-
Paper Information
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(3): 1551-1554
doi:10.5923/j.ajmms.20261603.148
Received: Mar. 3, 2026; Accepted: Mar. 28, 2026; Published: Mar. 31, 2026

Expression of Heat Shock Proteins HSP70 and HSP90 in Adrenal Tissues Under Acute and Chronic Stress Exposure
Ro`ziqulova Farangis Umarjon qizi1, Yariyev Alisher Alijonovich2, Xolmatova Charos Akramovna1
1Termez Branch of Tashkent State Medical University, Uzbekistan
2Syrdarya Branch of the Republican Scientific Center for Emergency Medical Care, Uzbekistan
Correspondence to: Ro`ziqulova Farangis Umarjon qizi, Termez Branch of Tashkent State Medical University, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

This experimental study investigated the expression of heat shock proteins HSP70 and HSP90 in rat adrenal tissues under conditions of acute and chronic stress exposure. Stress was modeled using the forced swimming test for 4 and 10 days. The results demonstrated that HSP70 expression significantly increased as early as day 4 of stress exposure and remained elevated on day 10, indicating the formation of a stable cellular adaptive response. In contrast, HSP90 expression showed pronounced upregulation during the early phase of stress exposure, followed by partial reduction during prolonged stress. The obtained findings indicate the phase-dependent nature of the cellular stress response and confirm the important role of heat shock proteins in the mechanisms of adrenal adaptation to stress.
Keywords: Stress, General adaptation syndrome, HSP70, HSP90, Heat shock proteins, Adrenal glands, Experiment, Western blot, Adaptation
Cite this paper: Ro`ziqulova Farangis Umarjon qizi, Yariyev Alisher Alijonovich, Xolmatova Charos Akramovna, Expression of Heat Shock Proteins HSP70 and HSP90 in Adrenal Tissues Under Acute and Chronic Stress Exposure, American Journal of Medicine and Medical Sciences, Vol. 16 No. 3, 2026, pp. 1551-1554. doi: 10.5923/j.ajmms.20261603.148.
1. Introduction
- General Adaptation Syndrome (GAS) represents a universal physiological response of the organism to a wide spectrum of stressors. It begins with the activation of nonspecific adaptive mechanisms and may culminate in impaired functional stability under prolonged stress exposure [1,2,6]. In contemporary science, GAS is considered an integrative framework that unites neuroendocrine, immune, and behavioral responses, with a central role attributed to the hypothalamic–pituitary–adrenal (HPA) axis. The HPA axis ensures the mobilization of energy resources, regulates homeostatic balance, and determines individual adaptive capacity during stress signal activation [3,7].Recent studies emphasize that acute stress may exert adaptive effects, enhancing resistance to subsequent challenges. However, prolonged or repetitive stress exposure leads to HPA axis dysregulation, impaired cortisol regulation, and the development of pathological conditions [9]. This transition from adaptation to exhaustion is consistent with the classical three-phase model of GAS—alarm, resistance, and exhaustion—and reflects mechanistic alterations underlying multisystem pathological responses [6].The practical significance of clarifying the mechanisms of GAS is обусловлена the high prevalence of chronic stress in modern society, which is associated with an increased risk of psychosomatic disorders, depressive conditions, and somatic diseases, including cardiometabolic disturbances [8]. Understanding the biological correlates of the adaptive response and the mechanisms underlying the shift toward pathological states represents an essential step toward the identification of diagnostic biomarkers, outcome prediction, and the development of targeted interventions aimed at preventing stress-associated disorders. Therefore, the study of GAS remains highly relevant and warrants further in-depth interdisciplinary investigation through 2026 and beyond.
2. Materials and Methods
- The study was conducted in accordance with generally accepted experimental protocols at a center accredited to perform research in compliance with Good Laboratory Practice (GLP) standards [4,5]. A total of 30 male rats weighing 180–240 g were included in the experiment and randomly assigned to three groups (n = 10 per group). All animals underwent at least one week of acclimatization prior to the initiation of the experimental procedures.The animals were distributed as follows:• Control group (n = 10): no stress exposure.• Acute stress group (n = 10): animals were subjected to forced swimming for four consecutive days.• Chronic stress group (n = 10): animals underwent the same procedure for ten consecutive days.Stress Induction ProtocolStress was induced using the forced swimming test. Each animal was placed individually in a transparent cylindrical tank (40 cm in height, 10 cm in diameter) filled with water to a depth of 30 cm. Water temperature was maintained at 20–23°C. Each session lasted 10 minutes. After completion of the procedure, animals were gently dried and returned to their cages [10].Adrenal gland tissues were collected on days 4 and 10 for subsequent Western blot analysis.Western Blot AnalysisAdrenal tissue samples (20–100 mg) were homogenized in 5–10 volumes of lysis buffer supplemented with protease and phosphatase inhibitors. The homogenates were incubated on ice for 30 minutes and then centrifuged at 12,000 rpm for 30 minutes at 4°C. The supernatant was collected, and protein concentration was determined using the Bradford assay.Proteins were denatured in 1× Laemmli buffer at 100°C for 10 minutes, cooled, briefly centrifuged, and stored at −20°C until analysis.Electrophoresis. Samples were separated by SDS–PAGE. Gels were placed into the electrophoresis chamber filled with 1× running buffer. Four microliters of molecular weight marker were loaded into the first well, followed by 10 µL of each protein sample into subsequent wells. Electrophoresis was performed at 60–80 V for 10 minutes (stacking gel), followed by 100 V for approximately 1 hour.Protein Transfer (Blotting). Polyvinylidene fluoride (PVDF) membranes were activated in 100% methanol for 1 minute and equilibrated in cold 1× transfer buffer (4°C) for 10 minutes. The transfer sandwich was assembled in the following order: sponge – filter paper – gel – membrane – filter paper – sponge. Protein transfer was performed at 15 V for 1 hour and 10 minutes.Blocking and Antibody Incubation. Following transfer, membranes were incubated overnight at 4°C with gentle agitation in blocking buffer (5% BSA in TBST). Membranes were then incubated overnight at 4°C with primary antibodies diluted in the same buffer (5% BSA/TBST). After three washes in 1× TBST (10 minutes each), membranes were incubated with appropriate HRP-conjugated secondary antibodies overnight at 4°C. Subsequently, membranes were washed three times in TBST.Detection. Protein bands were detected using enhanced chemiluminescence (ECL) reagents (1:1 mixture of reagents A and B). Membranes were incubated with the substrate for 1–5 minutes, and signals were visualized using a chemiluminescent imaging system. Quantitative analysis of band intensity was performed using appropriate image analysis software.
3. Results
- To evaluate the cellular stress response in rats subjected to experimental stress, the expression levels of heat shock proteins HSP70 and HSP90 were assessed using Western blot analysis.As shown in Figure 1, compared with the control group, HSP70 expression was markedly increased already on day 4 in all three samples and remained elevated on day 10. A moderate inter-lane variability was observed. The β-actin bands were generally comparable across samples, allowing qualitative comparison; however, accurate quantitative evaluation requires normalization to β-actin.
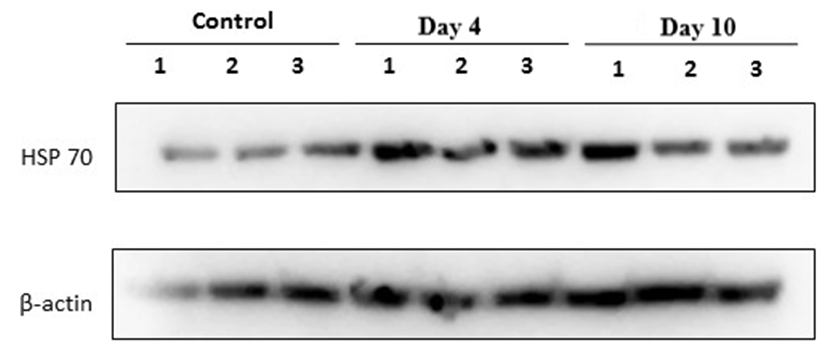 | Figure 1. Western blot analysis of heat shock protein HSP70 and the loading control β-actin in rat adrenal gland tissues in a forced swimming–induced stress model |
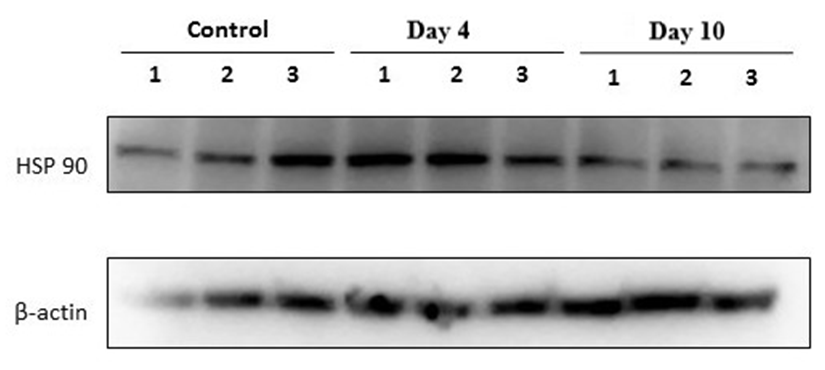 | Figure 2. Western blot analysis of heat shock protein HSP90 and the loading control β-actin in rat adrenal gland tissues in a forced swimming–induced stress model |
4. Discussion
- The obtained results demonstrated that HSP70 expression in adrenal tissues increased as early as after four days of forced swimming and remained elevated after ten days of exposure. This finding reflects the activation of stress-induced molecular defense mechanisms aimed at maintaining protein structural stability and preventing denaturation under prolonged stress conditions. The sustained upregulation of HSP70 suggests its pivotal role as a key regulator of endocrine tissue adaptation to chronic stress.In contrast to HSP70, HSP90 exhibited a distinct expression pattern. A marked increase in HSP90 levels was observed after four days of stress in all samples, which may indicate its involvement in the regulation and stabilization of glucocorticoid receptors within the adrenal cortex. Given that HSP90 functions as an essential molecular chaperone for steroid hormone receptors, its early upregulation likely reflects activation of the hypothalamic–pituitary–adrenal (HPA) axis during the initial adaptive phase. However, by day 10, HSP90 expression partially declined toward control levels. This reduction may be associated with cellular adaptation to sustained stress exposure and a decreased requirement for intensive molecular chaperone activity.Thus, HSP70 appears to mediate a sustained protective response under chronic stress conditions, whereas HSP90 is predominantly involved in the early phase of adaptation. The coordinated activation of both proteins underscores the complex and phase-dependent nature of adrenal cellular defense mechanisms during prolonged stress exposure.
5. Conclusions
- 1. Stress modeling using the forced swimming paradigm results in a significant increase in HSP70 expression in rat adrenal tissues as early as day 4, with sustained elevation observed on day 10, indicating the formation of a stable cellular adaptive response.2. HSP90 expression is characterized by marked upregulation during the early phase of stress exposure (day 4), followed by partial reduction by day 10, reflecting its predominant involvement in the initial stage of adaptation and in the regulation of glucocorticoid receptor stability.3. The differential expression dynamics of HSP70 and HSP90 confirm the phase-dependent nature of the General Adaptation Syndrome and highlight the coordinated yet functionally distinct roles of chaperone proteins in the mechanisms of stress-induced adrenal adaptation.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML