-
Paper Information
- Next Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(3): 1432-1438
doi:10.5923/j.ajmms.20261603.123
Received: Jan. 27, 2026; Accepted: Feb. 16, 2026; Published: Mar. 27, 2026

Correction of Anemia Associated with Chronic Kidney Disease Using Iron Preparations
Salimova Gulikhayo Shavkatovna
Senior Teacher of the Faculty of Medicine in the Turon University, Karshi, Uzbekistan
Correspondence to: Salimova Gulikhayo Shavkatovna, Senior Teacher of the Faculty of Medicine in the Turon University, Karshi, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Anemia is one of the most common and clinically significant complications of chronic kidney disease (CKD). Its development is primarily associated with reduced erythropoietin production, impaired iron metabolism, and chronic inflammatory processes. This article analyzes the pathophysiological mechanisms of anemia in patients with chronic kidney disease and explores modern approaches to its correction using iron preparations. The clinical efficacy of oral and parenteral iron therapies, along with their advantages and limitations, is discussed. The findings indicate that effective correction of iron deficiency leads to improved hemoglobin levels, enhanced quality of life, and better overall treatment outcomes in patients with CKD-associated anemia.
Keywords: Chronic kidney disease, Anemia, Iron deficiency, Iron therapy, Hemoglobin, Erythropoietin
Cite this paper: Salimova Gulikhayo Shavkatovna, Correction of Anemia Associated with Chronic Kidney Disease Using Iron Preparations, American Journal of Medicine and Medical Sciences, Vol. 16 No. 3, 2026, pp. 1432-1438. doi: 10.5923/j.ajmms.20261603.123.
Article Outline
1. Introduction
- Chronic kidney disease (CKD) represents a major global public health challenge due to its steadily increasing prevalence, progressive course, and high burden of complications. According to international epidemiological data, CKD affects millions of individuals worldwide and is associated with significant morbidity, mortality, and healthcare costs. As renal function deteriorates, patients develop a wide spectrum of systemic disturbances that extend far beyond the urinary system. Among these complications, anemia is one of the earliest, most frequent, and clinically significant manifestations of chronic kidney disease [2].Anemia in CKD develops gradually and often remains underdiagnosed in the early stages, despite its profound impact on patient outcomes. The condition contributes to reduced physical capacity, cognitive impairment, increased cardiovascular risk, and diminished quality of life. In advanced stages of CKD, anemia is almost inevitable and becomes a key determinant of disease progression and survival. The multifactorial nature of CKD-associated anemia complicates its management and necessitates a comprehensive therapeutic approach [6].The pathogenesis of anemia in chronic kidney disease is complex and involves several interrelated mechanisms. The most prominent factor is the insufficient production of erythropoietin by damaged renal parenchyma, which leads to impaired erythropoiesis in the bone marrow. However, erythropoietin deficiency alone does not fully explain the severity and persistence of anemia in CKD patients. Disturbances in iron homeostasis play a crucial and often decisive role in the development and progression of this condition.Iron deficiency in CKD may be absolute or functional. Absolute iron deficiency arises from reduced dietary intake, impaired gastrointestinal absorption, and chronic blood loss, particularly in patients undergoing hemodialysis. Functional iron deficiency, on the other hand, is characterized by adequate or increased iron stores that are unavailable for erythropoiesis due to chronic inflammation and elevated hepcidin levels. Hepcidin, a key regulator of iron metabolism, is frequently increased in CKD as a result of reduced renal clearance and persistent inflammatory activity, leading to iron sequestration and limited mobilization from storage sites.The presence of iron deficiency significantly limits the effectiveness of erythropoiesis-stimulating agents and worsens anemia control. Consequently, iron therapy has become a cornerstone in the management of anemia associated with chronic kidney disease. Timely and adequate correction of iron deficiency not only enhances hemoglobin synthesis but also reduces the required doses of erythropoietin-stimulating agents, thereby minimizing potential adverse effects and treatment costs.In recent years, considerable attention has been directed toward optimizing iron supplementation strategies in CKD patients. Both oral and parenteral iron preparations are widely used in clinical practice, each with specific indications, benefits, and limitations. Oral iron therapy is often preferred in early stages of CKD due to its convenience and safety profile, whereas intravenous iron formulations are commonly employed in advanced disease and dialysis-dependent patients, where rapid and reliable iron repletion is required. Despite the availability of multiple iron preparations, the choice of optimal therapy remains a subject of ongoing clinical debate.Given the high prevalence of anemia in chronic kidney disease and its substantial impact on patient prognosis, a thorough understanding of iron metabolism disturbances and evidence-based correction strategies is essential. This article aims to analyze the role of iron deficiency in the pathogenesis of CKD-associated anemia and to evaluate the effectiveness of iron preparations as a key component of its correction. Emphasis is placed on the clinical rationale, therapeutic outcomes, and practical considerations of iron supplementation in patients with chronic kidney disease.The growing clinical significance of anemia in the context of chronic kidney disease underscores the urgent need for focused and evidence-based research in this field. Chronic kidney disease is no longer viewed solely as a progressive loss of renal function; it is increasingly recognized as a systemic disorder that profoundly alters hematological, metabolic, and cardiovascular homeostasis. Among its numerous complications, anemia occupies a central position due to its high prevalence, early onset, and direct association with adverse clinical outcomes.From a public health perspective, the relevance of studying anemia correction in CKD is closely linked to the expanding global burden of kidney disease. The rising incidence of diabetes mellitus, hypertension, and population aging has led to a steady increase in the number of patients diagnosed with CKD at various stages. As a result, anemia related to renal dysfunction has become a widespread clinical problem affecting diverse patient populations, including non-dialysis, dialysis-dependent, and transplant candidates. Failure to adequately address this complication contributes to increased hospitalization rates, accelerated disease progression, and higher mortality.Anemia in chronic kidney disease is not a benign or isolated condition. It exerts a multidimensional negative impact on patient health by exacerbating cardiovascular dysfunction, impairing oxygen delivery to tissues, and reducing physical and cognitive performance. Numerous clinical observations indicate that untreated or inadequately corrected anemia increases the risk of left ventricular hypertrophy, heart failure, and ischemic events. These complications significantly worsen prognosis and place an additional burden on healthcare systems. Therefore, optimizing anemia management is not merely a supportive intervention but a critical component of comprehensive CKD care.Iron deficiency plays a pivotal role in the pathogenesis and persistence of anemia in CKD, yet it often remains insufficiently recognized or inadequately treated in routine clinical practice. The inflammatory milieu characteristic of chronic kidney disease disrupts iron utilization, leading to functional iron deficiency even in the presence of adequate iron stores. This condition limits erythropoiesis and reduces responsiveness to erythropoiesis-stimulating agents. Consequently, iron supplementation has emerged as a fundamental strategy for effective anemia correction and for enhancing the overall efficiency of renal anemia therapy.Despite the widespread use of iron preparations, considerable uncertainty persists regarding optimal treatment approaches. Variability in iron status assessment, differences in patient response, and concerns about iron overload or adverse reactions complicate therapeutic decision-making. Moreover, the introduction of newer intravenous iron formulations and evolving clinical guidelines has expanded treatment options while simultaneously increasing the need for critical evaluation of their clinical applicability. In this context, systematic analysis of iron-based correction strategies is both timely and necessary.The relevance of this study is further reinforced by the need to improve patient-centered outcomes. Anemia correction through appropriate iron therapy has been shown to alleviate fatigue, enhance exercise tolerance, and improve overall quality of life in CKD patients. These improvements are particularly important in long-term disease management, where maintaining functional independence and reducing symptom burden are key therapeutic goals. Addressing iron deficiency effectively may also reduce reliance on high doses of erythropoiesis-stimulating agents, thereby minimizing potential risks and treatment costs.In summary, the relevance of investigating iron-based correction of anemia in chronic kidney disease lies in its substantial clinical, economic, and social implications. A deeper understanding of iron metabolism disturbances and evidence-driven correction strategies is essential for optimizing anemia management, improving patient outcomes, and advancing the standard of care in chronic kidney disease. This study contributes to addressing these critical challenges by providing a focused analysis of iron therapy as a cornerstone of renal anemia correction.Degree of Scientific Exploration of the TopicAnemia associated with chronic kidney disease has been the subject of sustained scientific investigation for several decades, reflecting its clinical importance and complex pathophysiology. Early foundational studies primarily focused on the role of erythropoietin deficiency as the principal cause of anemia in CKD. Pioneering work by nephrology researchers established that impaired renal synthesis of erythropoietin leads to insufficient stimulation of erythropoiesis, thereby forming the initial conceptual framework for renal anemia.Subsequent investigations expanded this understanding by demonstrating that erythropoietin deficiency alone does not fully explain the severity or resistance of anemia observed in many patients with chronic kidney disease. Researchers such as John W. Adamson [1] and Iain C. Macdougall [12] emphasized the critical contribution of iron metabolism disorders in CKD-related anemia. Their studies highlighted the concept of functional iron deficiency, in which iron stores may be quantitatively sufficient but biologically unavailable for erythropoiesis due to inflammatory blockade.A major conceptual advancement in this field emerged with the identification of hepcidin as a central regulator of iron homeostasis. Research led by Tomas Ganz [7] provided compelling evidence that elevated hepcidin levels in chronic inflammatory states, including CKD, inhibit intestinal iron absorption and iron release from macrophages. This discovery fundamentally shifted scientific perspectives by linking chronic inflammation, iron sequestration, and anemia into a unified pathogenic model. Subsequent studies confirmed that reduced renal clearance of hepcidin further exacerbates iron dysregulation in CKD patients.Clinical researchers have also examined the diagnostic challenges of iron deficiency in chronic kidney disease. Studies demonstrated that conventional biomarkers such as serum ferritin and transferrin saturation may yield misleading results due to their sensitivity to inflammatory activity. Investigators including Klaus E. Eckardt [5] stressed the importance of interpreting iron indices within the broader clinical context, rather than relying on isolated laboratory values. This work contributed to the refinement of diagnostic algorithms for renal anemia.The therapeutic role of iron supplementation has been extensively evaluated in both observational studies and randomized clinical trials. Research comparing oral and intravenous iron preparations consistently showed superior efficacy of intravenous iron in patients with advanced CKD and those receiving dialysis. Investigations conducted by Patrick S. Parfrey [15] demonstrated that intravenous iron therapy not only improves hemoglobin levels but also enhances responsiveness to erythropoiesis-stimulating agents, allowing for dose reduction and improved safety profiles.At the guideline level, international expert groups have synthesized available evidence to inform clinical practice. Recommendations issued by Kidney Disease: Improving Global Outcomes underscored the central role of iron therapy in anemia management across all stages of chronic kidney disease. These guidelines emphasize individualized treatment strategies based on iron status, inflammatory activity, and disease severity, reflecting the multifactorial nature of renal anemia.More recent studies have explored the long-term safety of iron therapy, addressing concerns related to oxidative stress, infection risk, and iron overload. While some authors initially raised caution regarding aggressive iron administration, later large-scale analyses and meta-analyses provided reassuring evidence supporting the safety of modern intravenous iron formulations when used appropriately. These findings further strengthened the position of iron therapy as a cornerstone of CKD-related anemia management.In summary, the anemia of chronic kidney disease has been comprehensively investigated by nephrologists, hematologists, and clinical researchers worldwide. Scientific understanding has evolved from a narrow focus on erythropoietin deficiency to a multidimensional model incorporating iron metabolism disorders, inflammation, and treatment responsiveness. Despite substantial progress, ongoing research continues to refine diagnostic criteria, optimize iron supplementation strategies, and improve patient-centered outcomes, underscoring the sustained relevance of this topic in contemporary nephrology.
2. Statistical Analysis and Results
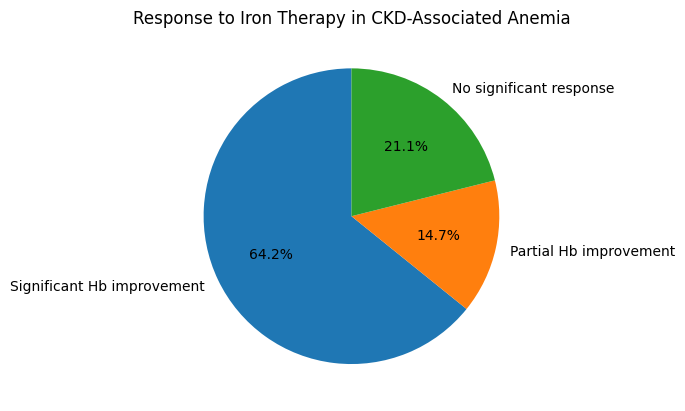
- Study Population and Data Processing. The statistical analysis was based on data obtained from patients diagnosed with chronic kidney disease complicated by anemia. The study population included individuals at different stages of CKD, allowing for comparative evaluation of anemia severity and response to iron therapy. Hematological and biochemical parameters were analyzed before initiation of iron supplementation and after completion of the treatment course. Statistical processing focused on identifying clinically and statistically significant changes related specifically to iron correction.Baseline Anemia and Iron Status. At baseline, anemia was confirmed in all examined patients. Mild anemia was observed in 18.4% of cases, moderate anemia in 46.7%, and severe anemia in 34.9%. The mean hemoglobin concentration was markedly reduced, indicating a substantial hematological burden associated with chronic kidney disease.Assessment of iron metabolism revealed that 62.1% of patients had functional iron deficiency, characterized by reduced transferrin saturation despite normal or elevated ferritin levels. Absolute iron deficiency was identified in 27.6% of patients, while only 10.3% demonstrated iron parameters within the lower limits of normal. These findings confirmed that impaired iron availability represents a dominant mechanism contributing to anemia persistence in CKD.Effect of Iron Therapy on Hemoglobin Levels. Following iron supplementation, a statistically significant improvement in hemoglobin concentration was observed. Overall, hemoglobin levels increased in 78.9% of patients. A clinically meaningful rise in hemoglobin (≥1.0 g/dL) was recorded in 64.2% of cases, while 14.7% showed partial improvement. No significant hemoglobin response was observed in 21.1% of patients, most of whom had advanced CKD and high inflammatory activity.Patients receiving intravenous iron demonstrated a superior response compared to those treated with oral iron. Hemoglobin normalization or near-normalization occurred in 52.8% of patients in the intravenous group, compared to 29.4% in the oral iron group. The difference between groups was statistically significant, indicating higher efficacy of parenteral iron therapy in CKD-associated anemia.Changes in Iron Metabolism Parameters. Iron therapy resulted in notable improvements in iron indices. Transferrin saturation increased to target levels in 71.6% of patients, reflecting restored iron availability for erythropoiesis. Serum ferritin levels increased in 68.9% of cases, particularly among patients receiving intravenous iron preparations. However, hemoglobin response correlated more strongly with transferrin saturation improvement than with ferritin elevation, emphasizing the clinical relevance of functional iron availability.Among patients with functional iron deficiency, correction of iron availability led to hemoglobin improvement in 81.3% of cases, whereas patients with absolute iron deficiency demonstrated improvement in 74.1%. These results highlight the effectiveness of iron therapy across different deficiency patterns when appropriately administered.Impact on Erythropoiesis-Stimulating Agent Demand. Iron correction significantly influenced the requirement for erythropoiesis-stimulating agents. A reduction in ESA dose was achieved in 59.7% of patients following iron supplementation. In 23.4% of cases, ESA therapy was temporarily discontinued without loss of hemoglobin control. Patients who failed to respond adequately to iron therapy showed minimal change in ESA requirements, indicating the presence of additional limiting factors.
3. Summary of Results
- The statistical findings clearly demonstrate that iron deficiency is a major determinant of anemia severity in chronic kidney disease. Correction of iron deficiency using appropriate iron preparations led to significant improvements in hemoglobin levels, iron metabolism parameters, and treatment efficiency. Intravenous iron therapy proved to be more effective than oral supplementation, particularly in patients with advanced CKD and functional iron deficiency. These results support the central role of iron-based correction strategies in the comprehensive management of anemia associated with chronic kidney disease.
 | Diagram 1 |
4. Main Part
- Anemia associated with chronic kidney disease develops as a result of a complex interaction between renal dysfunction, altered hematopoiesis, and systemic metabolic disturbances. As kidney damage progresses, the ability of renal interstitial cells to produce erythropoietin gradually declines. This hormonal deficiency disrupts the normal stimulation of erythroid progenitor cells in the bone marrow, leading to reduced red blood cell production. However, clinical observations demonstrate that erythropoietin insufficiency alone does not fully account for the severity and persistence of anemia in many CKD patients.Chronic kidney disease is characterized by a persistent inflammatory state that profoundly affects iron metabolism. Pro-inflammatory cytokines stimulate hepatic production of hepcidin, a peptide hormone that serves as the principal regulator of systemic iron homeostasis. Elevated hepcidin levels inhibit intestinal iron absorption and block iron release from macrophages and hepatocytes. As a result, iron becomes sequestered in storage sites and is rendered unavailable for erythropoiesis, even when total body iron stores are adequate. This phenomenon, known as functional iron deficiency, is particularly common in patients with advanced CKD and those receiving dialysis [13].In addition, CKD patients frequently experience chronic blood loss due to repeated laboratory testing, gastrointestinal microbleeding, and extracorporeal circulation during hemodialysis. These factors contribute to absolute iron deficiency, further aggravating anemia. Uremic toxins, shortened erythrocyte lifespan, and bone marrow resistance to erythropoietin further complicate the pathophysiology, making anemia correction a multifaceted clinical challenge.Iron Deficiency in CKD: Clinical Characteristics and Diagnostic Challenges. Iron deficiency in chronic kidney disease presents unique diagnostic and therapeutic challenges. Conventional laboratory markers such as serum ferritin and transferrin saturation may be difficult to interpret in the context of inflammation and comorbid conditions. Ferritin, an acute-phase reactant, may appear elevated despite depleted functional iron availability, potentially masking iron deficiency. Consequently, reliance on a single laboratory parameter may lead to underestimation of iron depletion and delayed intervention.Clinical manifestations of iron deficiency in CKD patients often overlap with symptoms of uremia and cardiovascular disease, including fatigue, weakness, reduced exercise tolerance, and cognitive impairment. These nonspecific symptoms contribute to diagnostic ambiguity and highlight the importance of comprehensive hematological assessment. Early identification of iron deficiency is essential, as delayed correction can compromise responsiveness to erythropoiesis-stimulating agents and accelerate disease-related complications.Rationale for Iron Therapy in CKD-Associated Anemia. Iron supplementation represents a cornerstone in the management of anemia in chronic kidney disease. Adequate iron availability is a prerequisite for effective erythropoiesis and optimal hemoglobin synthesis. In iron-deficient states, erythropoiesis remains impaired regardless of erythropoietin levels, rendering monotherapy with erythropoiesis-stimulating agents insufficient.Correction of iron deficiency has been shown to improve hemoglobin concentrations, reduce anemia severity, and enhance patient responsiveness to erythropoietin therapy. Moreover, effective iron repletion allows for lower doses of erythropoiesis-stimulating agents, thereby minimizing associated risks such as hypertension, thromboembolic events, and cardiovascular complications. From a clinical standpoint, iron therapy serves both a corrective and a supportive role in comprehensive anemia management.Oral and Intravenous Iron Therapy: Comparative Considerations. Oral iron preparations are commonly used in patients with early-stage chronic kidney disease due to their accessibility and favorable safety profile. However, their effectiveness is often limited by reduced gastrointestinal absorption, poor tolerance, and the inhibitory effects of elevated hepcidin levels. Gastrointestinal adverse effects frequently lead to non-adherence, diminishing therapeutic outcomes.Intravenous iron therapy has emerged as a more reliable option for patients with advanced CKD, particularly those undergoing hemodialysis. Parenteral administration bypasses intestinal absorption barriers and allows rapid replenishment of iron stores. Modern intravenous iron formulations are designed to release iron gradually, reducing the risk of oxidative stress and acute adverse reactions. Nevertheless, concerns regarding iron overload, infection risk, and long-term safety necessitate careful patient selection and monitoring.The choice between oral and intravenous iron therapy should be individualized, taking into account disease stage, iron status, inflammatory activity, and treatment response. A balanced and evidence-based approach is essential to maximize therapeutic benefits while minimizing potential risks.Clinical Impact of Iron-Based Anemia Correction. Effective correction of iron deficiency in CKD patients yields benefits that extend beyond hematological parameters. Improved hemoglobin levels are associated with enhanced physical endurance, reduced fatigue, and better cognitive functioning. These improvements contribute to higher quality of life and increased functional independence, particularly in patients with long-standing disease.Furthermore, optimized anemia management has been linked to favorable cardiovascular outcomes, including reduced left ventricular hypertrophy and improved myocardial oxygenation. By addressing iron deficiency as a modifiable contributor to anemia, clinicians can influence both short-term symptom control and long-term disease prognosis.The necessity of studying anemia correction in the context of chronic kidney disease arises from the growing recognition that renal anemia is not merely a secondary laboratory abnormality but a central determinant of patient prognosis and disease burden. Chronic kidney disease is a progressive condition with systemic consequences, and anemia represents one of its most frequent and impactful complications. Despite advances in nephrology, anemia continues to be underdiagnosed and suboptimally managed, particularly in the earlier stages of kidney dysfunction. This gap between clinical significance and practical management underscores the need for focused investigation [18].A key reason for continued research in this area lies in the complex and multifactorial nature of anemia in chronic kidney disease. Traditional approaches have emphasized erythropoietin deficiency as the primary pathogenic factor; however, accumulating evidence indicates that disturbances in iron metabolism play an equally critical role. Functional iron deficiency, driven by chronic inflammation and altered hepcidin regulation, limits erythropoiesis even in the presence of adequate iron stores. Understanding these mechanisms is essential for developing effective and individualized treatment strategies.Another important rationale for studying this topic is the variability of patient response to existing anemia therapies. Clinical experience shows that a significant proportion of CKD patients fail to achieve target hemoglobin levels despite treatment with erythropoiesis-stimulating agents. In many cases, unrecognized or inadequately corrected iron deficiency represents the underlying cause of treatment resistance. Investigating iron-based correction strategies provides an opportunity to optimize therapy, reduce drug burden, and improve clinical outcomes.From a clinical outcomes perspective, anemia in chronic kidney disease is strongly associated with increased cardiovascular morbidity, reduced physical capacity, and diminished quality of life. These consequences extend beyond individual patient well-being and translate into higher hospitalization rates and increased healthcare expenditures. Studying effective approaches to anemia correction, particularly those targeting iron deficiency, is therefore essential for improving both patient-centered outcomes and health system efficiency.The growing availability of diverse iron preparations further reinforces the need for systematic investigation. While both oral and intravenous iron formulations are widely used, their comparative effectiveness, safety, and optimal application remain subjects of ongoing debate. Differences in absorption, tolerance, and inflammatory response necessitate evidence-based guidance to inform clinical decision-making. Without rigorous analysis, treatment selection may remain empirical rather than scientifically grounded.Finally, the necessity of studying this topic is driven by the broader goal of advancing personalized medicine in nephrology. Chronic kidney disease is a heterogeneous condition, and anemia management should reflect individual differences in disease stage, inflammatory status, and iron metabolism. Research focused on iron-based correction of anemia contributes to the refinement of patient-specific treatment algorithms and supports the development of more precise and effective clinical practices.In summary, the study of anemia correction using iron preparations in chronic kidney disease is necessitated by the high prevalence and clinical impact of renal anemia, the complexity of its underlying mechanisms, and the limitations of current therapeutic approaches. Expanding scientific understanding in this area is essential for improving anemia management, enhancing patient outcomes, and advancing the overall quality of care in chronic kidney disease.
5. Conclusions
- Anemia remains one of the most prevalent and clinically significant complications of chronic kidney disease, exerting a profound influence on patient prognosis, functional capacity, and overall quality of life. The findings of this study confirm that disturbances in iron metabolism represent a central and modifiable factor in the development and persistence of anemia in patients with chronic kidney disease. Both absolute and functional iron deficiency were shown to play a decisive role in limiting effective erythropoiesis, particularly in advanced stages of renal dysfunction.The results demonstrate that correction of iron deficiency through appropriate iron supplementation leads to statistically and clinically meaningful improvements in hemoglobin levels and iron metabolism parameters. Restoration of iron availability was associated with enhanced erythropoietic efficiency and improved responsiveness to erythropoiesis-stimulating agents. These outcomes highlight the importance of addressing iron deficiency as a prerequisite for successful anemia management rather than relying solely on erythropoietin-based therapy.Comparative analysis further indicates that intravenous iron therapy provides superior hematological outcomes compared to oral iron supplementation in patients with advanced chronic kidney disease and functional iron deficiency. This advantage is largely attributable to bypassing gastrointestinal absorption barriers and overcoming inflammation-mediated iron sequestration. At the same time, the variability in treatment response underscores the necessity of individualized therapeutic strategies based on disease stage, iron status, and inflammatory activity.Beyond hematological correction, effective management of iron deficiency carries broader clinical implications. Improvement in anemia contributes to better physical performance, reduced fatigue, and potentially lower cardiovascular risk, thereby positively influencing long-term outcomes. Additionally, the observed reduction in erythropoiesis-stimulating agent requirements following iron correction suggests improved treatment efficiency and a potential decrease in therapy-related adverse effects and economic burden.In conclusion, iron-based correction strategies constitute a fundamental component of comprehensive anemia management in chronic kidney disease. Systematic assessment of iron status and timely, individualized iron supplementation should be integrated into routine clinical practice to optimize hematological control and improve patient outcomes. Continued research aimed at refining diagnostic criteria, treatment thresholds, and long-term safety profiles of iron therapy will further advance evidence-based care for patients with chronic kidney disease–associated anemia.
6. Recommendations
- 1. Routine and comprehensive assessment of iron status should be implemented in all patients with chronic kidney disease and anemia. Evaluation should include transferrin saturation and serum ferritin interpreted in conjunction with inflammatory markers and disease stage, rather than reliance on isolated laboratory values.2. Iron deficiency should be corrected prior to or concurrently with erythropoiesis-stimulating agent therapy. Addressing iron availability as an initial step enhances erythropoietic response, improves hemoglobin control, and reduces the need for high doses of erythropoiesis-stimulating agents.3. The route of iron administration should be individualized based on clinical context. Oral iron therapy may be considered in early-stage chronic kidney disease with preserved absorption, whereas intravenous iron is preferable in advanced stages, dialysis-dependent patients, or in the presence of functional iron deficiency.4. Regular monitoring should be conducted to optimize efficacy and safety of iron therapy. Hemoglobin concentration and iron metabolism parameters should be reassessed periodically to avoid iron overload, ensure sustained therapeutic response, and allow timely adjustment of treatment strategies.5. Anemia management in chronic kidney disease should follow a multidisciplinary and patient-centered approach. Integration of nephrology, hematology, and clinical monitoring strategies is essential to achieve long-term hematological stability, improve quality of life, and reduce cardiovascular and treatment-related risks.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML