-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(3): 901-904
doi:10.5923/j.ajmms.20261603.11
Received: Feb. 10, 2026; Accepted: Mar. 1, 2026; Published: Mar. 4, 2026

Evidence-Based Approaches to Treating Ovarian Endometriosis
Nazirova Zilola Mukhamadovna
PhD, Associate Professor, Department of Obstetrics and Gynecology No.1, Andijan State Medical Institute, Andijan, Uzbekistan
Correspondence to: Nazirova Zilola Mukhamadovna, PhD, Associate Professor, Department of Obstetrics and Gynecology No.1, Andijan State Medical Institute, Andijan, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Ovarian endometriosis is a prevalent gynecological disorder that significantly affects women of reproductive age and is commonly associated with chronic pelvic pain, dysmenorrhea, infertility, and a progressive decline in ovarian reserve. The condition represents one of the most clinically challenging manifestations of endometriosis due to its complex pathogenesis and potential impact on reproductive function. Despite advances in diagnostic imaging and surgical techniques, the optimal management strategy for ovarian endometriosis remains a subject of ongoing debate. This controversy is largely driven by heterogeneity in diagnostic criteria, variability in disease severity, and concerns regarding the possible detrimental effects of surgical intervention on healthy ovarian tissue. According to global epidemiological data, endometriosis affects approximately 10–15% of women of reproductive age, with ovarian endometriomas identified in up to 30–44% of diagnosed cases. The clinical and socio-economic burden of the disease is substantial, as patients often experience a prolonged diagnostic delay, averaging 7–10 years from the onset of symptoms to definitive diagnosis. Such delays contribute to disease progression, increased pain severity, reduced quality of life, and worsening reproductive outcomes. Surgical treatment, including cystectomy, ablation, and minimally invasive alternatives such as sclerotherapy, is frequently employed; however, each approach carries inherent risks, particularly related to postoperative reduction in ovarian reserve. Therefore, individualized treatment selection based on reproductive goals, ovarian reserve status, and disease characteristics remains critically important. Ovarian endometriosis is a common cause of infertility and decreased ovarian reserve in women of reproductive age. The choice of surgical treatment remains controversial due to the risk of ovarian tissue damage. A total of 110 women with ovarian endometriosis underwent surgical treatment and were divided into three groups according to ovarian reserve, endometrioma size, and surgical technique: cold cystectomy, bipolar ablation, or aspiration–sclerotherapy. Ovarian reserve was assessed using ultrasound and AMH levels. Cold cystectomy provided better ovarian tissue preservation in women with normal ovarian reserve. Bipolar ablation was effective for small endometriomas. In patients with recurrent disease and reduced ovarian reserve, aspiration–sclerotherapy showed favorable outcomes with minimal invasiveness. Treatment of ovarian endometriosis should be individualized. Organ-preserving techniques and fertility-oriented surgical strategies are essential to optimize reproductive outcomes.
Keywords: Ovarian endometriosis, Ovarian reserve, Laparoscopy, Fertility preservation, Minimally invasive gynecology
Cite this paper: Nazirova Zilola Mukhamadovna, Evidence-Based Approaches to Treating Ovarian Endometriosis, American Journal of Medicine and Medical Sciences, Vol. 16 No. 3, 2026, pp. 901-904. doi: 10.5923/j.ajmms.20261603.11.
1. Introduction
- Disputes regarding the optimal method for treating ovarian endometriosis largely stem from the broad and, at times, imprecise application of the diagnosis. Often, the diagnosis is based solely on clinical symptoms such as lower abdominal pain or dysmenorrhea, even though these symptoms may be present in various other gynecological conditions. For instance, dysmenorrhea is an independent clinical entity and not necessarily indicative of endometriosis. This diagnostic oversimplification can lead to mismanagement and the performance of unnecessary interventions, complicating the treatment strategy for women affected by this condition.Endometriomas negatively impact ovarian function. Numerous studies have documented a significant reduction in follicular density within endometriotic cysts as low as 6.3 per mm³, compared to 25.1 per mm³ in healthy ovarian tissue [1,6,7]. Even unilateral lesions of moderate size can lead to a decrease in the number of antral follicles one of the primary markers of ovarian reserve [3,4,6].Over the past three years, external genital endometriosis (EGE) has consistently ranked as the leading indication for surgical intervention in women with infertility in the Andijan Valley.Advanced forms of endometriosis requiring rectal mobilization and ureterolysis were performed in 5% of cases. Adnexectomy was indicated in 3% of women due to recurrent endometriomas, complete loss of healthy ovarian tissue, or age over 40 with completed reproductive plans. In 7% of cases, salpingectomy was carried out prior to planned IVF cycles due to tubal pathology.Ovarian endometriosis is one of the most common manifestations of endometriosis and remains a significant clinical challenge due to its impact on female reproductive function [4,6]. It is frequently associated with chronic pelvic pain, menstrual irregularities, and infertility. Globally, endometriosis affects approximately 10–15% of women of reproductive age, and ovarian endometriomas are present in up to 44% of those affected. Despite the high prevalence, the disease is often diagnosed late with delays averaging 7–10 years due to its non-specific clinical presentation and overlap with other gynecological disorders.The pathophysiological impact of ovarian endometriomas includes not only mechanical distortion of ovarian tissue but also biochemical changes that impair folliculogenesis and reduce ovarian reserve. Several studies have shown a significant decrease in follicular density and antral follicle count in ovaries affected by endometriotic cysts. These findings raise serious concerns regarding the timing and method of surgical intervention, especially in women desiring future fertility [7,9].The choice of surgical strategy remains controversial. While cystectomy is considered the gold standard, it carries the risk of reducing ovarian reserve due to the inadvertent removal of healthy cortical tissue. Alternative techniques such as laser ablation, bipolar coagulation, or sclerotherapy are aimed at minimizing this risk but vary in effectiveness and recurrence rates [10,11]. There is currently no universally accepted algorithm for the optimal management of ovarian endometriomas in women of reproductive age.
2. Material and Research Methods
- In this study, all 110 participants underwent surgical treatment either operative laparoscopy or transvaginal sclerotherapy. Indications for surgery primarily included ovarian masses with sonographic features suggestive of endometriosis, pelvic floor obliteration, and persistent lesions observed for over three months. Additional factors such as chronic pelvic pain and infertility were considered but not regarded as primary indications, as these symptoms were variably associated with ovarian masses detected on clinical or sonographic examination.
3. Results
- Based on the surgical techniques employed, patients were categorized into three groups:Group 1 (n=37) included women with normal AMH levels and endometriomas 4–6 cm in diameter, often with adhesions involving adjacent organs as seen on preoperative ultrasound. A “cold” cystectomy technique was used, employing aquadissection and avoiding high-energy surgical tools (electrocautery or laser) to minimize thermal damage to the ovarian cortex and preserve ovarian reserve.Group 2 (n=34) consisted of women with endometriomas smaller than 4 cm and normal AMH levels. These patients underwent ablation of the endometriotic lining using bipolar electrocautery.Group 3 included patients with diminished ovarian reserve and recurrent disease. All had ultrasound-confirmed involvement of adjacent organs in the adhesion process. Endometriomas in this group did not exceed 4 cm.Complete cystectomy for endometriomas is associated with a significant risk of reducing ovarian reserve a concern particularly in bilateral cases, where reproductive potential may be severely compromised. According to the World Health Organization, the reproductive age in women ranges from 18 to 35 years, highlighting the importance of preserving ovarian function in this demographic [6,12].
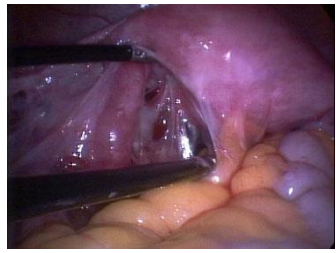 | Figure 1. Technique of “cold” cystectomy using aquadissection and avoidance of high-energy instruments to preserve ovarian tissue |
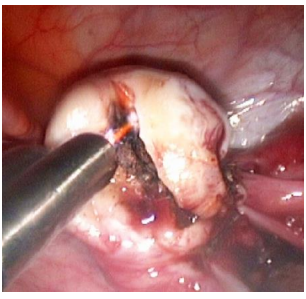 | Figure 2. Ablation of ovarian endometrioma using bipolar electrocautery. |
4. Conclusions
- The results of this study demonstrate that the choice of surgical treatment for ovarian endometriosis must be carefully individualized, taking into account the patient’s age, ovarian reserve status, size and localization of endometriomas, presence of recurrence, and the extent of pelvic adhesions.Organ-preserving techniques such as “cold” cystectomy with aquadissection have shown clear advantages in minimizing thermal injury to the ovarian cortex and preserving ovarian reserve in women of reproductive age [2,8]. Ablation of the endometriotic lining using bipolar electrocautery under controlled power settings can also serve as a viable alternative for small-sized endometriomas. In cases of recurrent disease with diminished ovarian reserve, intraoperative aspiration-sclerotherapy has proven to be a promising method, offering both efficacy and low invasiveness [3,9,10].These findings underscore the importance of a comprehensive approach to the management of ovarian endometriosis, which should include thorough clinical and imaging assessments, the use of fertility-preserving surgical technologies, and continuous monitoring of ovarian reserve [13]. Special attention must be given to young women who have not yet fulfilled their reproductive potential, for whom fertility preservation remains a top priority.Therefore, the optimization of surgical strategies in ovarian endometriosis should be guided by principles of individualization, minimally invasive intervention, and multidisciplinary care [1,2,7]. This will improve treatment efficacy and reproductive outcomes in this patient population.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML