-
Paper Information
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(3): 1471-1475
doi:10.5923/j.ajmms.20261603.130
Received: Feb. 15, 2026; Accepted: Mar. 9, 2026; Published: Mar. 27, 2026

Immunological Profile Features in Patients with Bronchial Asthma Comorbid with Type 2 Diabetes Mellitus
Ubaydullaeva N. N.1, Khaidarova F. A.2, Ismatullaeva S. S.2
1Center for the Development of Professional Qualifications of Medical Workers, Tashkent, Uzbekistan
2Republican Specialized Scientific-Practical Medical Center of Endocrinology named after Academic Yo.Kh. Turakulov, Tashkent, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

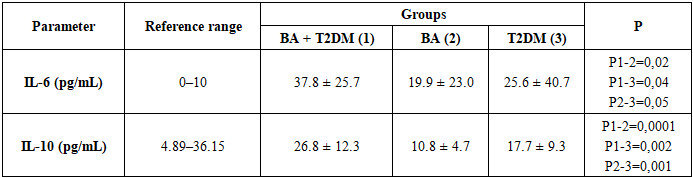
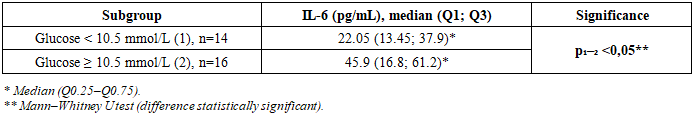
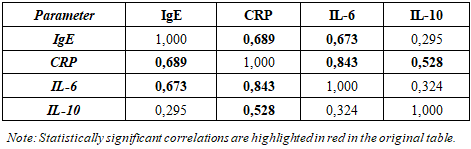
Aim. To assess the characteristics of the cytokine profile (interleukin-6 [IL-6], interleukin-10 [IL-10]) in patients with bronchial asthma (BA), type 2 diabetes mellitus (T2DM), and their comorbidity, and to determine the associations of cytokine levels with parameters of carbohydrate and lipid metabolism, leptin, inflammatory markers, and pulmonary function. Materials and methods. A total of 90 inpatients were examined and allocated to three sex- and age-matched groups (n=30 each): BA+T2DM, isolated BA, and isolated T2DM. Clinical and laboratory assessments included fasting glucose, glycated hemoglobin (HbA1c), insulin, leptin, lipid profile, immunoglobulin E (IgE), C-reactive protein (CRP), and spirometry. Serum IL-6 and IL-10 concentrations were measured by enzyme-linked immunosorbent assay (ELISA). Statistical analyses were performed using Statistica 8.0; differences were considered significant at p<0.05. Results. All groups demonstrated elevated IL-6 levels relative to reference values. The highest IL-6 concentrations were observed in the BA+T2DM group (37.8±25.7 pg/mL), which was significantly higher than in isolated BA (19.9±23.0 pg/mL; p=0.02) and isolated T2DM (25.6±40.7 pg/mL; p=0.04). IL-10 levels were also maximal in the BA+T2DM group (26.8±12.3 pg/mL) and significantly exceeded those in isolated BA (10.8±4.7 pg/mL; p=0.0001) and isolated T2DM (17.7±9.3 pg/mL; p=0.002). In patients with BA+T2DM, IL-6 was higher at glucose ≥10.5 mmol/L compared with glucose <10.5 mmol/L (median 45.9 vs 22.05 pg/mL; p<0.05). Leptin showed positive correlations with IL-6 (r=0.703) and IL-10 (r=0.434), correlated positively with atherogenic lipid parameters, and correlated negatively with high-density lipoprotein cholesterol (HDL-C) (r=−0.581). IL-6 was strongly correlated with CRP (r=0.843) and IgE (r=0.673). A strong inverse correlation was identified between IL-6 and forced vital capacity (FVC) (ρ=−0.77; p<0.001); no significant associations were found between IL-10 and spirometric indices. Conclusion. Comorbid BA and T2DM is characterized by more pronounced pro-inflammatory activity (elevated IL-6), activation of a compensatory anti-inflammatory response (elevated IL-10), and close immunometabolic interactions (leptin–cytokines–lipids). Heightened systemic inflammation is associated with poorer pulmonary function, while increasing glycemia is associated with higher IL-6 levels.
Keywords: Bronchial asthma, Type 2 diabetes mellitus, IL-6, IL-10, Leptin, Systemic inflammation, IgE, C-reactive protein, Spirometry
Cite this paper: Ubaydullaeva N. N., Khaidarova F. A., Ismatullaeva S. S., Immunological Profile Features in Patients with Bronchial Asthma Comorbid with Type 2 Diabetes Mellitus, American Journal of Medicine and Medical Sciences, Vol. 16 No. 3, 2026, pp. 1471-1475. doi: 10.5923/j.ajmms.20261603.130.
1. Relevance
- Bronchial asthma (BA) and type 2 diabetes mellitus (T2DM) are among the most prevalent chronic non-communicable diseases and constitute a substantial medical and societal burden. According to the World Health Organization, approximately 262 million people were living with asthma in 2019, and the condition is associated with considerable disability and mortality [1]. According to the International Diabetes Federation (IDF; Diabetes Atlas, 11th edition), in 2024 there were about 589 million adults aged 20–79 years living with diabetes worldwide (≈11.1% of the adult population); the proportion of undiagnosed cases remains high, and prevalence is expected to continue to rise [2].The comorbidity of BA and T2DM is of particular clinical importance in the context of increasing obesity and metabolic disturbances, which may contribute to the development of a “systemic” inflammatory phenotype in a subset of patients with BA. Current evidence indicates a bidirectional association between BA and T2DM (an increased risk of T2DM among patients with BA and vice versa), and suggests that this comorbidity is linked to poorer clinical outcomes and greater difficulty achieving disease control [3,4]. Contemporary asthma management strategies emphasize the need for proactive identification and correction of comorbidities and risk factors (including metabolic abnormalities and obesity) as an integral component of improving asthma control [5].The pathogenetic basis of an unfavorable course in comorbid BA and T2DM is considered to be chronic low-grade inflammation involving pro-inflammatory cytokines and adipokines. Interleukin-6 (IL-6) is implicated in the mechanisms of insulin resistance and hyperglycemia and is regarded as one of the key markers of the inflammatory component in T2DM; systematic reviews have demonstrated an association between elevated IL-6 levels and the risk of developing T2DM [6–8]. At the same time, obesity-associated BA is characterized by alterations in systemic and airway inflammation, and leptin—an adipokine—has been shown to correlate with inflammatory and clinical manifestations of BA, potentially serving as a link in immunometabolic interactions [9]. Nevertheless, data on the balance between pro-inflammatory (IL-6) and anti-inflammatory (IL-10) cytokines, their relationships with glycemic control parameters, as well as with markers of allergic inflammation and pulmonary function in the setting of comorbid BA and T2DM remain limited. This underscores the need for studies examining the immunological profile in patients with this comorbidity.Study aim. This article examines peripheral blood plasma cytokine levels in patients with bronchial asthma, type 2 diabetes mellitus, and their comorbidity, with the aim of identifying associations between inflammatory markers and hormonal–metabolic parameters that may influence the clinical course of asthma in patients with T2DM.
2. Materials and Methods
- The study included 90 patients who were hospitalized in the Endocrinology and Pulmonology departments of City Clinical Hospital No. 1 and Republican specialized scientific-practical medical center of endocrinology named after academic Yo.Kh. Turakulov (Tashkent, Uzbekistan). Patients were allocated to three sex- and age-matched groups (n=30 each): comorbid BA and T2DM, isolated BA, and isolated T2DM.All participants underwent a comprehensive clinical and laboratory evaluation, including hormonal and metabolic assessments with measurement of glycated hemoglobin (HbA1c), blood glucose, insulin, and leptin, as well as pulmonary function testing. BA was diagnosed according to the classification and criteria of the international consensus on asthma diagnosis and management (Global Initiative for Asthma—GINA, 2016). T2DM was diagnosed by an endocrinologist in accordance with established standards [10].Plasma IL-6 and IL-10 concentrations were determined by ELISA using standard protocols and commercial kits (Vector-Best; DRG, Germany) on a Mindray MR-96A microplate reader at 450–620 nm, with point-to-point calibration curve construction.Statistical analyses were performed using Microsoft Statistica 8.0. Depending on data distribution, parametric and non-parametric methods were applied; differences were considered statistically significant at p < 0.05.
3. Results
- We performed a comparative analysis of plasma IL-6 and IL-10 levels in patients with bronchial asthma, patients with comorbid bronchial asthma and type 2 diabetes mellitus, and patients with type 2 diabetes mellitus (Table 1).
|
|
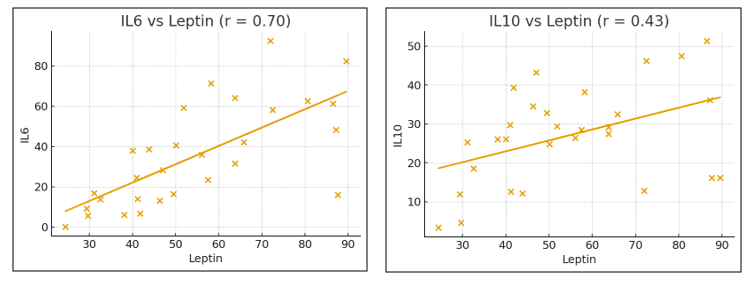 | Figure 1. Correlations between leptin and IL-6/IL-10 levels in patients with comorbid bronchial asthma and type 2 diabetes mellitus |
|
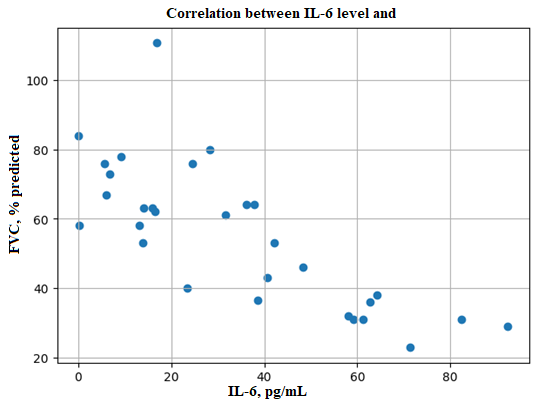 | Figure 2. Correlation between forced vital capacity (FVC) and IL-6 levels in patients with isolated bronchial asthma |
4. Discussion
- The present findings confirm that both BA and T2DM are accompanied by a systemic inflammatory background; however, in the setting of comorbidity the inflammatory response appears more pronounced and complex. This allows BA–T2DM comorbidity to be viewed as a state of mutual potentiation of inflammatory mechanisms: metabolic dysregulation in T2DM enhances systemic inflammation and endothelial dysfunction, whereas chronic airway inflammation in BA sustains persistent activation of pro-inflammatory cascades. Consequently, a more stable inflammatory phenotype may develop, potentially associated with poorer symptom control and a higher frequency of exacerbations.Of particular relevance is the observed association between IL-6 levels and glycemia. This suggests that inadequate glycemic control is accompanied by an intensified pro-inflammatory response. Pathogenetically, this relationship is plausible: hyperglycemia promotes oxidative stress, the formation of advanced glycation end products, and усиление cytokine-mediated inflammation, which may aggravate BA by increasing airway hyperresponsiveness and maintaining a systemic inflammatory milieu.IL-10, an anti-inflammatory cytokine, also reached its highest levels in the BA+T2DM group. At first glance, this may appear paradoxical; however, such a pattern is most plausibly interpreted as compensatory activation of anti-inflammatory pathways in response to an excessive pro-inflammatory burden. Concurrently, the significant correlations of IL-10 with IL-6 and CRP indicate that elevated IL-10 does not “switch off” inflammation, but rather reflects an attempt to limit its magnitude in the context of a chronic process.Particular attention should be paid to the identified immunometabolic interactions. Leptin occupied a central position within the correlation network: its positive associations with IL-6 and IL-10, as well as with atherogenic lipid fractions, support the role of dysfunctional adipose tissue as a source of adipokines and inflammatory mediators. Thus, in BA–T2DM comorbidity a “vicious circle” emerges: adipokine-driven inflammation enhances cytokine activity, while systemic inflammation exacerbates metabolic disturbances, thereby sustaining the chronic course of both diseases.Correlations between IL-6 and IgE point to a clinically important interplay between allergic and metabolic inflammation. The association of IL-6 with IgE suggests that, in the setting of comorbidity, not only systemic inflammation but also immunological activity related to the allergic component of BA is amplified, which may compromise the effectiveness of disease control. The functional relevance of this inflammatory burden is further supported by the inverse correlation between IL-6 levels and FVC: greater systemic inflammation is associated with reduced ventilatory capacity. Taken together, these findings indicate that IL-6 may be considered a marker reflecting not only laboratory evidence of inflammatory activity but also a potentially adverse impact on respiratory functional parameters.Additional considerationsRecent studies have emphasized the potential role of anti-aging genes, particularly Sirtuin-1 (SIRT1), in the regulation of inflammatory and metabolic processes associated with chronic diseases such as bronchial asthma and type 2 diabetes mellitus. SIRT1 is a NAD⁺-dependent deacetylase involved in the control of cellular metabolism, oxidative stress, and inflammatory signaling pathways.Decreased activity of SIRT1 has been associated with the development of hyperglycemia, insulin resistance, and excessive inflammatory responses. In patients with type 2 diabetes mellitus, reduced SIRT1 expression may contribute to increased production of pro-inflammatory cytokines, including interleukin-6, which plays an important role in systemic inflammation. In bronchial asthma, impaired SIRT1 signaling may also enhance airway inflammation and worsen disease severity.Therefore, assessment of Sirtuin-1 as a potential biomarker may be useful in patients with bronchial asthma combined with type 2 diabetes mellitus. Monitoring SIRT1 levels may help identify patients at higher risk of inflammatory and metabolic complications and may contribute to the development of more targeted therapeutic approaches.
5. Conclusions
- Elevated IL-6 levels relative to reference values were identified in patients with BA, T2DM, and their comorbidity, with the most pronounced increase observed in the BA+T2DM group, reflecting усиление of systemic inflammation in the setting of comorbidity.In BA+T2DM, pro-inflammatory activity was associated with metabolic disturbances: IL-6 levels were significantly higher at glucose ≥10.5 mmol/L; leptin correlated with IL-6/IL-10 and with atherogenic lipid profile parameters (with an inverse association with HDL-C), supporting the contribution of immunometabolic mechanisms.IL-6 was associated with markers of systemic and allergic inflammation (CRP and IgE) and showed an inverse correlation with forced vital capacity (FVC), indicating that greater inflammatory activity is linked to poorer pulmonary function.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML