-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(3): 1539-1544
doi:10.5923/j.ajmms.20261603.145
Received: Feb. 1, 2026; Accepted: Feb. 22, 2026; Published: Mar. 31, 2026

Immunohistochemical Characteristics of Various Forms of Adenoids in Children
Shoyatbek Rustamjonovich Khusanov1, Bahodir Salijonovich Mamajonov2
1Independent Researcher, Andijan State Medical Institute, Andijan, Uzbekistan
2DSc, Doctor of Medical Sciences, Andijan State Medical Institute, Andijan, Uzbekistan
Correspondence to: Shoyatbek Rustamjonovich Khusanov, Independent Researcher, Andijan State Medical Institute, Andijan, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

The sclerotic hypertrophic form of the adenoid is predominantly observed in older adolescents with chronic adenoiditis. This condition is characterized by pronounced proliferation of connective tissue originating from the base of the pharyngeal lymphoid tissue, particularly in the region adjacent to the palatine bone, and extending toward the periosteum. The connective tissue bundles display a hypocellular, coarse fibrous architecture and are arranged as thick strands extending from the basal layer of the adenoid to the superficial layers. In addition, these fibrotic changes are accompanied by structural remodeling of the lymphoid component, including partial replacement of lymphoid follicles and disruption of the mucosa-associated lymphoid tissue (MALT) organization. Vascular alterations, such as angiomatous changes, along with signs of interstitial proliferative inflammation, are frequently observed, reflecting long-standing chronic inflammatory stimulation. Collectively, these morphological features indicate progressive fibrosis and reduced immunological functionality of the adenoid tissue in chronic disease states.
Keywords: Interstitial proliferative inflammation, Angiomatous changes, Hyperplasia, Mucosa-associated lymphoid tissue (MALT), Lymphoid follicle
Cite this paper: Shoyatbek Rustamjonovich Khusanov, Bahodir Salijonovich Mamajonov, Immunohistochemical Characteristics of Various Forms of Adenoids in Children, American Journal of Medicine and Medical Sciences, Vol. 16 No. 3, 2026, pp. 1539-1544. doi: 10.5923/j.ajmms.20261603.145.
1. Introduction
- Adenoid hypertrophy and chronic adenoiditis are among the most prevalent otorhinolaryngological conditions in pediatric populations and pose a significant public health concern due to their high frequency and potential complications. Epidemiological studies indicate that adenoid hypertrophy affects a substantial proportion of children, with prevalence estimates ranging from 20% to over 60%, depending on age, diagnostic criteria, and environmental factors [1]. The peak incidence is observed between 3 and 7 years of age, corresponding to the period when adenoid tissue reaches its maximal size prior to physiological involution during adolescence [2,3].Clinically, children with adenoid hypertrophy frequently present with nasal obstruction, mouth breathing, snoring, and recurrent upper respiratory tract infections, which may lead to sleep-disordered breathing and otitis media with effusion [4,5]. These symptoms not only impair daily functioning and sleep quality but may also influence craniofacial development and growth patterns [6]. Allergic conditions, particularly allergic rhinitis, are commonly associated with increased severity of adenoid hypertrophy, highlighting the interplay between immune sensitization and lymphoid tissue response [7].Environmental factors, including exposure to air pollution, passive smoking, and aeroallergens, have been linked to a higher risk and earlier onset of adenoid hypertrophy, emphasizing the role of exogenous factors in disease progression [8,9]. Immunologically, the adenoid functions as part of the mucosa-associated lymphoid tissue (MALT), maintaining local immune surveillance and inducing antigen-specific adaptive immune responses. Chronic antigenic stimulation, recurrent infections, or immune dysregulation can result in structural remodeling, fibrosis, and altered lymphocyte populations, compromising its immunological function [10,11].Constitutional and genetic factors, such as lymphatic-hypoplastic diathesis and features of thymicolymphatic status, can also contribute to adenoid hypertrophy independent of active inflammation, often leading to secondary immunodeficiency and reduced responsiveness to conservative therapy [12]. These multifactorial mechanisms underscore the need for comprehensive morphological and immunohistochemical studies to optimize diagnosis and treatment strategies in pediatric otolaryngology.
2. Materials and Methods
- This study was conducted between 2020 and 2024 at the ADTI Clinic and the IMPULS Private Clinic. A total of 64 children, aged 1 to 10 years, diagnosed with advanced adenoid disease, were included.The immunological characteristics of adenoid tissue were evaluated using immunohistochemical methods, focusing on the expression patterns of immune markers within the nasopharyngeal tonsils. Clinical and anamnesis data were collected to assess the age-related prevalence and immunological features of adenoid enlargement. Statistical analyses were performed to compare findings across age groups and to determine the significance of observed immunohistochemical patterns.
3. Results and Discussion
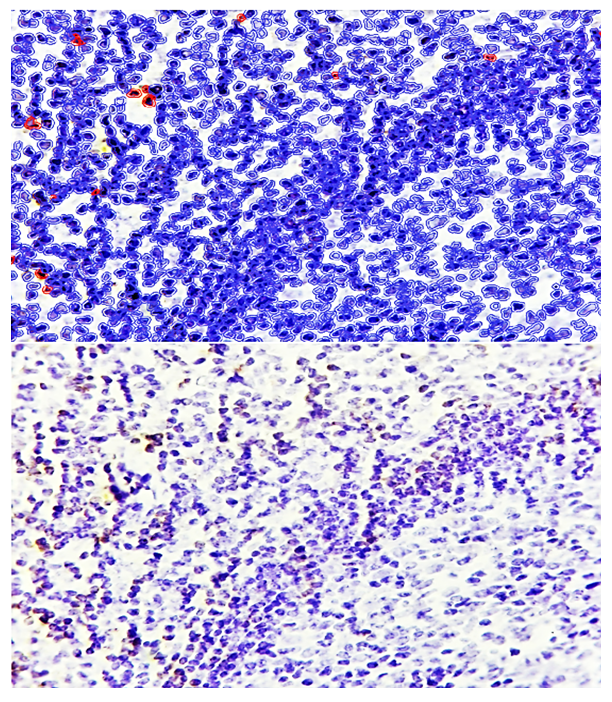
- The Bcl-2 protein, located on human chromosome 18, is a homolog of six anti-apoptotic proteins that inhibit the apoptotic process. With a molecular weight of approximately 22 kDa, Bcl-2 is localized in the cell and nuclear membranes, the sarcoplasm, and the mitochondrial membrane. High expression of Bcl-2 has been shown to block calcium ion release, slow lipid peroxidation, reduce antioxidant activity, and suppress nitric oxide synthase (NOS) activity. The primary function of Bcl-2 is to prevent the release of mitochondrial apoptotic molecules such as cytochrome c, AIF, and ATP through mitochondrial pores. By binding to the mitochondrial membrane, Bcl-2 closes these pores, inhibiting pro-apoptotic signaling and preventing apoptosis.Morphological examination of adenoid tissue in children with adenoid hypertrophy revealed several histotopographic forms, indicating structural heterogeneity within the lymphoid tissue. Based on these observations, immunohistochemical studies were performed to evaluate the expression of Bcl-2 across different histotopographic patterns of adenoid tissue, allowing a comparative analysis of molecular and morphological features.In the lymphoproliferative hypertrophy form of adenoid tissue, immunohistochemical analysis demonstrated that Bcl-2 expression was largely absent in the germinal centers of lymphoid follicles, whereas low-level expression was observed in the B-cell mantle zone (Figure 1). This finding suggests that the anti-apoptotic activity of Bcl-2 is largely suppressed within the germinal centers, potentially facilitating apoptosis in these regions, while the mantle zone retains limited anti-apoptotic protection. Such a distribution may reflect a mechanism by which lymphoid tissue balances proliferation and programmed cell death, maintaining immune homeostasis within hypertrophic adenoids.These results highlight the importance of spatial regulation of apoptotic markers in the pathogenesis of adenoid hypertrophy. Differential Bcl-2 expression across follicular compartments may contribute to selective survival of B-cell populations and influence the progression of lymphoproliferative forms of the disease. Further studies are warranted to correlate these molecular findings with clinical severity and potential response to therapy, providing a more comprehensive understanding of adenoid pathophysiology in pediatric patients (see Figure 1).
 | Figure 1. Lymphoproliferative form of adenoid tissue, showing low-level expression of the Bcl-2 marker around the lymphoid follicles. Staining: DAB chromogen. Magnification: 10×40 |
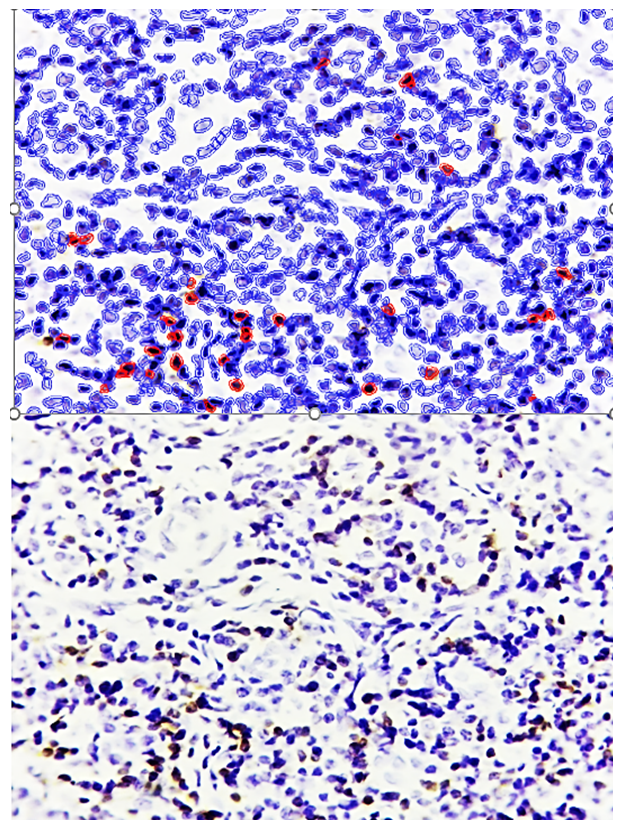 | Figure 3. Interstitial proliferative inflammatory hypertrophy form of adenoid tissue, showing Bcl-2 expression in certain cells within the infiltrate. Staining: DAB chromogen. Magnification: 10×40 |
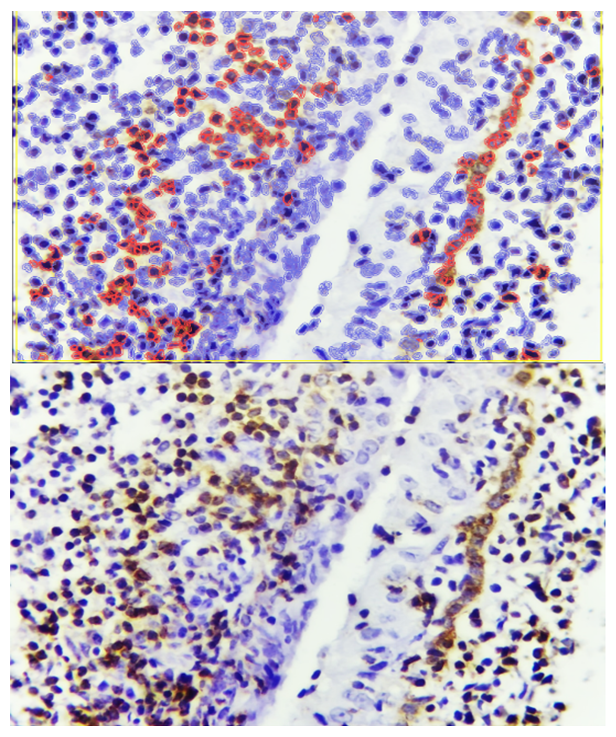 | Figure 4. Interstitial proliferative inflammatory form of adenoid tissue, showing stronger Bcl-2 expression around the covering epithelium. Staining: DAB chromogen. Magnification: 10×40 |
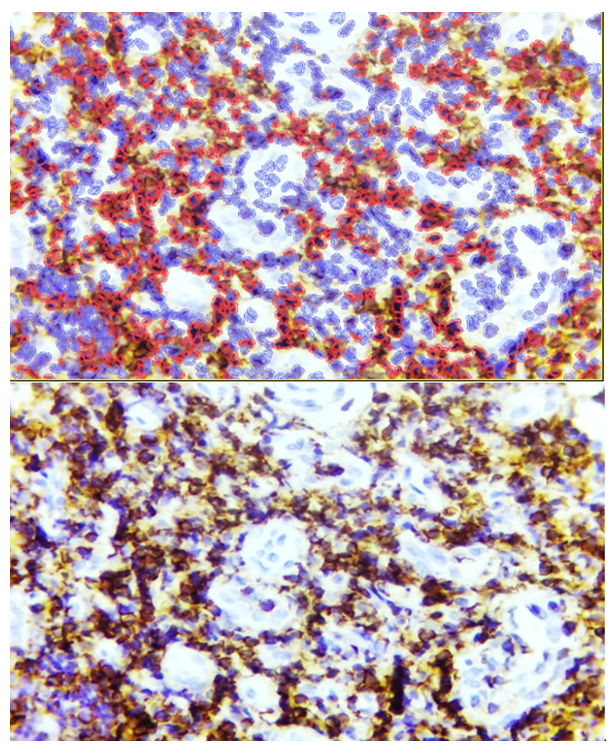 | Figure 5. Angiomatoid hypertrophy form of adenoid tissue, showing strong Bcl-2 expression in cells located between blood vessels. Staining: DAB chromogen. Magnification: 10×40 |
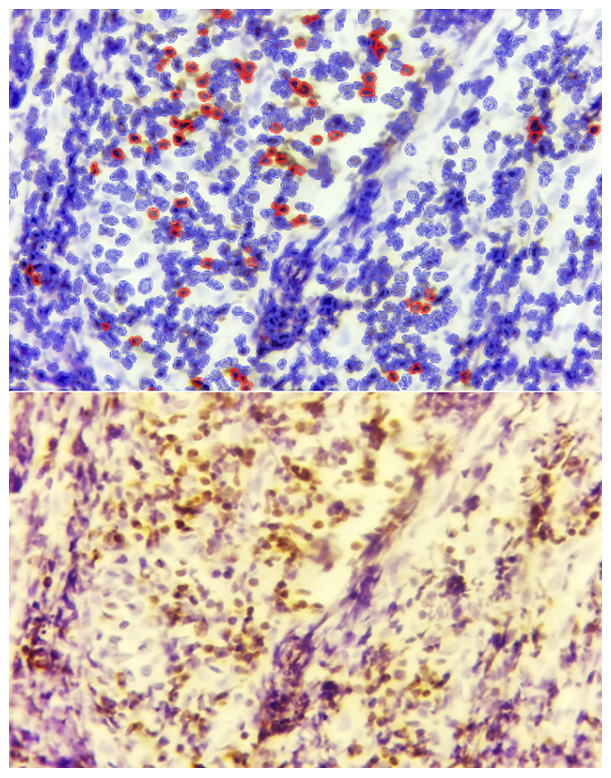 | Figure 6. Sclerosed hypertrophy form of adenoid tissue, showing moderate positive Bcl-2 expression in lymphoid cells within the sclerotic bundles. Staining: DAB chromogen. Magnification: 10×40 |
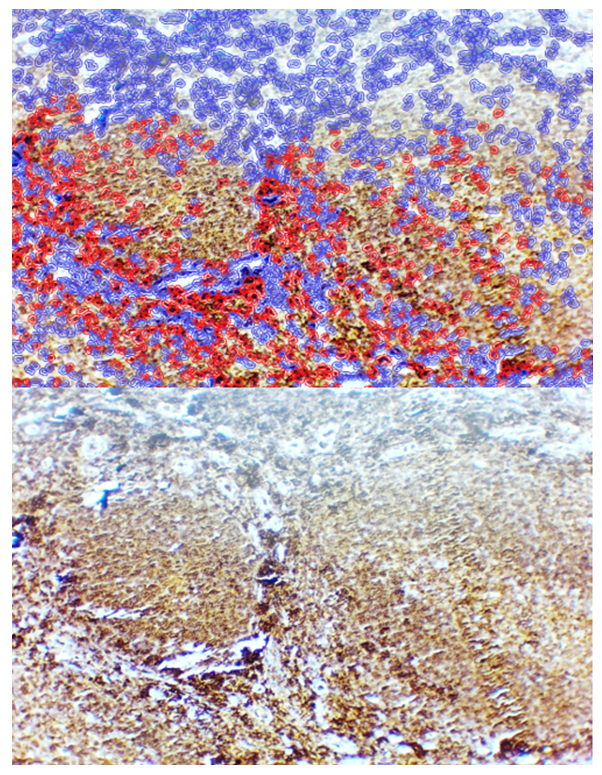 | Figure 7. Lymphofollicular hypertrophy form of adenoid tissue, showing CD45 expression throughout all tissue compartments. Staining: DAB chromogen. Magnification: 10×40 |
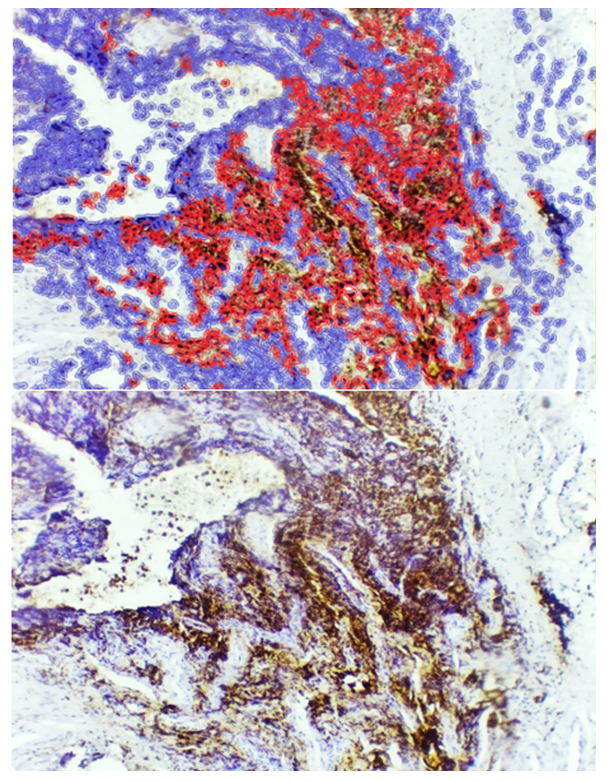 | Figure 8. Sclerosed hypertrophy form of adenoid tissue, showing low-level CD45 expression within the sclerotic bundles. Staining: DAB chromogen. Magnification: 10×40 |
4. Conclusions
- The present study comprehensively analyzed the expression patterns of Bcl-2, an anti-apoptotic protein, and CD45, a pan-leukocyte marker, across different histotopographic forms of adenoid tissue in children. The findings indicate that the level and localization of these markers vary significantly depending on the structural and pathological characteristics of the adenoid.Specifically, Bcl-2 expression was observed to be low in lymphofollicular hypertrophy, moderate in interstitial proliferative inflammatory forms, high in angiomatoid hypertrophy, and moderate in sclerosed hypertrophy. These differences reflect the varying degrees of anti-apoptotic activity within lymphoid, reticular, and stromal compartments. In particular, strong Bcl-2 expression was consistently noted in reticular stromal cells and lymphoid cells surrounding proliferating blood vessels in the angiomatoid form, whereas in sclerosed adenoids, Bcl-2 expression was restricted and heterogeneous due to fibrosis and tissue atrophy.Similarly, CD45 expression demonstrated form-specific distribution. In lymphofollicular hypertrophy, CD45 was positively expressed throughout all tissue compartments, indicating active presence of lymphocytes, monocytes, and granulocytes. In interstitial proliferative forms, expression was scattered among lymphoid and histiocytic cells, while in angiomatoid hypertrophy, CD45 positivity was detected in lymphoid and monocyte-macrophage cells surrounding proliferating vessels, but absent in endothelial and pericyte cells. In the sclerosed form, residual lymphoid and monocyte cells showed low-level CD45 expression, particularly in fibrotic areas. Quantitative morphometric analysis confirmed that CD45 expression was highest in the angiomatous form (36.7%), moderate in interstitial forms (24.2%), and lowest in the sclerosed form (16.5%).Overall, these findings demonstrate that both Bcl-2 and CD45 exhibit distinct, form-dependent expression patterns, reflecting the balance between apoptosis regulation, immune cell infiltration, and tissue remodeling in adenoid hypertrophy. The results provide valuable insight into the pathophysiological heterogeneity of adenoid disease, highlighting the importance of immunohistochemical profiling in understanding disease progression and potential therapeutic targeting of apoptotic and immune pathways.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML