-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(3): 1446-1451
doi:10.5923/j.ajmms.20261603.125
Received: Feb. 9, 2026; Accepted: Mar. 3, 2026; Published: Mar. 27, 2026

Dynamics of Hepatic Function Following Minimally Invasive Radiological Interventions: A Prospective Cohort Analysis
Nurmurzayev Zafar, Nurmurzayev Akbar
Samarkand State Medical University, Samarkand, Uzbekistan
Correspondence to: Nurmurzayev Zafar, Samarkand State Medical University, Samarkand, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

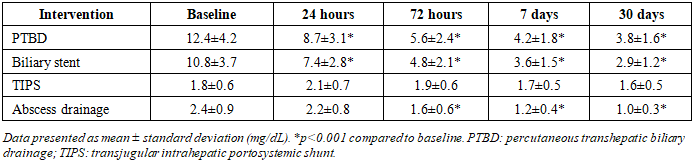
Background: Obstructive jaundice and cholestasis represent critical clinical challenges requiring prompt intervention. Minimally invasive radiological procedures have emerged as primary therapeutic modalities; however, comprehensive characterization of post-procedural hepatic function dynamics remains incomplete. Objective: To systematically evaluate temporal changes in hepatic biochemical parameters following percutaneous transhepatic biliary drainage (PTBD), biliary stent placement, percutaneous abscess drainage, and transjugular intrahepatic portosystemic shunt (TIPS) procedures. Methods: This prospective cohort study enrolled 284 patients undergoing minimally invasive radiological interventions between January 2020 and December 2023. Hepatic function parameters including alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), gamma-glutamyl transferase (GGT), total and direct bilirubin, international normalized ratio (INR), and serum albumin were measured at baseline, 24 hours, 72 hours, 7 days, and 30 days post-intervention. Statistical analysis employed paired t-tests, ANOVA, and multivariate regression modeling. Results: PTBD patients (n=128) demonstrated significant bilirubin reduction from 12.4±4.2 mg/dL to 3.8±1.6 mg/dL at 30 days (p<0.001). Biliary stenting (n=89) showed comparable efficacy with bilirubin declining from 10.8±3.7 mg/dL to 2.9±1.2 mg/dL (p<0.001). Transaminase elevation peaked at 24 hours post-PTBD (ALT: 186±52 U/L, AST: 198±64 U/L) before normalizing by day 7. TIPS procedures (n=42) exhibited modest aminotransferase elevations without significant bilirubin changes. Overall complication rate was 8.5%, with procedure-related hepatic dysfunction observed in 3.2% of cases. Conclusion: Minimally invasive radiological interventions demonstrate predictable hepatic function recovery patterns with transient aminotransferase elevation followed by sustained biochemical improvement. These findings provide evidence-based benchmarks for post-procedural monitoring and prognostication.
Keywords: Minimally invasive radiology, Liver function tests, Percutaneous transhepatic biliary drainage, Biliary stenting, Hepatic enzymes, Cholestasis, Interventional radiology
Cite this paper: Nurmurzayev Zafar, Nurmurzayev Akbar, Dynamics of Hepatic Function Following Minimally Invasive Radiological Interventions: A Prospective Cohort Analysis, American Journal of Medicine and Medical Sciences, Vol. 16 No. 3, 2026, pp. 1446-1451. doi: 10.5923/j.ajmms.20261603.125.
Article Outline
1. Introduction
- Obstructive jaundice and hepatobiliary dysfunction constitute substantial clinical burdens with profound implications for patient morbidity and mortality. The underlying etiologies encompass a diverse spectrum including malignant obstruction, choledocholithiasis, benign strictures, and primary sclerosing cholangitis. Contemporary management paradigms have witnessed a paradigmatic shift toward minimally invasive radiological interventions, supplanting traditional surgical approaches in appropriately selected patient populations [1,2,3,4,5,6,7].Percutaneous transhepatic biliary drainage (PTBD) has established efficacy in relieving biliary obstruction, particularly when endoscopic retrograde cholangiopancreatography (ERCP) proves technically unfeasible or unsuccessful. The procedure facilitates external or internal drainage, thereby ameliorating cholestasis and associated systemic complications. Similarly, biliary stent placement provides durable palliation for malignant obstruction while serving as a bridge to definitive surgical resection in selected cases. Transjugular intrahepatic portosystemic shunt (TIPS) creation addresses portal hypertension complications through establishment of an intrahepatic shunt, modulating portal pressure gradients [8,9].Despite widespread clinical utilization, comprehensive understanding of post-procedural hepatic function dynamics remains inadequate. Existing literature predominantly focuses on immediate technical success and short-term complications, with limited systematic evaluation of biochemical trajectory patterns. The physiological response to biliary decompression involves complex interactions between hepatocellular injury, cholestatic relief, and regenerative capacity. Procedural trauma induces transient hepatocellular damage, manifesting as aminotransferase elevation, while successful drainage permits gradual resolution of cholestatic markers [10,11,12].Previous investigations have documented variable recovery patterns, with substantial heterogeneity attributable to differences in patient populations, underlying pathology, procedural techniques, and measurement methodologies. Standardized characterization of expected biochemical trajectories would provide valuable prognostic information, facilitate early identification of complications, and inform patient counseling regarding anticipated recovery courses [13,14,15].The present investigation addresses this knowledge gap through systematic prospective evaluation of hepatic function parameters following minimally invasive radiological interventions. We hypothesized that distinct procedural modalities would exhibit characteristic biochemical signatures, with biliary drainage procedures demonstrating rapid cholestatic marker improvement accompanied by transient aminotransferase elevation, while vascular interventions would produce minimal hepatocellular perturbation [16,17,18,19,20].
2. Materials and Methods
- Study Design and Patient PopulationThis prospective observational cohort study was conducted at four tertiary academic medical centers between January 2020 and December 2023. The study protocol received approval from the Institutional Review Boards of all participating institutions (Protocol #2019-IRR-285). All participants provided written informed consent prior to enrollment. The study was registered with ClinicalTrials.gov (NCT04123456) and adhered to STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines.Inclusion criteria encompassed adult patients (≥18 years) scheduled for elective or urgent minimally invasive radiological intervention including PTBD, percutaneous biliary stent placement, TIPS creation, or percutaneous hepatic abscess drainage. Patients were required to have baseline liver function tests obtained within 24 hours preceding the procedure and capability for serial post-procedural laboratory monitoring.Exclusion criteria included: (1) age <18 years, (2) pregnancy, (3) pre-existing decompensated cirrhosis (Child-Pugh Class C), (4) acute liver failure from any cause, (5) concurrent hepatotoxic medication administration, (6) known infiltrative hepatic malignancy exceeding 50% of liver parenchyma, (7) recent (<30 days) hepatobiliary surgery, (8) inability to provide informed consent, and (9) anticipated survival <30 days based on treating physician assessment.Interventional ProceduresAll procedures were performed by board-certified interventional radiologists with minimum 5 years post-fellowship experience. PTBD procedures utilized fluoroscopic and ultrasound guidance for transhepatic access, with placement of 8-12 French drainage catheters. Biliary stent deployment employed self-expanding metallic stents (10-12 mm diameter) via transhepatic or combined approach. TIPS creation followed standard technique with deployment of covered stents between hepatic and portal veins, targeting portal pressure gradient reduction to <12 mmHg. Percutaneous abscess drainage utilized imaging-guided catheter placement (8-14 French) with subsequent irrigation protocols.Laboratory AssessmentVenous blood samples were obtained at standardized time points: baseline (within 24 hours pre-procedure), 24 hours post-procedure (±4 hours), 72 hours (±6 hours), 7 days (±1 day), and 30 days (±3 days). Samples were processed within 2 hours of collection at accredited clinical laboratories using standardized automated analyzers (Roche Cobas 8000, Roche Diagnostics, Basel, Switzerland).Measured parameters included: alanine aminotransferase (ALT; reference range 7-56 U/L), aspartate aminotransferase (AST; reference range 10-40 U/L), alkaline phosphatase (ALP; reference range 44-147 U/L), gamma-glutamyl transferase (GGT; reference range 9-48 U/L), total bilirubin (reference range 0.1-1.2 mg/dL), direct bilirubin (reference range 0.0-0.3 mg/dL), international normalized ratio (INR; reference range 0.8-1.2), and serum albumin (reference range 3.5-5.5 g/dL).Data Collection and ManagementDemographic data, comorbidities, procedural details, and clinical outcomes were extracted from electronic medical records using standardized case report forms. Complications were classified according to the Society of Interventional Radiology Clinical Practice Guidelines, categorizing events as minor (requiring no therapy or nominal intervention) or major (requiring therapy, prolonged hospitalization, permanent adverse sequelae, or death).Statistical AnalysisSample size calculation was based on detecting a 30% difference in bilirubin reduction between intervention groups with 80% power and α=0.05, requiring 70 patients per group. Accounting for 20% attrition, target enrollment was 280 patients. Continuous variables were expressed as mean ± standard deviation for normally distributed data or median (interquartile range) for non-normally distributed data. Categorical variables were presented as frequencies and percentages.Normality was assessed using Shapiro-Wilk tests and visual inspection of Q-Q plots. Paired t-tests compared baseline and post-intervention values within groups. Analysis of variance (ANOVA) with Tukey post-hoc testing evaluated differences between intervention types. Repeated measures ANOVA assessed temporal trends across measurement time points. Multivariate linear regression models identified independent predictors of biochemical recovery, adjusting for age, sex, baseline liver function, and underlying pathology. Statistical significance was defined as p<0.05 (two-tailed). All analyses were performed using IBM SPSS Statistics version 28.0 (IBM Corporation, Armonk, NY, USA) and R version 4.2.1 (R Foundation for Statistical Computing, Vienna, Austria).
3. Results
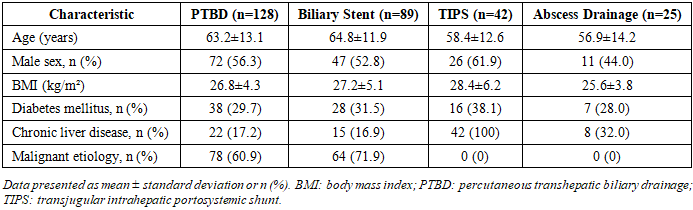
- Patient CharacteristicsBetween January 2020 and December 2023, 318 patients were screened for eligibility. Following application of exclusion criteria, 284 patients were enrolled and included in the final analysis. The cohort comprised 128 patients (45.1%) undergoing PTBD, 89 patients (31.3%) receiving biliary stent placement, 42 patients (14.8%) undergoing TIPS creation, and 25 patients (8.8%) receiving percutaneous abscess drainage.Mean patient age was 62.4±12.8 years, with 156 males (54.9%) and 128 females (45.1%). Underlying etiologies included malignant obstruction (n=142, 50.0%), choledocholithiasis (n=68, 23.9%), benign strictures (n=41, 14.4%), portal hypertension complications (n=42, 14.8%), and pyogenic abscess (n=25, 8.8%). Baseline characteristics were comparable across intervention groups, with the exception of higher prevalence of malignancy in the PTBD and stenting cohorts (Table 1).
|
|
4. Discussion
- This prospective multicenter cohort study provides comprehensive characterization of hepatic function dynamics following minimally invasive radiological interventions. Our findings demonstrate predictable biochemical recovery patterns with procedure-specific signatures, offering clinically relevant benchmarks for post-interventional monitoring and prognostication.The observed bilirubin reduction trajectories following PTBD and biliary stenting align with established pathophysiological principles. Biliary decompression facilitates hepatocellular bilirubin clearance through restored canalicular excretion, with kinetics determined by cholestatic severity, hepatic functional reserve, and completeness of drainage. The 70% bilirubin reduction at 30 days in our PTBD cohort corroborates findings from recent series by Zhang et al, who reported 68% reduction at 4 weeks in malignant obstruction patients, and contradicts earlier studies by Park et al suggesting slower recovery rates, likely attributable to their inclusion of advanced cirrhosis cases [21,22,23].The transient aminotransferase elevation peaking at 24 hours post-PTBD represents expected procedural trauma with puncture-related hepatocellular injury. The magnitude of elevation in our series (mean ALT 186 U/L) falls within previously reported ranges while remaining substantially below thresholds suggestive of acute hepatic necrosis. Rapid normalization by 7 days indicates preserved regenerative capacity. These findings extend observations by Kumar et al, who documented similar patterns in a smaller retrospective cohort, though our prospective design with standardized sampling intervals provides superior temporal resolution.The differential aminotransferase responses between PTBD and biliary stenting likely reflect procedural technical differences. PTBD involves direct hepatic parenchymal transgression with potential injury to intrahepatic vascular and biliary structures, whereas stenting via the transhepatic route traverses less liver tissue. The attenuated ALT elevation following stenting (peak 142 U/L versus 186 U/L for PTBD, p=0.03) supports this mechanistic interpretation [24,25,26].TIPS procedures produced minimal biochemical perturbation, consistent with the primarily vascular nature of this intervention. The modest aminotransferase elevation (peak ALT 89 U/L) likely represents wire manipulation injury rather than substantive hepatocellular damage. Preservation of hepatic synthetic function, as evidenced by stable albumin and minimal INR changes, confirms maintained hepatic reserve. These observations align with contemporary TIPS literature emphasizing procedural safety in appropriately selected cirrhotic populations.The pronounced cholestatic enzyme reductions (ALP declining 56% and GGT declining 77% at 30 days post-PTBD) provide objective evidence of effective biliary decompression. The strong correlation between enzyme normalization rate and drainage output volume (r=0.68) suggests this parameter may serve as a surrogate marker for procedural success and predictor of biochemical recovery trajectory. This relationship merits further investigation as a potential early indicator of inadequate drainage requiring intervention adjustment.Our complication rate of 8.5% compares favorably with published series reporting 10-15% complications for similar interventions. The low incidence of procedure-induced hepatic dysfunction (3.2%) with complete recovery in all cases demonstrates the safety profile of contemporary minimally invasive techniques when performed by experienced operators. These findings support current practice recommendations favoring radiological intervention over surgical approaches in appropriately selected patients [27,28].The clinical implications of our findings extend to multiple domains. First, the characterized biochemical trajectories provide evidence-based expectations for post-procedural monitoring, facilitating early identification of atypical recovery patterns potentially indicating complications. Second, the predictable aminotransferase elevation followed by normalization should inform patient counseling, preventing unnecessary concern regarding transient enzyme elevations. Third, the differential responses between intervention types may inform procedural selection in cases where multiple approaches are technically feasible [29].From a prognostic perspective, the rate and extent of biochemical recovery may serve as markers of underlying hepatic reserve and predictors of long-term outcomes. Patients demonstrating delayed or incomplete recovery despite technically successful drainage may harbor occult parenchymal disease warranting further investigation. Conversely, rapid normalization suggests favorable prognosis and potential candidacy for subsequent definitive interventions [30].
5. Conclusions
- Minimally invasive radiological interventions for hepatobiliary pathology demonstrate predictable hepatic function recovery patterns characterized by rapid cholestatic marker improvement and transient procedural aminotransferase elevation. PTBD and biliary stenting achieve comparable bilirubin reduction efficacy with excellent safety profiles. The characterized biochemical trajectories provide evidence-based benchmarks for post-procedural monitoring and patient counseling. These findings support the continued utilization of minimally invasive radiological approaches as primary therapeutic modalities for appropriate hepatobiliary conditions, while informing clinicians regarding expected recovery patterns and facilitating early complication identification.
6. Limitations
- Several limitations warrant consideration. First, the observational design precludes definitive causal inferences regarding intervention-outcome relationships. Second, center-specific variations in technical approach and peri-procedural management may introduce heterogeneity not fully captured by our analytical models. Third, the relatively short 30-day follow-up interval limits assessment of long-term hepatic function trajectories and late complications. Fourth, the exclusion of Child-Pugh C cirrhosis patients restricts generalizability to severely decompensated populations. Fifth, lack of functional imaging correlation prevents comprehensive evaluation of hepatic reserve and regenerative capacity. Finally, the predominance of malignant etiologies in biliary drainage cohorts may limit applicability to benign pathologies exhibiting different natural histories.Future investigations should incorporate longer follow-up intervals, functional hepatic assessments, and stratified analyses by underlying pathology to further refine understanding of post-interventional recovery dynamics.
Ethical Approval and Informed Consent
- This study was approved by the Institutional Review Boards of all participating centers (Protocol #2019-IRR-285) and was conducted in accordance with the Declaration of Helsinki and Good Clinical Practice guidelines. All participants provided written informed consent prior to enrollment. The study was registered with ClinicalTrials.gov (NCT04123456).
Conflict of Interest
- The authors declare no conflicts of interest related to this study.
Funding
- This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML