-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(3): 881-886
doi:10.5923/j.ajmms.20261603.07
Received: Feb. 7, 2026; Accepted: Feb. 23, 2026; Published: Mar. 4, 2026

Features of the Dynamics of the Circadian Rhythm of Mean Blood Pressure in Severe Combined Traumatic Brain Injury in Children
Mukhitdinova Khura Nuritdinovna1, Shomurodov Akmal Yangiboy ogli2, Dadabaeva Yorkinoy Isakzhanovna3
1Professor, Center for the Development of Professional Qualifications of Medical Workers under the Ministry of Health of the Republic of Uzbekistan, Tashkent, Uzbekistan
2Anesthesiologist-Resuscitator, City Children’s Clinical Hospital No. 2, Tashkent, Uzbekistan
3Physician, Department of Pediatric Anesthesiology and Intensive Care Namangan Branch of the Republican Research Center of Emergency Medical Care, Uzbekistan
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

In Group 2, a decrease in the mesor of the circadian rhythm of mean arterial pressure (MAP) to normal values was identified. The severity of traumatic brain injury (TBI) had a significant impact on the effectiveness of comprehensive treatment of severe combined traumatic brain injury (SCTBI) and requires improvement of therapeutic approaches in children of Subgroup 3. It is possible that the comparatively more pronounced changes in the amplitude and circadian variability of MAP in Group 2 may be attributed to a more sparing stress-protective correction, which allowed more active involvement of compensatory mechanisms in the ongoing adaptive reactions of all systems, including the vascular system, in the postoperative period. In all subgroups of both groups, a strong direct correlation between MAP and diastolic arterial pressure (DAP) was revealed, which allows us to conclude that changes in DAP have a predominant influence on MAP levels in children with SCTBI.
Keywords: Circadian rhythm, Mean arterial pressure, Severe combined traumatic brain injury, Children
Cite this paper: Mukhitdinova Khura Nuritdinovna, Shomurodov Akmal Yangiboy ogli, Dadabaeva Yorkinoy Isakzhanovna, Features of the Dynamics of the Circadian Rhythm of Mean Blood Pressure in Severe Combined Traumatic Brain Injury in Children, American Journal of Medicine and Medical Sciences, Vol. 16 No. 3, 2026, pp. 881-886. doi: 10.5923/j.ajmms.20261603.07.
1. Introduction
- In severe traumatic brain injury (TBI), mean arterial pressure (MAP) must be maintained above 90 mmHg to ensure adequate cerebral perfusion pressure (CPP) and prevent cerebral ischemia and neuronal death, as autoregulatory mechanisms are impaired and cerebral blood flow becomes directly dependent on systemic arterial pressure [5,4,6]. Correction of arterial hypotension in patients with severe TBI should be performed based on comprehensive assessment of all hemodynamic components, taking into account the individual variability of changes at different stages of the disease. Some authors recommend maintaining MAP not lower than 80 mmHg [1,2,6]. To achieve target arterial pressure levels, various infusion solutions, vasopressor agents, inotropic drugs, or their combinations are used. However, irrational administration of vasopressors may lead to adverse effects associated with excessive vasoconstriction and impaired microcirculation, while excessive infusion therapy may result in pulmonary, intestinal, and systemic edema [4,6]. According to published data, normal MAP values in children are age-dependent: 73–77 mmHg at 3–7 years and 80–86 mmHg at 8–15 years. When adjusted for age, the mean MAP in healthy pediatric populations is approximately 79 ± 4 mmHg [2,3].ObjectiveTo investigate and evaluate alterations in the circadian rhythm of mean arterial pressure in children with severe combined traumatic brain injury (SCTBI).
2. Materials and Methods
- A comparative cohort study was conducted based on monitoring data of pediatric patients aged 3–18 years treated for severe combined traumatic brain injury (SCTBI) at the Republican Scientific Center for Emergency Medical Care between 2016–2021 (Group 1) and 2022–2024 (Group 2). In Group 1, injuries were primarily caused by road traffic accidents (n=23) and falls from height (n=11), whereas in Group 2 road traffic accidents accounted for 32 cases and falls (catatrauma) for 5 cases. In both cohorts, patients were stratified into three subgroups according to treatment strategy: Subgroup 1 included patients managed conservatively due to absence of surgical indications; Subgroup 2 comprised patients who underwent extracranial corrective surgical procedures within the first hours after admission; and Subgroup 3 included patients who received combined cranial and extracranial surgical interventions within the first 24 hours with multidisciplinary involvement. In Group 1, 13 patients received conservative therapy, 16 underwent extracranial surgery, and 8 underwent combined procedures; identical subgroup distributions were observed in Group 2 (13, 16, and 8 patients, respectively). No statistically significant differences in age distribution were identified between groups. A slight predominance of male patients was observed across most subgroups. There were no significant differences in injury severity or Glasgow Coma Scale (GCS) scores at admission between subgroups in Group 1. However, Injury Severity Score (ISS) values were significantly lower in Subgroups 2 and 3 of Group 2 (p<0.05), likely reflecting improvements in prehospital care and implementation of the “golden hour” principle. All patients underwent continuous hourly hemodynamic monitoring in the intensive care unit. Circadian rhythm parameters of mean arterial pressure (MAP), including mesor, acrophase, bathyphase, amplitude, and daily variability, were calculated to assess temporal hemodynamic patterns and to compare the effectiveness of surgical and conservative management strategies. Patients were transferred to specialized departments upon stabilization of hemodynamics, respiratory function, and neurological status.
3. Results and Discussion
- During the first 7 days, statistically significant differences between the groups were identified in the parameters of mesor, acrophase, and bathyphase (Table 1). In Subgroup 2 of Group 2, the mesor level of the circadian rhythm of mean arterial pressure (MAP) was 7% lower than in Group 1, while MAP values in the acrophase were 10% lower and in the bathyphase 11% lower (p<0.05, respectively).
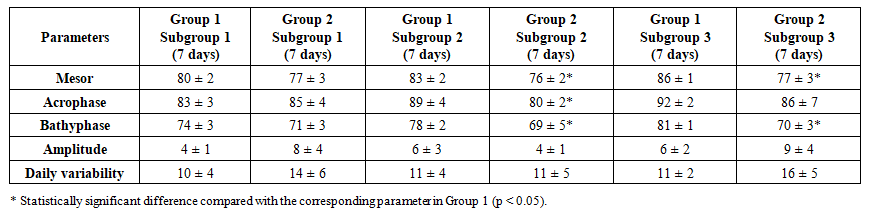 | Table 1. Comparative evaluation of the mean values of the circadian rhythm of mean arterial pressure during the first 7 days |
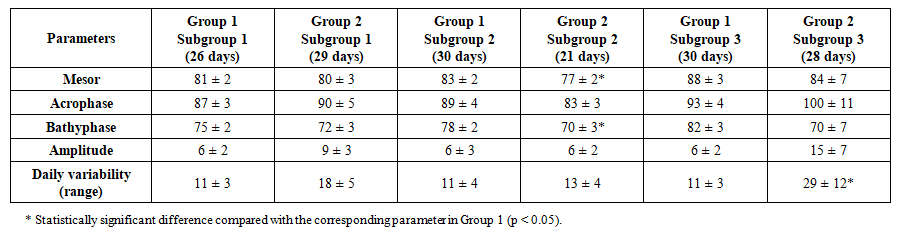 | Table 2. Comparative evaluation of mean circadian MAP values during ICU stay |
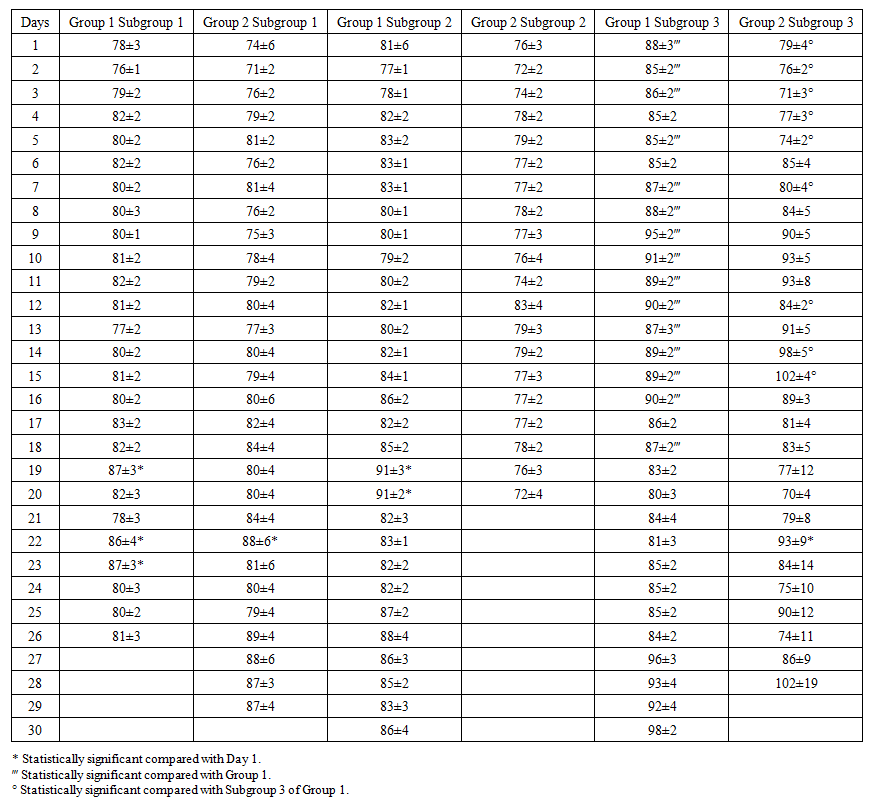 | Table 3. Dynamics of the mesor of the circadian rhythm of mean arterial pressure (MAP) |
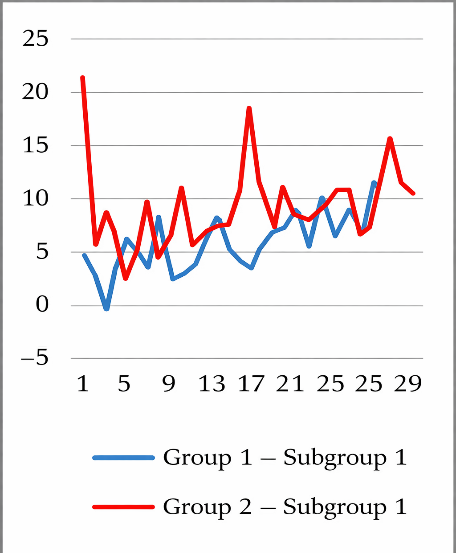 | Figure 1. Amplitude of the Circadian Rhythm of Mean Arterial Pressure (Subgroup 1) |
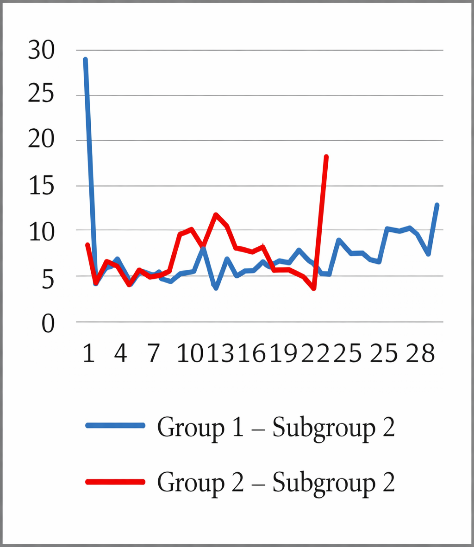 | Figure 2. Amplitude of the Circadian Rhythm of Mean Arterial Pressure (Subgroup 2) |
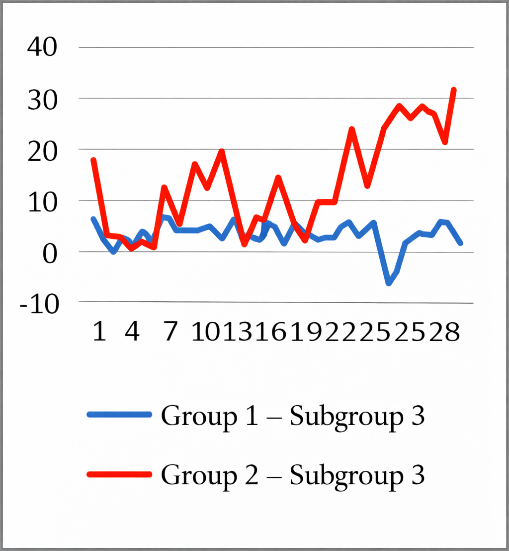 | Figure 3. Amplitude of the Circadian Rhythm of Mean Arterial Pressure (Subgroup 3) |
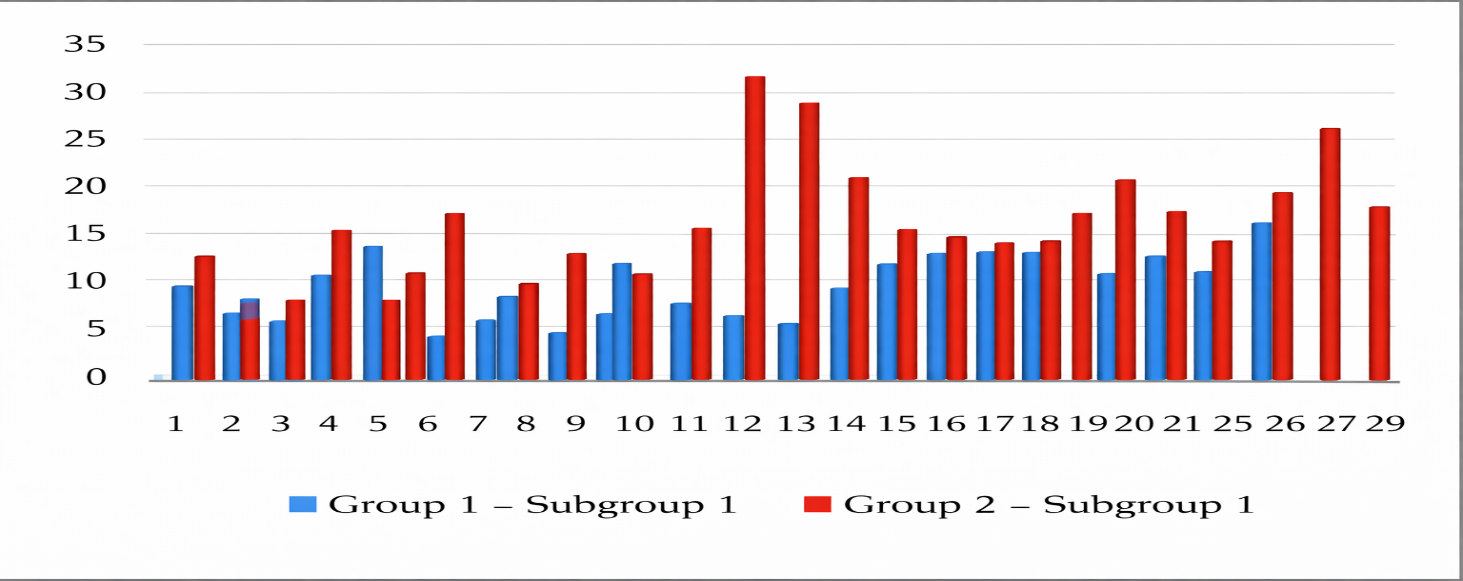 | Figure 4. Daily Fluctuations of Mean Arterial Pressure in Subgroup 1 |
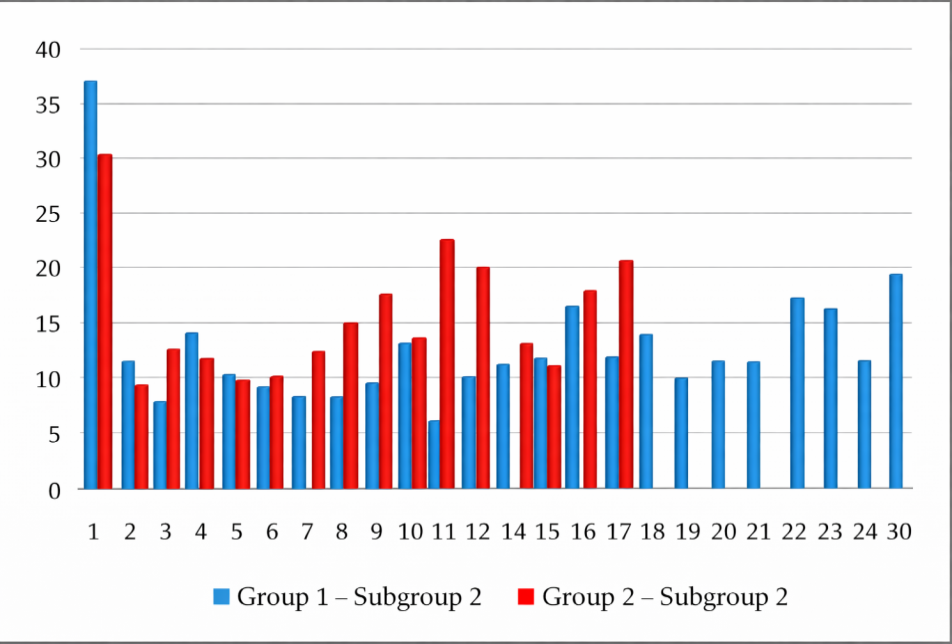 | Figure 5. Daily Fluctuations of Mean Arterial Pressure in Subgroup 2 |
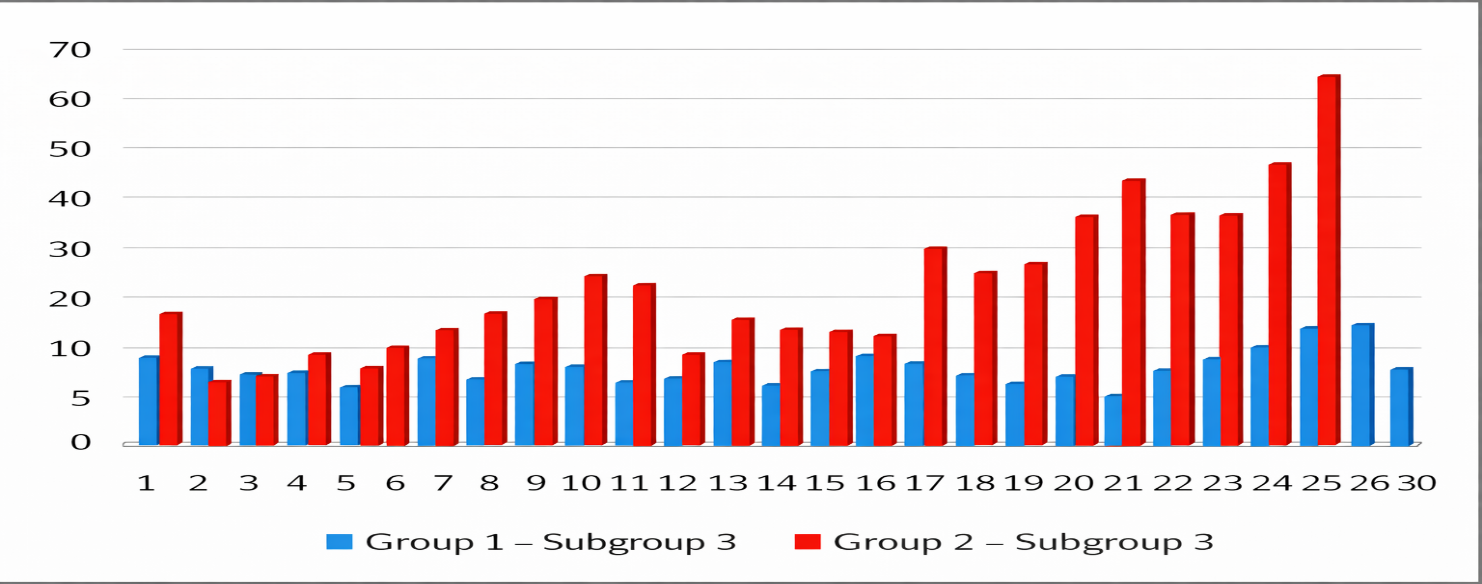 | Figure 6. Daily Fluctuations of Mean Arterial Pressure in Subgroup 3 |
4. Conclusions
- In Group 2, normalization of the mesor of the circadian rhythm of mean arterial pressure (MAP) was observed. The severity of traumatic brain injury significantly influenced the effectiveness of comprehensive treatment of severe combined traumatic brain injury (SCTBI) and indicates the need to optimize therapeutic strategies in children of Subgroup 3. The comparatively more pronounced changes in MAP amplitude and daily variability in Group 2 may be associated with a more sparing stress-protective correction, which allowed greater activation of compensatory mechanisms in ongoing adaptive responses of all systems, including the vascular system, during the postoperative period. In all subgroups of both groups, a strong positive correlation between MAP and diastolic arterial pressure (DAP) was identified, confirming the predominant influence of DAP changes on MAP levels in children with SCTBI.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML