-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(3): 1548-1550
doi:10.5923/j.ajmms.20261603.147
Received: Feb. 22, 2026; Accepted: Mar. 16, 2026; Published: Mar. 31, 2026

Scientific Substantiation of the Quality Assessment of the Laboratory and Diagnostic Service of the Khorezm Region
Nurmetova Yulduz Baltayevna
Department of Military Field Therapy, Hematology and Diagnostics, Urgench State Medical Institute, Khorezm, Uzbekistan
Correspondence to: Nurmetova Yulduz Baltayevna, Department of Military Field Therapy, Hematology and Diagnostics, Urgench State Medical Institute, Khorezm, Uzbekistan.
| Email: | 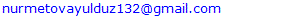 |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

In the context of reforming the healthcare system of Uzbekistan, which provides for the creation of 198 centralized laboratories by 2030, the development of regional models for assessing the quality of laboratory services is becoming particularly relevant. The article presents a scientific justification for a comprehensive system for assessing the quality of the laboratory diagnostic service of the Khorezm region based on international standards (ISO 15189), quality indicators, and the methodology for analyzing the pre-analytical, analytical, and post-analytical phases of the laboratory process. The proposed model includes 24 key quality indicators adapted to regional conditions, and an external quality assessment mechanism (EFCA) using the Six Sigma scale for quantitative efficiency assessment.
Keywords: Laboratory diagnostics, Quality management, Quality indicators, ISO 15189, Khorezm region, External quality assessment, Pre-analytical phase
Cite this paper: Nurmetova Yulduz Baltayevna, Scientific Substantiation of the Quality Assessment of the Laboratory and Diagnostic Service of the Khorezm Region, American Journal of Medicine and Medical Sciences, Vol. 16 No. 3, 2026, pp. 1548-1550. doi: 10.5923/j.ajmms.20261603.147.
1. Introduction
- Laboratory diagnostics is one of the key components of the modern healthcare system, providing up to 60-70% of objective information necessary for diagnosing, choosing treatment tactics, and evaluating the effectiveness of therapy [1,2]. The quality of laboratory tests directly affects the patient's safety, clinical effectiveness, and economic feasibility of medical care [3]. In the Republic of Uzbekistan, within the framework of the implementation of the Healthcare Development Strategy for 2021-2025 and subsequent stages of reform (including Presidential Decree No. PP-311 dated 05.09.2024 and measures to create 198 centralized inter-district laboratories by 2030), special attention is paid to the modernization of laboratory diagnostic services (LDS), the transition to centralized models, and the introduction of unified quality management standards [4,5]. The Khorezm region is characterized by a number of specific features that significantly affect the organization and quality of laboratory diagnostics: - a significant share of the rural population (about 75%) and the concentration of medical institutions; - high workload on district laboratories in conditions of limited access to modern equipment and reagents;- climatic factors (high temperature and humidity in the summer period), increasing the risk of pre-analytical errors during the transportation of biomaterial; - persisting fragmentation of the laboratory network, especially in the northern districts of the region;- Shovot, Khankin (Khonka), Gurlen, and Yangiarik.Despite positive trends (implementation of laboratory information systems in a number of quality assessment departments, participation in external quality assessment programs), significant problems remain in the indicated districts: - high percentage of pre-analytical defects (from 2.5 to 4.5% depending on the district); - insufficient participation in external quality assessment programs (EFA) - from 68% to 92% in 2024-2025; - uneven equipment and personnel training; - lack of a unified regional quality indicator monitoring system. International standards (primarily ISO 15189:2022 "Medical Laboratories. Quality and Competence Requirements") and WHO recommendations emphasize the need for a comprehensive approach to quality management covering all aspects of the laboratory process - pre-analytical, analytical, and post-analytical [6,7]. At the same time, regional adapted quality assessment systems that take into account local logistics, personnel, and climate characteristics are gaining special value.Research objectivesDevelop and scientifically substantiate a system for assessing the quality of the laboratory diagnostic service of the Khorezm region based on international standards and quality indicators adapted to regional conditions.
2. Materials and Methods
- The quality assessment system is developed based on: international standard ISO 15189:2022 "Medical Laboratories.Requirements for quality and competence"; WHO recommendations on the laboratory quality management system; the concept of a complete laboratory process (Total Testing Process) with an emphasis on the pre-analytical (up to 68% of errors), analytical, and post-analytical phases.Research methodsØ Adaptation of IFCC WG-LEPS quality indicators;Ø External Quality Assessment (QQA) through interlaboratory comparison;Ø Calculation is sigmo-metric (σ = (TEa - Bias) / CV);Ø Descriptive Statistics and Trend Analysis (Excel, SPSS).Object and scope of research. Analysis of the work of 12 clinical and diagnostic laboratories of the Khorezm region (including CROs of the Shavatsky, Khankinsky, Gurlensky, Yangiaryk districts) for 2024-2025. Data obtained by:- audit according to the checklist ISO 15189;- monthly monitoring of 24 quality indicators;- results of participation in VOC programs;- analysis of reports from the regional health department. Observation period
 January 3, 2024 - December 2025. The study was conducted in accordance with the requirements of the reform of the laboratory service of Uzbekistan (PP-311 dated 05.09.2024 and others) and national adaptations of quality standards.
January 3, 2024 - December 2025. The study was conducted in accordance with the requirements of the reform of the laboratory service of Uzbekistan (PP-311 dated 05.09.2024 and others) and national adaptations of quality standards.3. Results and Discussion
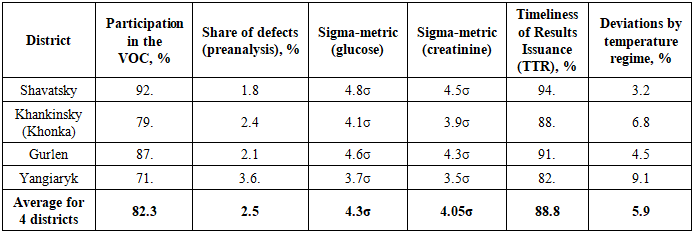
- Results of the assessment of the quality of laboratory diagnostic services in the Shavatsky, Khankinsky, Gurlensky, and Yangiaryk districts (2024-2025).
|
|
4. Conclusions
- 1. A quality assessment system for the laboratory diagnostic service of the Khorezm region, integrating the requirements of the ISO 15189 standard, quality indicators by phases of the laboratory process, and a methodology for quantitative assessment through sigma metrics, has been scientifically substantiated.2. The proposed model of 24 indicators allows for the identification of bottlenecks in laboratory work, especially in the pre-analytical phase, where up to 68% of errors are concentrated. 3. The system is adapted to the regional conditions of the Khorezm region and can serve as a basis for creating a unified digital register of the quality of laboratory services within the framework of the national reform of the laboratory service of Uzbekistan.4. For successful implementation, it is necessary: to create a regional center for external quality assessment on the basis of the Republican Emergency Medical Center in the city of Urgench, to train personnel according to pre-analytics standards, and to implement a unified information system for monitoring indicators.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML