Isaxanova Nigora Xikmatovna1, Shamansurova Elmira Amanullayevna2
1Ph.D., Associate Professor Tashkent State Medical University, Tashkent, Uzbekistan
2Dsc., Professor, Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: Isaxanova Nigora Xikmatovna, Ph.D., Associate Professor Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Abstract
In recent years, major advances have been achieved in reducing the number of cardiac complications in children through early prenatal and postnatal diagnosis of congenital heart defects, as well as through the early application of surgical interventions in cardiac surgery. Background: To study the characteristics of adaptive potential in children with congenital heart defects, including patients before and after surgical correction. Methods: The study was conducted at the Pediatric Clinic of Tashkent State Medical University and was based on the analysis of adaptive capacity in 294 children aged 1 to 7 years during the preoperative and postoperative periods. All patients were divided into two groups: Group 1 (160 children) — children with cyanotic congenital heart defects; Group 2 (134 children) — children with acyanotic congenital heart defects. The research methods included assessment of growth and development, evaluation of acid–base balance parameters in the blood, and measurement of biochemical marker levels. Results. The obtained results indicate that in children with acyanotic congenital heart defects, changes in acid–base status and electrolyte parameters are less pronounced compared to the cyanotic group and, in most cases, are compensated more rapidly after surgery. However, significant changes in gas exchange, electrolyte levels, and glucose concentration during the cardiopulmonary bypass stage indicate the need for regular monitoring even in this group of patients. Conclusions. The study results demonstrate that the majority of congenital heart defects are diagnosed within the first year of life, confirming the critical importance of early diagnosis and the organization of targeted specialized dispensary follow-up for this category of children.
Keywords:
Cyanotic congenital heart defects, Acid–base parameters, Biochemical markers
Cite this paper: Isaxanova Nigora Xikmatovna, Shamansurova Elmira Amanullayevna, Indicators of Adaptation Mechanisms in Children with Congenital Heart Defects, American Journal of Medicine and Medical Sciences, Vol. 16 No. 3, 2026, pp. 916-921. doi: 10.5923/j.ajmms.20261603.15.
1. Introduction
In recent years, early prenatal and postnatal diagnosis of congenital heart defects, along with the early application of surgical interventions in cardiac surgery, has been considered a major advancement leading to a reduction in the number of cardiac complications observed in children.According to the results of global epidemiological analyses, the number of children born with congenital heart defects is steadily increasing worldwide. According to data from the World Health Organization, approximately 303,000 children with congenital anomalies die within the first four weeks of life. Congenital defects often lead to long-term disability, which in turn has a significant impact on affected individuals, their families, healthcare systems, and society as a whole.Congenital heart defects (CHDs), classified under ICD-10 codes Q20–Q28, are the most common congenital anomalies and represent one of the leading causes of child mortality. Considering the above, the prevalence and incidence of heart diseases, their early detection, diagnosis, and the development of preventive measures are among the most pressing issues in modern healthcare. The increasing prevalence of this condition in the population, along with insufficiently studied pathogenetic mechanisms and prognostic factors, necessitates further scientific research in this field. Compared with other congenital anomalies worldwide, congenital heart defects occupy a leading position in terms of prevalence among children and as one of the main causes of mortality [1,2,3].In children with congenital heart defects, certain alterations are observed in the acid–base status and electrolyte balance of the blood, and these parameters change dynamically during the various stages of surgical interventions.Chronic hypoxia in childhood, especially in conditions associated with congenital heart defects (CHDs), is considered one of the key pathogenetic factors affecting the body. One of the central molecular mechanisms activated in response to hypoxia is the hypoxia-inducible factor-1 alpha (HIF-1α) signaling pathway. This factor is a crucial regulator that ensures angiogenesis, erythropoiesis, and metabolic adaptation of tissues in the pediatric organism [8-12].In pediatric practice, it is well known that under hypoxic conditions, the degradation of HIF-1α is inhibited, allowing it to translocate into the cell nucleus and activate the transcription of genes involved in vascular growth and oxygen transport, including VEGF, PD-ECGF/TP, and erythropoietin (EPO). This mechanism is regarded as a physiological response aimed at compensating for oxygen deficiency in children [5,6].Furthermore, according to the literature, children with cyanotic congenital heart defects exhibit significantly higher levels of HIF-1α and related pro-angiogenic factors in myocardial tissue compared with those with acyanotic forms and healthy children. In addition, elevated mRNA levels of HIF-1α, VEGF, and EPO in the blood of newborns with cyanosis and persistent pulmonary hypertension are considered early laboratory markers of generalized hypoxia [10,15].
2. Materials and Methods
The adaptive status of pediatric patients with a confirmed diagnosis of congenital heart defects was studied during the preoperative and postoperative periods. Children with congenital heart defects were divided into two main groups: Group 1 included 160 children (47.4%) with cyanotic congenital heart defects, and Group 2 included 134 children (39.7%) with acyanotic congenital heart defects.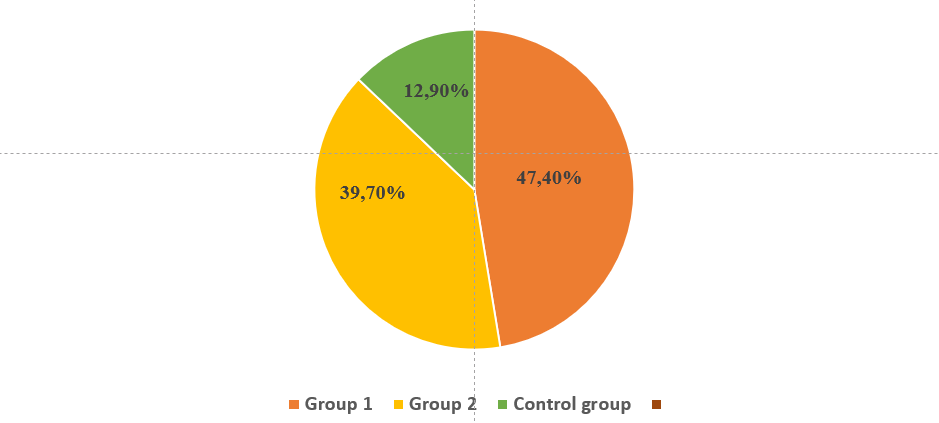 | Figure 1. Distribution of the examined pediatric patients by study groups |
The analysis of sex distribution among children with congenital heart defects showed a predominance of female patients among the examined children. According to the results of the sex-based analysis, in Group 1 the number of boys was 71 (44.6%), while girls accounted for 89 (55.4%). In Group 2, there were 62 boys (46.4%) and 72 girls (53.6%). In the control group, boys comprised 8 children (40.0%) and girls 12 children (60.0%).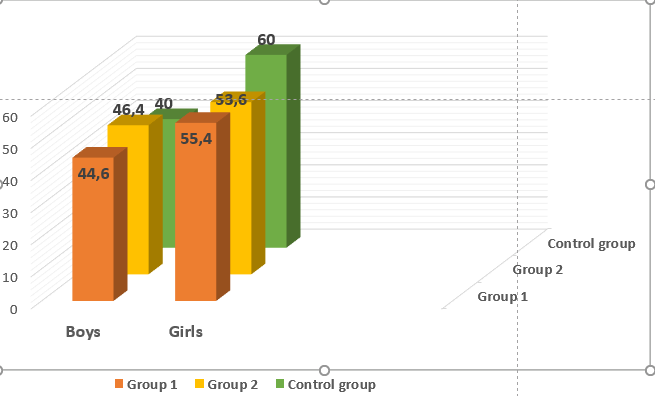 | Figure 2. Distribution of patients by sex (absolute numbers) |
The age range of the examined children was from birth to 7 years. In the age group of 0–1 year, the proportion of children was 78.9% in Group 1, 51.5% in Group 2, and 16.7% in the control group. Children aged 1–3 years accounted for 14.2% in Group 1, 22.6% in Group 2, and 43.3% in the control group. The proportion of children aged 3–7 years was 4.3% in Group 1, 13.86% in Group 2, and 39.0% in the control group. Children older than 7 years constituted 2.6% in Group 1, 12.6% in Group 2, and 1% in the control group.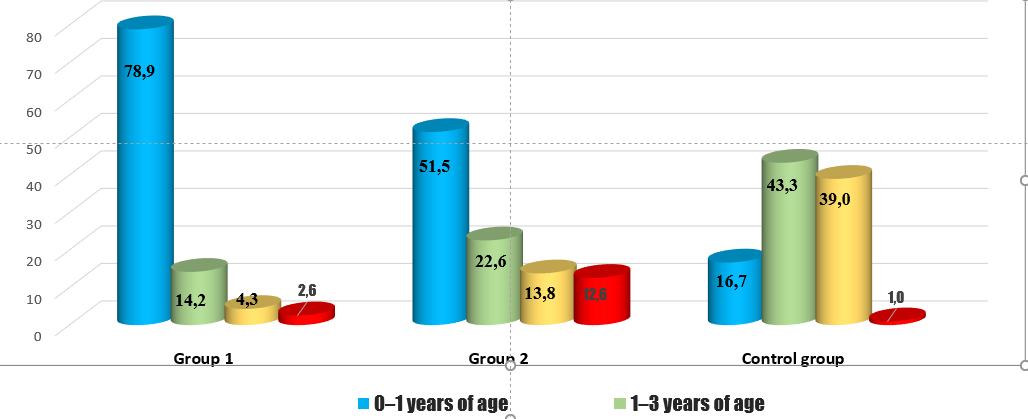 | Figure 3. Age distribution of the examined pediatric patients by study groups |
3. Result and Discussion
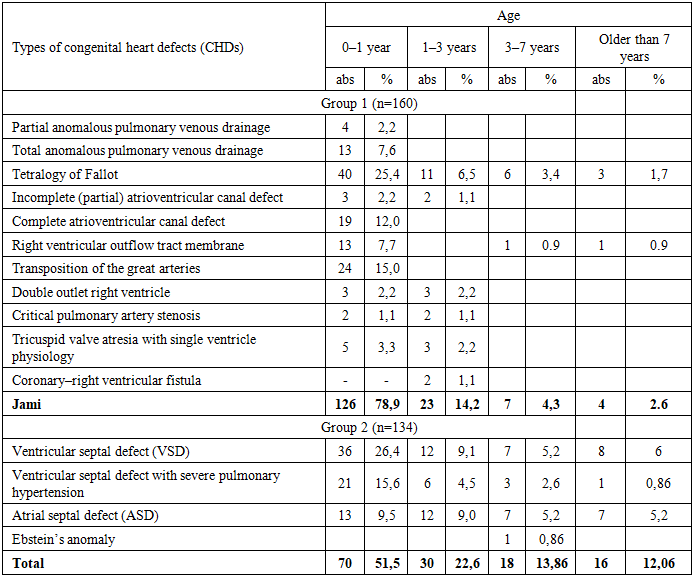
Analysis of the distribution of defect types and age among children in Group 1 with congenital heart defects (CHDs) showed that partial anomalous pulmonary venous drainage (PAPVD) was identified in 4 children (2.2%) aged from birth to 1 year. Total anomalous pulmonary venous drainage (TAPVD) was observed in 13 children (7.6%) in the 0–1 year age group.Tetralogy of Fallot was diagnosed in 40 children (25.2%) aged 0–1 year, in 11 children (6.5%) aged 1–3 years, in 6 children (3.4%) aged 3–7 years, and in 3 children (1.7%) older than 7 years.The incomplete form of atrioventricular canal defect (partial AV canal) was detected in 3 children (2.2%) aged 0–1 year and in 2 children (1.1%) aged 1–3 years.The complete form of atrioventricular canal defect (complete AV canal) was recorded in 19 children (12.0%) aged 0–1 year.Right ventricular outflow tract membrane (RVOT membrane) was identified in 13 children (7.7%) aged 0–1 year, in 1 child (0.9%) aged 3–7 years, and in 1 child (0.9%) older than 7 years.Transposition of the great arteries (TGA) was observed in 25 children (14.4%) aged 0–1 year. Double outlet right ventricle (DORV) was recorded in 3 children (2.2%) aged 0–1 year and in 3 children (2.2%) aged 1–3 years. Critical pulmonary artery stenosis was diagnosed in 2 children (1.1%) aged 0–1 year and in 1 child (1.1%) aged 1–3 years. Tricuspid valve atresia with a single ventricle physiology was observed in 5 children (3.3%) aged 0–1 year and in 2 children (2.2%) aged 1–3 years. Coronary–right ventricular fistula was observed only in 2 children (1.1%) aged 0–1 year (Table 1).Table 1. Description of congenital heart defect types and age distribution in the examined children
 |
| |
|
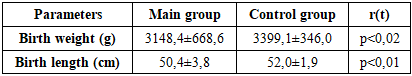
Growth and development in children with congenital heart disease were assessed using body weight, body length, and growth and developmental indices based on the reference growth curves recommended by the World Health Organization (WHO). In children with congenital heart defects, growth and development were assessed using body weight, body length, and growth-and-development indices based on the WHO-recommended growth curves.The majority of children with congenital heart defects—231 patients (78.7%)—had a birth weight above 2500 g, while 63 children (21.3%) had low birth weight (1500–2500 g). Low birth weight was observed only in the main study group, demonstrating a statistically significant difference between the main and control groups (21.3% vs. 0%, respectively; p < 0.001).Overall, the mean birth weight of children with congenital heart defects was significantly lower than that of children in the control group—3148.4 ± 668.6 g versus 3399.1 ± 346.0 g, respectively (p < 0.02) (Table 2).Table 2. Anthropometric parameters at birth
 |
| |
|
The mean birth weight of full-term children with congenital heart defects was 3323.3 ± 434.7 g, whereas in preterm infants this parameter was 2436.2 ± 184.9 g. In the main group, the mean body length was 50.4 ± 3.8 cm, which was significantly lower than that of children in the control group (52.0 ± 1.9 cm; p < 0.01). Among full-term children with congenital heart defects, body length averaged 52.0 ± 2.2 cm, while in preterm infants it was 46.7 ± 1.1 cm.In children with congenital heart defects, certain changes in the acid–base status and electrolyte balance of the blood were observed, and these parameters varied dynamically during different stages of surgical intervention.In children with cyanotic congenital heart defects, the blood pH level before surgery was 7.4 ± 0.3 and remained stable during cardiopulmonary bypass (CPB) and in the postoperative period (7.4 ± 0.4). This indicates a relatively compensated acid–base status.The partial pressure of carbon dioxide in the blood (pCO₂) was 34.1 ± 0.8 mmHg before surgery, increased to 36.7 ± 0.4 mmHg during CPB (p < 0.01), and reached 37.7 ± 0.4 mmHg in the postoperative period. These changes reflect functional alterations in the respiratory and gas exchange systems.The partial pressure of oxygen (pO₂) was 96.1 ± 2.8 mmHg before surgery and 99.1 ± 0.4 mmHg during cardiopulmonary bypass (CPB), after which it decreased to 85.6 ± 0.4 mmHg following CPB (p < 0.01) and subsequently increased to 98.2 ± 0.4 mmHg in the postoperative period. At the same time, oxygen saturation (SatO₂) remained high at all stages and reached 98.8 ± 0.4% after surgery.Blood glucose levels were 4.8 ± 0.1 mmol/L before surgery and increased significantly to 9.6 ± 0.4 mmol/L after CPB (p < 0.001), then decreased to 5.6 ± 0.4 mmol/L in the postoperative period. This indicates the development of stress-induced hyperglycemia.Bicarbonate (HCO₃⁻) and total CO₂ (TCO₂) levels remained relatively stable throughout all stages of surgery, indicating adequate metabolic compensation of the acid–base status. The base excess (BE) value also showed no marked deviations, decreasing to 0.7 ± 0.4 mmol/L after surgery.In the electrolyte analysis, sodium (Na⁺) levels remained within the normal range at all stages, whereas potassium (K⁺) levels decreased significantly during and after cardiopulmonary bypass (CPB), reaching 3.2 ± 0.4 mmol/L (p < 0.001). Calcium (Ca²⁺) levels decreased to 1.3 ± 0.4 mmol/L after CPB and were partially restored in the postoperative period (1.8 ± 0.4 mmol/L).In children with acyanotic congenital heart defects, changes in acid–base status and electrolyte parameters were less pronounced compared with the cyanotic group. In these patients, pH, pCO₂, and pO₂ values remained close to physiological norms throughout all stages of surgery, and no severe metabolic or respiratory acidosis was observed.Although a moderate increase in glucose levels was noted during CPB, a faster tendency toward normalization was observed in the postoperative period. Electrolyte analysis showed that alterations in potassium and calcium levels were less profound and compensated more rapidly than in the cyanotic CHD group.Overall, indicators of acid–base status and electrolyte balance in children with acyanotic congenital heart defects differed to some extent from those in the cyanotic group.The blood pH level before surgery was 7.4 ± 0.3 and slightly decreased to 7.3 ± 0.4 during cardiopulmonary bypass (CPB). After CPB, the pH returned to 7.4 ± 0.3, while in the postoperative period it was 7.35 ± 0.4, indicating a mild compensated respiratory–metabolic state.The partial pressure of carbon dioxide (pCO₂) was 34.1 ± 0.8 mmHg before surgery, increased to 37.5 ± 0.4 mmHg during CPB (p < 0.001), and reached 40.7 ± 0.4 mmHg after CPB (p < 0.001). In the postoperative period, pCO₂ was 36.4 ± 0.4 mmHg (p < 0.01), reflecting the presence of elements of hypoventilation.The partial pressure of oxygen (pO₂) before surgery was 96.1 ± 2.8 mmHg and remained at 96.8 ± 0.4 mmHg during cardiopulmonary bypass (CPB) (p < 0.001). After CPB, pO₂ decreased to 93.5 ± 0.4 mmHg (p < 0.001) and was restored to 97.4 ± 0.4 mmHg in the postoperative period.Oxygen saturation (SatO₂) was 98.5 ± 0.8% before surgery, 97.5 ± 0.4% during CPB, and decreased significantly to 92.6 ± 0.4% after CPB (p < 0.001), subsequently increasing to 97.7 ± 0.3% after surgery.Blood glucose levels were 4.8 ± 0.1 mmol/L before surgery, 4.0 ± 0.3 mmol/L during cardiopulmonary bypass (CPB), and increased sharply to 10.6 ± 0.4 mmol/L after CPB (p < 0.001). In the postoperative period, glucose levels decreased to 5.6 ± 0.4 mmol/L.Bicarbonate (HCO₃⁻) levels were 24.2 ± 0.8 mmol/L before surgery, 22.4 ± 0.3 mmol/L during CPB, 26.0 ± 0.4 mmol/L after CPB, and 23.4 ± 0.4 mmol/L in the postoperative period.Total CO₂ (TCO₂) levels were 23.5 ± 0.7 mmol/L before surgery, increased to 24.6 ± 0.4 mmol/L during CPB, rose significantly to 26.9 ± 0.3 mmol/L after CPB (p < 0.001), and measured 25.8 ± 0.4 mmol/L in the postoperative period (p < 0.05).The base excess (BE) level before surgery was 1.2 ± 0.01 mmol/L, increased to 1.7 ± 0.4 mmol/L during cardiopulmonary bypass (CPB), and then decreased significantly to −3.4 ± 0.4 mmol/L after CPB (p < 0.01). In the postoperative period, BE recovered to 1.8 ± 0.4 mmol/L.Electrolyte analysis showed that sodium (Na⁺) levels were 140.3 ± 4.4 mmol/L before surgery, 135.6 ± 0.4 mmol/L during CPB, 145.3 ± 0.4 mmol/L after CPB, and 138.5 ± 0.3 mmol/L in the postoperative period.Potassium (K⁺) levels were 5.2 ± 0.1 mmol/L before surgery, decreased significantly to 3.5 ± 0.4 mmol/L during CPB (p < 0.001), further declined to 3.0 ± 0.4 mmol/L after CPB (p < 0.001), and reached 3.8 ± 0.4 mmol/L in the postoperative period.Calcium (Ca²⁺) levels were 2.4 ± 0.1 mmol/L before surgery, increased to 3.2 ± 0.4 mmol/L during cardiopulmonary bypass (CPB), then decreased to 1.0 ± 0.3 mmol/L after CPB, and were restored to 2.7 ± 0.3 mmol/L in the postoperative period.It is noted that HIF-1α exerts both neuroprotective and neurotoxic effects following hypoxia–ischemia. These effects depend on the cell type and the severity of hypoxia. A deeper understanding of these complex functions of HIF-1α is of great importance for the development of protective therapeutic strategies in hypoxic–ischemic injury.The obtained results demonstrated that HIF-1α levels differed significantly between the two groups of patients.In patients of Group 1, the HIF-1α level before surgery was 7.2 ± 1.2 ng/mL (p < 0.001), during surgery it was 7.4 ± 0.8 ng/mL (p < 0.01), and on the 3rd postoperative day it measured 7.3 ± 0.9 ng/mL (p < 0.01).In patients of Group 2, HIF-1α levels were 9.9 ± 1.7 ng/mL before surgery (p < 0.001), increased to 11.7 ± 1.6 ng/mL during surgery (p < 0.001), and were 9.9 ± 0.9 ng/mL on the 3rd postoperative day (p < 0.001).In Group 1, HIF-1α levels increased 3.1-fold before surgery, 3.2-fold during surgery, and 3.1-fold after surgery. In Group 2, the corresponding increases were 4.3-fold, 5.0-fold, and 4.3-fold, respectively.Comparative analysis between the groups showed that in Group 1 patients, HIF-1α levels differed from those of Group 2 by 1.3-fold before surgery (p < 0.05), 1.58-fold during surgery (p < 0.05), and 1.35-fold after surgery (p < 0.05).In children with cyanotic congenital heart defects, chronic hypoxia led to activation of the HIF-1α signaling pathway, one of the key molecular mechanisms of the hypoxic response.In this group, preoperative HIF-1α levels were significantly higher than those in the control group, which was interpreted as a compensatory response to prolonged hypoxemia. During surgery, cardiopulmonary bypass (CPB) and transient impairment of tissue perfusion resulted in a further increase in HIF-1α expression.On the 3rd postoperative day, HIF-1α levels showed a slight downward trend; however, they remained significantly higher than those in the control group. This finding indicates that in children with cyanotic congenital heart defects, the molecular response to hypoxia is stable and prolonged in nature.Table 3. Dynamic changes in HIF-1α levels (ng/mL) in children with congenital heart defects
 |
| |
|
4. Conclusions
Among patients in Group 1, the most common forms of congenital heart defects were tetralogy of Fallot, transposition of the great arteries, and the complete form of atrioventricular canal defect. The study results demonstrate that the majority of defects are diagnosed within the first year of life, confirming the critical importance of early diagnosis and the organization of targeted, specialized dispensary follow-up for this category of children.The obtained data indicate that in children with acyanotic congenital heart defects, alterations in acid–base status and electrolyte parameters are less pronounced than in the cyanotic group and, in most cases, are compensated more rapidly after surgery. However, the presence of significant changes in gas exchange, electrolyte balance, and glucose levels during the cardiopulmonary bypass stage highlights the need for regular monitoring even in these patients.In children with acyanotic congenital heart defects, activation of HIF-1α is primarily associated with surgical stress and hemodynamic changes, while persistent molecular remodeling related to chronic hypoxia is less evident. Significant differences in HIF-1α expression were identified between cyanotic and acyanotic congenital heart defects. In children with cyanotic CHDs, HIF-1α levels were higher at all observation stages, manifesting as a key molecular marker of adaptation to chronic hypoxia. In contrast, in children with acyanotic CHDs, changes in HIF-1α were mainly limited to transient hypoxic states associated with surgical intervention.These findings suggest that HIF-1α may serve as a promising biomarker for assessing the severity of hypoxia and adaptive potential in cyanotic congenital heart defects and may play an important role in the development of individualized management strategies.
References
| [1] | Shumakova O.V., Morozova N.Ya., Burov A.A., Podurovskaya Yu.L., Bokeria E.L., Nefedova I.E., Berishvili D.O., Degtyarev D.N. Features of hemostasis in children with congenital heart defects during the first 12 hours of life. Problems of Hematology/Oncology and Immunopathology in Pediatrics. 2019; 18(3): 29–34. |
| [2] | Dahaba AA. Thinking outside the box. Off-label use of Bispectral Index within context and limitations for conditions other than depth of anesthesia. Minerva Anestesiol. 2019 Feb; 85(2): 189-193. |
| [3] | Tsepokina A.V., Khutornaya M.V., Shabaldin A.V., et al. Distribution patterns of genotypes of polymorphic variants rs2234246 and rs4711668 of TREM-1 in children with ductus-dependent congenital heart defects. Translational Medicine. 2019; 6(4): 5–12. |
| [4] | Stepanova A.I., Alekhin M.N. Capabilities and limitations of speckle-tracking stress echocardiography. Siberian Journal of Clinical and Experimental Medicine. 2019; 34(1): 10–17. |
| [5] | Tairova S.B., Mukhtorov A.A., Ziyodullaeva M.S. Neurocognitive disorders in children with congenital heart defects (a literature review). Scientific Education and Practice. 2024; 3(28): 174–179. |
| [6] | Sprong M.C.A., van Brussel M., de Vries L.S., et al. Longitudinal Motor Developmental Outcomes in Infants with a Critical Congenital Heart Defect // Children (Basel). 2022. Vol. 9, No. 4. P. 570. doi: 10.3390/children 9040570. |
| [7] | Tairova S.B., Khushvaktova B.B. Clinical features of comorbid pathology in children with congenital septal heart defects (a literature review). Scientific Education and Practice. 2024; 3(28): 169–173. |
| [8] | Tokmakova K.A., Shilkin D.N., Kupryashov A.A. Hemodynamic activation of von Willebrand factor in children with congenital heart defects. Obstetrics, Gynecology and Reproduction. 2024; 18(5): 693–705. |
| [9] | Favier F.B., Britto F.A., Freyssenet D.G., Bigard X.A. HIF-1-guided adaptation of skeletal muscle to chronic hypoxia: a molecular understanding of muscle physiology. Cell. Like. Life Sci. 2015; V.72. P. 4681-4696. |
| [10] | Gregory M.B., Prouhet P.M., Russel C.L., Pfannenstiel B.R. Quality of Life for Parents of Children with Congenital Heart Defect: a systematic review. J Cardiovasc Nurs 2018; 33(4); 363-371. |
| [11] | Liu Y., Chen S., Zühlke L., et al. Global birth prevalence of congenital heart defects 1970–2017: Updated systematic review and meta-analysis of 260 studies // Int. J. Epidemiol. 2019. Vol. 48, No. 2. P. 455–463. doi: 10.1093/ije/dyz009. |
| [12] | Messerli–Bürgy N., Kakebeeke T.H., Meyer A.H., et al. Walking onset: a poor predictor for motor and cognitive skills in healthy preschool children // BMC Pediatr. 2021. Vol. 21, No. 1. P. 367. doi: 10.1186/s12887-021-02828-4. |





 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML

