's mechanisms of action, can help reduce the number of attacks and/or achieve remission [2].Zonisamide is a new-generation antiepileptic drug. It is registered for use as monotherapy, including initial monotherapy for focal seizures with or without secondary generalization. Since 2012, zonisamide has been one of the few antiepileptic drugs whose efficacy in the initial treatment of focal seizures in adults has been confirmed according to the strict standards of modern medicine established by the International League Against Epilepsy (level of evidence A), along with carbamazepine [1].The aim of the study was to retrospectively evaluate the frequency of adverse drug reactions observed after therapy with zonisamide in comparison with carbamazepine in patients with focal epilepsy.
2. Materials and Methods
- The study included a retrospective analysis of the frequency and nature of ADRs recorded in the medical records of 87 patients observed in the office for diagnosis and treatment of epilepsy and paroxysmal conditions by an epileptologist in 2024–2025. The study included patients meeting the following criteria: a) a reliably established diagnosis of structural focal epilepsies or focal epilepsies of unspecified etiology according to the International Classification of Epilepsy and Epileptic Syndromes (2017) [4]; b) use of monotherapy with carbamazepine or zonisamide. To assess the safety of antiepileptic drugs, the study used an adverse outcome, expressed as the number of patients with registered ADRs. Differences were considered statistically significant at p < 0.05.
3. Results
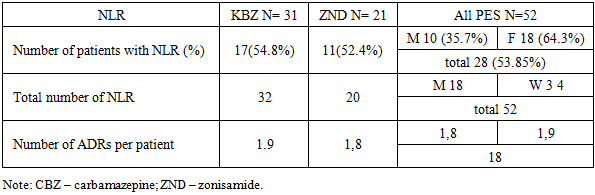
- The study included an analysis of ADRs recorded in the medical records of 52 patients with focal epilepsy receiving monotherapy with various AEDs. The clinical picture of epilepsy was characterized by the following types of seizures: simple partial – 8% (n= 4), complex partial (automatisms) – 23% (n=12), a large group consisted of patients with secondary generalized seizures – 69% (n=36). In the structure of clinical manifestations, focal seizures without impairment of consciousness prevailed, recorded in 38 patients (73%), while focal seizures with impaired consciousness were observed in 14 patients (27%). According to seizure frequency, the patients were distributed as follows: less than once a month – 18 people (34.5%); several times a month, but less than once a week – 24 people (46%); Several times a week, but less than once a day — 6 people (11.5%); once a day or more — 4 people (7.7%). Gender distribution: 34 (65.4%) men and 18 (34.6) women, of which the average age of men was 36.37 ± 8.14 years, women — 38.21 ± 5.06 years. Antiepileptic therapy in all patients was carried out as monotherapy. Carbamazepine was used in 31 patients (57.7%), while zonisamide was received by 21 patients (42.3%). The dose of carbamazepine was titrated starting from 200 mg per day, with an increase of 100 mg up to a daily dose of 500 mg, divided into two doses.During the study, a total of 52 adverse drug reactions were identified in 28 patients, accounting for 53.85% of the study group. Among patients with registered ADRs, 10 (35.7%) were men and 18 (64.3%) were women. The average number of ADRs per patient was 1.8 (Table 1). In women, the average number of ADRs per patient was higher and amounted to 1.9, while in men it was 1.8, with a statistically significant difference between the groups (p = 0.008).
|
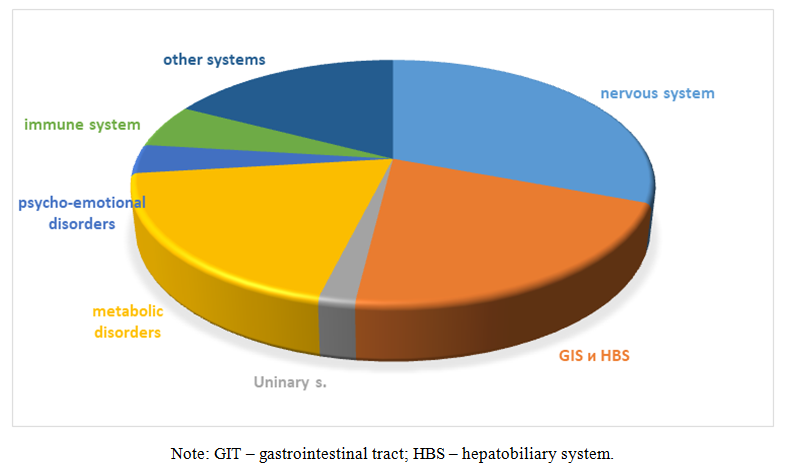 | Figure 1. Structure of NLR by organ systems during monotherapy,% (n=52) |
4. Conclusions
- The results of this study indicate that, overall, adverse events associated with central nervous system damage were most frequently reported with carbamazepine.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML