-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(3): 1466-1470
doi:10.5923/j.ajmms.20261603.129
Received: Feb. 12, 2026; Accepted: Mar. 6, 2026; Published: Mar. 27, 2026

The Course of Chronic Brucellosis in Women of Fertile Age
Nigora Aliyevna Djalalova
Department of Infectious Diseases and Pediatric Infectious Diseases, Phthisiology and Pulmonology, Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: Nigora Aliyevna Djalalova, Department of Infectious Diseases and Pediatric Infectious Diseases, Phthisiology and Pulmonology, Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

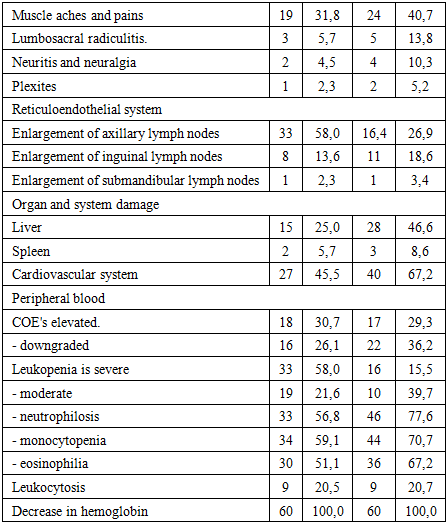
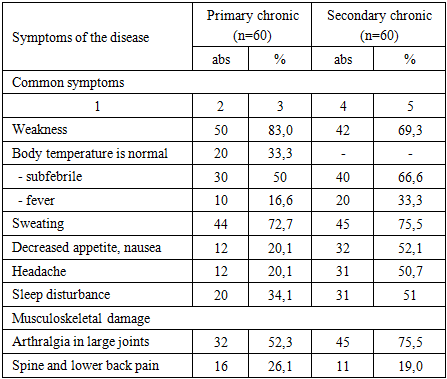
In women with primary chronic brucellosis, damage to the musculoskeletal system in 34.1% of cases was associated with damage to the peripheral nervous system, manifested by headache and poor sleep. In secondary chronic brucellosis, subjective data prevailed in patients - arthralgia in large joints of the arms and legs with predominant damage to large joints – bursitis and involvement of the nervous system – sciatica, neuritis, polyneuritis. Women with chronic brucellosis have a high incidence of concomitant extragenital pathology. It is possible that women with a history of high infection index are most susceptible to the development of chronic brucellosis, which is associated with a reduced state of the immune system. Thus, when assessing reproductive function, it was found that women with a history of chronic brucellosis are more likely to have early.
Keywords: Brucellosis, Chronic brucellosis, Fertile age, Brucellosis in women
Cite this paper: Nigora Aliyevna Djalalova, The Course of Chronic Brucellosis in Women of Fertile Age, American Journal of Medicine and Medical Sciences, Vol. 16 No. 3, 2026, pp. 1466-1470. doi: 10.5923/j.ajmms.20261603.129.
1. Introduction
- Recent years have witnessed significant advancements in the management of various infectious diseases, including brucellosis. Collaborative efforts among healthcare providers and veterinary specialists have effectively eradicated brucellosis from numerous previously endemic regions, leading to a marked reduction in human cases. However, despite these achievements, the incidence of brucellosis remains persistently high in specific geographical areas [1,2,3,4].Control of brucellosis continues to be a critical social concern due to its severe economic implications and health effects. Brucellosis infections can cause chronic illness, prolonged disability, and permanent impairments. While the disease has become relatively uncommon in many parts of the world, it still poses a significant health risk in Asia, particularly in endemic regions such as Central Asia and Kazakhstan [5,6,7].In Uzbekistan, recent data indicates a substantial increase in brucellosis across all demographic groups. Annually, Uzbekistan reports around 1.5 to 2.2 new brucellosis cases per 100,000 populations, a rate notably higher—between two to five times—than that observed in other CIS countries [8,9,10].Research conducted in Uzbekistan has shown that non-professional populations represent 81.6% of brucellosis cases, while the remaining 18.4% occur within professionally at-risk groups. Additionally, a correlation was established between clinical manifestations of brucellosis and the type of livestock farming practiced. Areas practicing mixed livestock farming demonstrated more severe disease progression, including significant organ involvement such as splenomegaly and hepatomegaly. A key factor identified was the prevalence of the highly virulent B. melitensis biovar 3 strain in these mixed farming areas. Uzbekistan also has circulating strains such as B. melitensis biovars 1 and 3, alongside B. abortus biovars 1, 3, and 6 [11].Another notable feature of brucellosis in Uzbekistan and Kazakhstan is the dominance of chronic disease forms, affecting up to 71.2% of cases, with primary chronic cases comprising up to 48.6% [12,13].The demographic most frequently affected by brucellosis includes individuals aged 20 to 40, typically engaged in meat and dairy production industries. Children, primarily consumers of dairy products, also exhibit a significant infection rate. Human-to-human transmission does not occur, rendering humans an epidemiological dead-end for brucellosis and other similar zoonotic infections [14,15].Over the past 25-30 years, limited research has explored the impact of chronic brucellosis on pregnancy outcomes. Studies, including those by E.M. Gvozdikova, have revealed notable clinical and pathological changes affecting female reproductive health due to brucellosis, such as menstrual irregularities (dysmenorrhea, oligomenorrhea, menorrhagia), impaired reproductive capacity, spontaneous miscarriages, and intrauterine fetal death [16].Our observations highlighted characteristic pathological changes in female reproductive organs caused by brucellosis, predominantly vascular alterations including sclerosis leading to significant vessel constriction or blockage. In acute brucellosis, inflammatory infiltrates and localized hemorrhages in ovaries were commonly found. Severe damage to female reproductive organs also occurs frequently in septic-metastatic forms, manifesting predominantly as menstrual irregularities, spontaneous abortions, and premature labor. Menstrual disorders, including amenorrhea, dysmenorrhea, metrorrhagia, and menorrhagia, have been extensively documented in affected individuals.Multiple studies indicate that brucellosis-related disturbances of menstrual cycles, such as amenorrhea and heteropolymenorrhea, frequently coexist with fertility issues, including infertility, spontaneous abortions, and increased risk of premature birth.Current immunocorrective therapies for brucellosis carry drawbacks such as high immunogenic potential and notable side effects. Therefore, enhancing therapeutic approaches for women with brucellosis is essential, emphasizing the correction of imbalanced cellular immunity, cooperative functioning of T- and B-lymphocytes and their regulatory subsets, and reduced phagocytic activity. This suggests integrating immunomodulatory agents into comprehensive brucellosis treatment regimens, which include etiotropic, anti-inflammatory, and symptomatic therapies.The purpose of this research was to analyze epidemiological and clinical characteristics of chronic brucellosis in reproductive-aged women.
2. Materials and Methods
- The base of clinical data collection is the Research Institute of Microbiology, Epidemiology and Infectious Diseases in Tashkent city. The study involved 120 female patients, aged between 19 and 39 years, diagnosed with chronic brucellosis. Diagnoses were confirmed through clinical examination, bacteriological cultures, serological tests, and statistical analysis.
3. Results and Discussion
- A total of 120 female patients with chronic brucellosis were included in our study. The selected age range, 19 to 39 years, corresponds to the reproductive phase of life. The overall average age of patients across all examined groups was 26.1±0.27 years, without statistically significant variations (P>0.05). Additionally, no significant disparities in age distribution were observed among subgroups. However, women aged 30 years and older (35.1%) and those aged between 20-24 years (29.1%) represented the largest subpopulations. A detailed summary of the age distribution is presented in Figure 1.
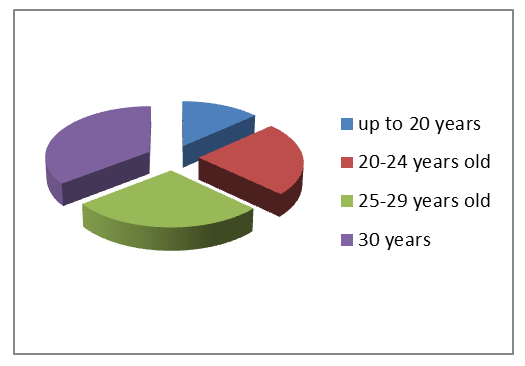 | Figure 1. Age of the patients |
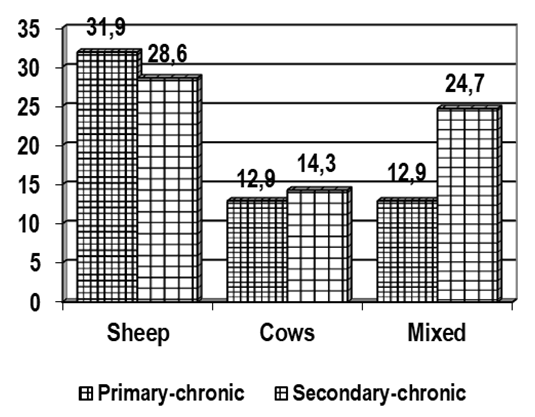 | Figure 2. Source of infection |
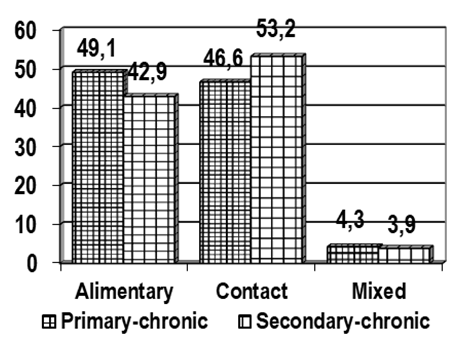 | Figure 3. Pathways of transmission |
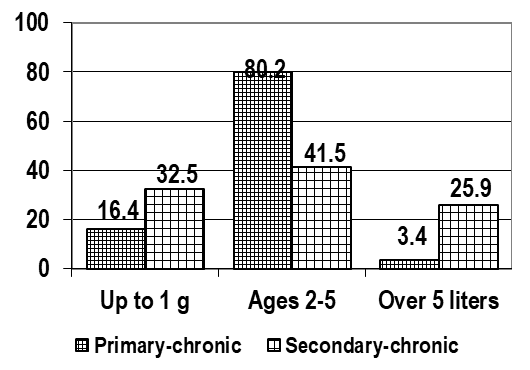 | Figure 4. Duration of the disease |
|
|
4. Conclusions
- Patients diagnosed with primary chronic brucellosis in 35% of cases had musculoskeletal disorders aggravated by neurological disorders. For example: headaches and sleep disorders. Patients with secondary chronic brucellosis predominantly reported subjective symptoms, including joint pain (arthralgia) in large joints of hands and feet, accompanied by bursitis and neurological issues like radiculitis, neuritis, and polyneuritis. Additionally, the presence of extragenital pathologies was notably high among the studied women. This may suggest that chronic brucellosis frequently develops in individuals with previous exposure to significant infectious events, likely due to compromised immune responses.Reproductive outcomes in women with chronic brucellosis frequently included early and late spontaneous abortions, premature labor, and increased perinatal mortality. The findings highlight the critical importance of prior infectious episodes, chronic inflammation, latent urinary tract infections, and delayed menstrual function development as risk factors. Understanding these clinical and immunological dynamics is essential for improving preventative and therapeutic strategies for chronic brucellosis.In conclusion, chronic brucellosis is significantly correlated with adverse reproductive outcomes, notably spontaneous miscarriages and premature births. These complications likely result from underlying inflammatory processes and immune disturbances characteristic of chronic infection, emphasizing the necessity for targeted clinical monitoring and management strategies in pregnant women with a history of brucellosis.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML