-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 837-841
doi:10.5923/j.ajmms.20261602.94
Received: Feb. 4, 2026; Accepted: Feb. 22, 2026; Published: Feb. 27, 2026

Characteristics of Autonomic Nervous System Dysfunction in Children and Adolescents with Migraine
Alidjanova Durdona Abdullajonovna1, Kadirova Robiya Mirabbasovna2
1MD, DSc, Associate Professor, Tashkent State Medical University, Tashkent, Uzbekistan
2Neurologist, SBJ MEDICAL, Tashkent, Uzbekistan
Correspondence to: Alidjanova Durdona Abdullajonovna, MD, DSc, Associate Professor, Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

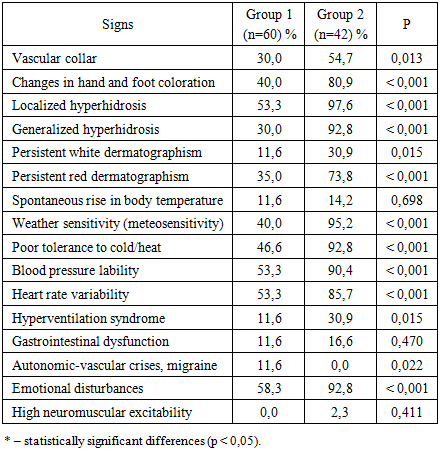
The study revealed a fundamental transformation of autonomic dysfunction depending on the patients’ age. In the first group (7–11 years), autonomic disorders were predominantly paroxysmal in nature, reflected by a higher prevalence of vegetative-vascular crises and migraines (p = 0.022). In the second group (12–18 years), a transition to systemic, persistent maladaptation was observed. Statistically significant differences (p < 0.001) in Group 2 were identified in almost all key parameters, namely local hyperhidrosis (97.6%), meteorological sensitivity (95.2%), emotional disturbances (92.8%), and blood pressure lability (90.4%). Such a high prevalence of symptoms indicates profound depletion of the body’s adaptive reserves and the formation of a stable pathological background.
Keywords: Autonomic dysfunction syndrome, Age-related characteristics, Maladaptation, Guillaume–Wayne questionnaire, Meteorological sensitivity
Cite this paper: Alidjanova Durdona Abdullajonovna, Kadirova Robiya Mirabbasovna, Characteristics of Autonomic Nervous System Dysfunction in Children and Adolescents with Migraine, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 837-841. doi: 10.5923/j.ajmms.20261602.94.
1. Introduction
- Migraine is one of the most disabling conditions affecting both adults and children. In adults, it causes difficulties in personal life, professional activity, and social functioning, whereas in children it may negatively affect academic performance and leisure activities, including frequent school absenteeism [1]. The clinical presentation of migraine differs significantly between adults and children. It is well known that in both age groups especially in children the disease is often accompanied by symptoms not directly related to headache [2]. These may manifest as episodic syndromes associated with migraine. Interestingly, such episodic syndromes were described in pediatric patients much earlier than in adults [3].The autonomic nervous system (ANS) plays a significant role in the pathogenesis of migraine. It should be noted that ANS disorders are considered the most common comorbid conditions accompanying migraine attacks (94.2%) [4]. Autonomic imbalance is characterized by the predominance of sympathetic over parasympathetic activity, with three principal directions of change identified. First, migraine with aura generally provokes more pronounced autonomic disturbances compared to migraine without aura. Second, autonomic dysfunction is observed more frequently in the sympathetic division than in the parasympathetic division. Third, sympathetic disturbances are often detected in the interictal period, with increased sympathetic tone during attacks, indicating adrenergic receptor hyperreactivity. Despite the available data on the role of the ANS in pediatric migraine, the relationship between migraine and autonomic dysfunction remains insufficiently understood, and it is still unclear which component is primary and which is secondary [5,6].It is important to understand that the ANS plays a key role in regulating emotional states, adaptation to environmental conditions, and metabolic processes, thereby ensuring optimal mental and physical activity. Its functional state significantly influences the course and prognosis of various pathologies across all age groups. Deviations in ANS function accompany nearly all diseases, including headache disorders, particularly migraine. Early detection of autonomic dysfunction in patients with headaches contributes to more accurate symptom management, improves overall well-being, and enhances quality of life [7,8]. This is especially relevant for children and adolescents, during whom maturation of segmental and suprasegmental brain structures occurs, influencing adaptive processes, disease development and progression, and the formation of pain syndromes, including cephalalgias.The clinical presentation of vegetative-vascular dystonia (VVD) in children and adolescents with migraine is highly variable and involves multiple organ systems. Several clinical forms may occur independently or in combination: arterial hypertension or hypotension; neurogenic syncope; non-infectious headache; visual and balance disturbances; fever associated with neurological disorders; functional cardiac insufficiency; hyperventilation syndrome; upper gastrointestinal motility disorders; lower gastrointestinal motility disorders (irritable bowel syndrome); vascular-trophic neuralgia; neurogenic bladder dysfunction; functional pulmonary hypertension; hyperhidrosis; neuroendocrine disorders; and episodes of autonomic dysfunction (panic attacks) [9].During clinical assessment, it is essential to consider the overall pattern of autonomic imbalance sympathicotonic, vagotonic, or mixed. For example, adolescence is characterized by enhanced sympathoadrenal activity, reflecting general activation of the neuroendocrine system associated with age-related changes [10]. Adolescents of the sympathicotonic type are typically slender, with reduced appetite. Their skin is dry and pale, with barely visible vascular patterns and persistent white dermographism. They often report cardialgia and show a predisposition to bradycardia and elevated blood pressure. Complaints may also include constipation and infrequent but abundant urination [11].Patients with vagotonia commonly demonstrate a tendency toward overweight despite poor appetite. They often experience a persistent feeling of coldness, increased sweating, and low-grade fever. The skin has a marbled appearance, feels cool to the touch; hands and feet remain cold and moist, frequently acquiring a cyanotic tint. Dermographism is pronounced, long-lasting, and sometimes raised. These adolescents frequently suffer from bradycardia or arrhythmias, tend toward hypotension, complain of cardiac pain, experience syncopal episodes, poorly tolerate travel, and report dyspnea. A significant proportion also exhibit abdominal pain, nausea, hypersalivation, flatulence, and frequent but low-volume urination [12].Objective of the Study. To determine the severity of autonomic disorders in children and adolescents with migraine.
2. Materials and Methods
- A total of 102 patients aged 7–18 years with a confirmed diagnosis of migraine (ICD-10: G43) were examined. Group I included 60 patients (58.8%) aged 7–11 years, of whom 33 (58.9%) were boys and 27 (58.7%) were girls. Group II consisted of 42 patients (41.1%) aged 12–18 years, including 23 boys (41.1%) and 19 girls (41.3%).All participants underwent a comprehensive clinical and neurological examination to identify key signs of involvement of the central, peripheral, and autonomic nervous systems [13]. Particular attention was paid to aggravating factors, including a history of frequent acute respiratory viral infections, chronic somatic diseases, vascular disorders, and emotional or physical stress.Autonomic nervous system (ANS) activity was assessed using clinical methods. Autonomic tone was evaluated according to the Guillaume–Wayne scale, where scores were assigned based on manifestations of sympathetic and parasympathetic activity across various organs and systems, followed by determination of the predominant orientation (sympathetic vs. parasympathetic). To diagnose autonomic dystonia syndrome (ADS), we used an 11-item patient questionnaire (a total score of ≥15 suggesting ADS) and a 13-item physician-completed checklist for detecting autonomic deviations (in individuals without pathology, the total score should not exceed 25) [14]. Statistical analysis was performed using the software package Statistica 8.0 (StatSoft, USA, Windows XP).
3. Results and Discussion
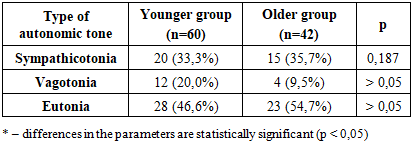
- According to medical history data, 24 patients (23.5%) had a complicated perinatal history, 28 patients (27.4%) demonstrated hereditary predisposition, and 51 patients (50.0%) had migraine attacks of unclear etiology. During interictal clinical neurological examination, no gross focal neurological deficits were detected; only scattered microfocal signs were observed.Paraclinical investigations revealed signs of angioencephalopathy on brain MRI in 32 patients (31.3%), demyelinating lesions in 11 patients (12.0%), and no detectable pathology in 58 children (57.0%). EEG findings showed brainstem dysfunction in 38 children (37.2%), epileptiform activity in 19 patients (18.6%), and diffuse brain changes in 45 children (44.1%). Transcranial Doppler ultrasonography identified vertebrobasilar insufficiency in 64 patients (62.7%) and congenital vascular anomalies in 17 children (16.6%).Analysis of baseline autonomic status according to the Guillaume–Wayne scale (modified for children) demonstrated a predominance of sympathicotonia in both groups: 20 patients (33.3%) in Group I and 15 patients (35.7%) in Group II. Although sympathetic predominance was more pronounced in the older group, no statistically significant intergroup differences were found (p = 0.187). Vagotonia was significantly more common in younger children 12 patients (20.0%) compared to 4 patients (9.5%) in the older group. Eutonia was recorded in 28 patients (46.6%) in Group I and 23 patients (54.7%) in Group II (Table 1).
|
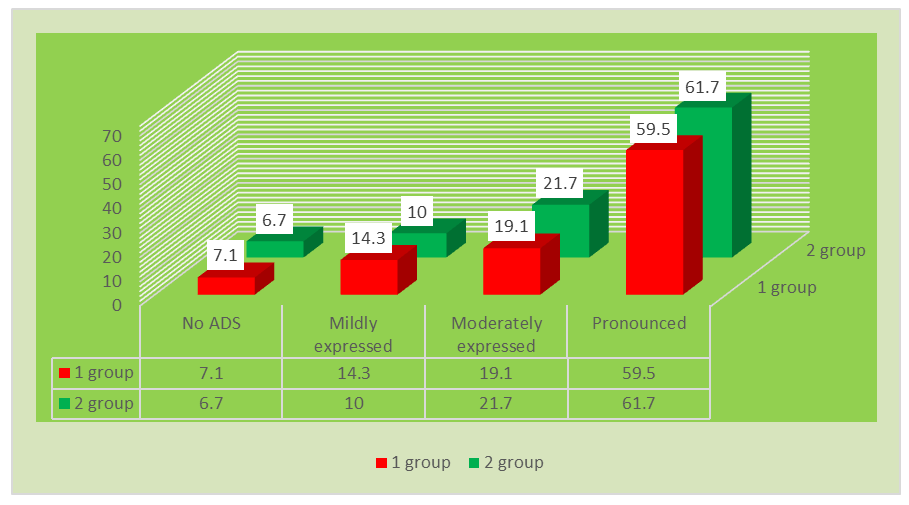 | Figure 1. Severity of autonomic dysfunction syndrome depending on age |
|
4. Conclusions
- Analysis of the autonomic profile in an age-related context allowed us to identify a fundamental pattern: the development of autonomic dysfunction progresses from the so-called “stormy” type (rare but intense crises) to the “diffuse” type (persistent lability of blood pressure, heart rate, and psychoemotional sphere). This pattern indicates a pronounced depletion of adaptive reserves with age in children with migraine and necessitates a shift in therapeutic focus from merely controlling acute migraine episodes to comprehensive neuroprotection and psychoautonomic correction. Finally, it should be emphasized that autonomic dysfunction in children with migraine is not a secondary symptom but an important predictor of disease severity.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML