-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 783-790
doi:10.5923/j.ajmms.20261602.82
Received: Jan. 22, 2026; Accepted: Feb. 20, 2026; Published: Feb. 27, 2026

Comparative Assessment of the Specific Activity of Various Synthetic Osteoplastic Materials for Bone Defect Restoration
Akbarov Avzal Nigmatullayevich, Nigmatova Nigora Rakhmatullayevna, Khabilov Bekhzod Nigmanovich
Tashkent State Medical University, Tashkent, Uzbekistan
Correspondence to: Nigmatova Nigora Rakhmatullayevna, Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

This review article compares the key classes of synthetic osteoplastic materials used for bone defect replacement: hydroxyapatite (HA), bioactive glasses (BG), and calcium phosphates (β-tricalcium phosphate, biphasic compositions, calcium phosphate cements). The analysis covers biocompatibility, osteointegration, osteoconductivity/osteoinductivity, biodegradation, and clinical limitations, as well as mechanisms of interaction with bone tissue. The main differences highlighted are: extremely slow resorption and high volumetric stability of HA; bioactivity and ion-induced stimulation of osteogenesis in BG, in some cases combined with an antimicrobial effect; and controlled resorption of calcium phosphates, allowing for material selection based on remodeling rates. Comparative tables are presented, which can serve as a practical algorithm for material selection in traumatology-orthopedics and maxillofacial surgery.
Keywords: Osteoplasty, Bone defects, Hydroxyapatite, Bioactive glass, β-tricalcium phosphate, Biphasic calcium phosphates, Calcium phosphate cements, Osteoconductivity, Osteoinductivity, Biodegradation
Cite this paper: Akbarov Avzal Nigmatullayevich, Nigmatova Nigora Rakhmatullayevna, Khabilov Bekhzod Nigmanovich, Comparative Assessment of the Specific Activity of Various Synthetic Osteoplastic Materials for Bone Defect Restoration, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 783-790. doi: 10.5923/j.ajmms.20261602.82.
1. Introduction
- The restoration of bone defects remains a pressing challenge in traumatology, orthopedics, and maxillofacial surgery. Osteoplastic materials are designed to replace or supplement bone autografts, avoiding the drawbacks of autogenous bone grafts (limited quantity, donor site morbidity, risk of complications). A modern synthetic bone substitute must meet several criteria: be biocompatible, ensure strong fusion with the bone (osteointegration), serve as a scaffold for new bone ingrowth (osteoconductivity), stimulate de novo bone formation (osteoinductivity), and gradually biodegrade while being replaced by new bone tissue. The process of implant degradation can occur both through material dissolution and cellular resorption [1,2,15,16].In practice, no class of materials fully meets all the listed requirements. Synthetic bioceramic materials based on calcium phosphates (CaP) and bioactive glasses are the most common bone substitutes; however, when used separately, they fulfill these functions only partially. Generally, such materials are osteoconductive and integrate well with bone, but have limited or no osteoinductivity. Nevertheless, over the past 10 years, evidence has emerged that certain compositions and microstructures are capable of inducing osteogenesis to some extent [1,2,15,16].This review article focuses on three main types of synthetic osteoplastic materials: hydroxyapatite (HA), bioactive glass (BG), and other calcium phosphates (e.g., β-tricalcium phosphate and biphasic compositions). Their osteoconductive and osteoinductive properties, biocompatibility and resorption, research results in vitro and in vivo, clinical applications, and mechanisms of interaction with bone tissue will be discussed. The comparative analysis is based on data from scientific publications, primarily from the last decade.
2. Materials and Methods
- Study design: narrative review with elements of comparative analysis.Sources: publications in peer-reviewed journals dedicated to synthetic osteoplastic materials (HA, BG, CaP) and their use in vitro, in vivo, and in clinical practice.The literature search was conducted using the following keywords: "hydroxyapatite," "bioactive glass," "45S5," "S53P4," "β-tricalcium phosphate," "biphasic calcium phosphate," "calcium phosphate cement," "osteoconduction," "osteoinduction," "bone defect repair," with priority given to publications from the last 10 years.Inclusion criteria: description of composition/microstructure, data on resorption and bone replacement, comparable outcomes (histology/CT/X-ray), and/or clinical results.Comparison parameters: (1) osteoconductivity/osteoinductivity, (2) biocompatibility and inflammatory response, (3) osteointegration mechanism and degradation products, (4) resorption rate, (5) mechanical limitations and application format (granules, blocks, coatings, cements).
3. Research Results
- Comparative analysis showed that all the materials under consideration possess pronounced osteoconductivity, but differ in bioactivity, osteoinductive potential, and resorption rate. The results for each class are presented below.Hydroxyapatite (HA)Hydroxyapatite, Ca10 (PO4) 6 (OH) 2, is the main mineral component of human bone tissue. About 70% of the bone matrix mass consists of carbonate-substituted hydroxyapatite, which is why synthetic HA is chemically similar to natural bone mineral. Due to this, it exhibits exceptionally high biocompatibility: after implantation, hydroxyapatite can directly fuse with the surrounding bone without a fibrous layer, ensuring strong osteointegration. Since its discovery, synthetic hydroxyapatite has been widely used in various forms - as a powder, porous granules, blocks, porous 3D scaffolds, and as a coating on metal implants.Hydroxyapatite is recognized as a classic osteoconductive material: it creates a favorable surface for the attachment of osteoblasts and the growth of new bone along the implant-bone interface. Newly formed bone can integrate porous HA by growing into its pores. However, the intrinsic osteoinductivity of hydroxyapatite is weakly expressed. Pure HA does not contain osteogenic factors and usually does not initiate the differentiation of stem cells into osteoblasts without additional stimulating factors. Nevertheless, there is evidence that porous CaP bioceramics, under certain conditions, can induce bone formation in soft tissues (ectopic bone formation) due to the adsorption of endogenous osteoinductive proteins from the blood and a specific pore microarchitecture. In practice, to enhance the osteoinductive properties of HA, it is combined with osteoinductive components or doped with ions (Si4+, Sr2+, etc.), which, as research in the last decade has shown, can stimulate osteogenic differentiation of cells [5,6,13,14].Biocompatibility and biodegradation of HA. Synthetic hydroxyapatite has excellent biocompatibility: it is non-toxic, does not cause a pronounced inflammatory reaction, and is gradually overgrown by bone tissue. Cases of long-term (>10 years) survival of HA implants without signs of rejection have been documented. However, the key characteristic of HA is its biostability. Sintered ceramic implants made from pure hydroxyapatite are practically not resorbed by the body: they can persist at the defect site for many years after implantation. The slow dissolution of HA under physiological conditions prevents its complete replacement with new bone. On one hand, this provides long-term mechanical support for the defect; on the other hand, excessive inertness can hinder bone remodeling. Porous and fine-crystalline HA is resorbed somewhat faster than coarse-crystalline or dense HA due to a larger surface area and osteoclast activity, but overall, the biodegradation rate of hydroxyapatite is noticeably lower than that of other calcium phosphates. Immune cells are capable of gradually resorbing HA, releasing calcium and phosphate ions, which can be reused by the body for bone mineralization. However, this process is slow, so HA is classified as a bioresorbable material with a very low resorption rate. Hydroxyapatite biodegradation is most often harmless to the body: dissolution products - calcium and phosphate - participate in normal mineral metabolism [3,4].Results in vitro and in vivo. In in vitro experiments, hydroxyapatite surfaces demonstrate good adhesion of osteoblast-like cells, promoting their proliferation and maturation. Cells perceive HA as a "native" material - an increase in alkaline phosphatase activity and mineralization is observed in cultures on HA carriers compared to inert materials (e.g., plastic or aluminum oxide). However, excessively rapid saturation of the medium with calcium ions upon contact with HA can temporarily reduce cell viability: for instance, one study showed that in the presence of hydroxyapatite material, the viability of preosteoblasts initially decreased. Nevertheless, overall, HA is not cytotoxic and supports the vital activity of bone tissue cells in vitro. In in vivo experiments (on animals), hydroxyapatite confirms its osteoconductivity: porous HA scaffolds facilitate the ingrowth of blood vessels and bone callus into the defect, leading to the formation of new bone on the surface and inside the implant. When HA granules are implanted into bone defects, their gradual "ingrowth" into the bone along the perimeter is observed. However, the rate of bone formation often lags behind the rate of resorption in quickly resorbing forms. Since hydroxyapatite degrades slowly, a significant amount of unresorbed material may remain in the defect zone after several months or even years. This does not impede functional regeneration, but histologically, the new bone forms mainly at the implant-bone interface, filling the pores to a depth of several millimeters. Some models report the appearance of bone tissue even inside the implant pores far from the original bone; however, such an osteoinductive effect for pure HA is rarely observed and typically occurs under specific conditions. Overall, in vivo studies confirm: HA provides reliable osteointegration and osteoconduction, but without active osteoinductive properties [3,6].Application in clinical practice. Hydroxyapatite is one of the most well-established synthetic materials for bone grafting in clinical practice. Bioceramics based on HA are used in traumatology and orthopedics, spinal surgery (fusion bridges), maxillofacial surgery and dentistry (alveolar ridge augmentation, sinus lifting, filling tooth extraction sockets), otolaryngology (reconstruction of auditory ossicles), and ophthalmology (orbital implants). Granulated osteoplastic materials, porous blocks and shapes, HA-based pastes, and composites are produced. Additionally, hydroxyapatite is widely used for applying biologically active coatings to metal endoprostheses and dental implants: a thin HA layer on the surface of a titanium implant promotes faster bone attachment (enhances implant osseointegration). Clinical experience with HA confirms its safety: the material does not cause immunological reactions or rejection. The main limitations of its use are relatively low strength (HA ceramics are brittle) and extremely slow resorption. Therefore, in load-bearing areas and for large defect volumes, HA is usually used in combination with stronger or more resorbable components (for example, as part of biphasic calcium phosphates or composites with polymers).Mechanism of interaction with bone tissue. The mechanism of biological action of hydroxyapatite is based on its chemical similarity to natural bone mineral. After implantation, a protein layer (albumin, immunoglobulins, osteopontin, etc.) can be adsorbed on the HA surface in contact with physiological fluids, which osteogenic cells subsequently adhere to. The hydroxyapatite surface provides nucleation of new mineral crystals: calcium and phosphate ions from the surrounding environment can precipitate on the HA, leading to the formation of an apatite layer continuous with the mineralizing bone matrix. Thus, the bone "recognizes" the implant as a related structure and continues to build its matrix on it. The absence of toxic degradation products allows cells to proliferate unhindered. At the same time, the material's passivity means it has little effect on biochemical signaling pathways: HA itself does not release osteostimulating factors. Therefore, its contribution is largely passive (serving as a framework and source of mineral ions). Nevertheless, even such a passive mechanism is sufficient for effective osteoconduction. Additionally, slow dissolution of HA can increase the local concentration of Ca2+ and PO43−, creating favorable conditions for the mineralization of the osteoid matrix.Bioactive glass (BG)Bioactive glasses refer to a group of synthetic silicate materials capable of actively interacting with biological tissues, especially bone. Typically, these are amorphous oxide glasses based on SiO2-CaO-P2O5 with additions of sodium oxide and other elements. The first bioactive glass (Bioglass® 45S5: 45% SiO2, 24.5% Na2O, 24.5% CaO, 6% P2O5) was developed by Larry Hench in the late 1960s. The key discovery was the chemical bonding of such glass with living bone - a phenomenon known as bioactivity. Unlike bioinert materials, bioactive glass doesn't simply "grow into" the bone, but induces the formation of a layer on its surface with a composition and structure identical to bone mineral, resulting in a strong fusion between the implant and bone. Subsequently, numerous compositions of bioglasses and glass-ceramics were developed for medical applications.Bioactive glass, like hydroxyapatite, possesses pronounced osteoconductivity - bone easily grows on its surface. However, a number of studies have shown that some bioactive glasses are capable of exhibiting osteoinductive properties as well. For instance, 45S5 granules implanted in rabbit femur defects stimulated faster bone formation compared to synthetic hydroxyapatite granules. In this case, osteogenesis was observed not only at the boundary with the adjacent bone, but also in the porous space inside the glass granules, i.e., far from contact with the original bone. This indicates that bioactive glass can indirectly induce the differentiation of mesenchymal cells into osteoblasts and the formation of bone de novo within the implant - which is defined as osteoinductivity. Reviews note that bioactive glasses are osteoconductive and osteoinductive materials capable of enhancing bone formation. The osteoinductive effect is associated with ion exchange and glass dissolution products, which act as osteostimulating signals. It should be noted that osteoinductivity depends on the glass composition: for example, boron-containing bioglass, which does not contain silicon, also demonstrated the ability to enhance bone formation, indicating the importance of the calcium component. Overall, compared to hydroxyapatite, bioactive glass often stimulates osteogenesis more strongly and can contribute to faster filling of the defect with bone tissue [8].Biocompatibility and biodegradation of BG. Most bioactive glasses have good biocompatibility, although their mechanism of interaction with tissues differs from that of inert materials. After implantation, bioglass reacts with extracellular fluid: due to the exchange of Na+ and Ca2+ ions for H+, a rapid increase in pH occurs in the microenvironment, along with the release of soluble silicon (Si (OH) 4) and calcium and phosphate ions. A local increase in alkalinity and osmotic concentration can have a moderate cytotoxic effect on the very first cells that come into contact with the implant. Nevertheless, this effect is temporary; overall, bioactive glasses are considered non-toxic to tissues, and their degradation products are harmless. Silicate bioglasses gradually dissolve in vivo: the amorphous silica matrix breaks down into orthosilicic acid Si (OH) 4, which is excreted in urine, while calcium and phosphate are utilized by the body. Thus, BG are biodegradable materials. The rate of biodegradation depends on the composition: classic 45S5 glass dissolves 50-60% in the body within a few months, while some phosphate-containing glasses can be almost completely resorbed within a similar timeframe. An important feature of BG is that their dissolution products stimulate regeneration. It has been shown that soluble ions released by the glass (Ca2+, Si4+, PO43−, etc.) not only participate in the formation of the apatite layer but also act as signaling molecules for cells, enhancing osteogenic differentiation. Thus, BG degradation is not just a decrease in material, but an active biochemical process accompanied by regulation of cellular activity. Bioglass does not cause chronic inflammation; on the contrary, some of its compositions (for example, those with boron or silver ions) have antibacterial properties that prevent the development of infection in the implantation zone. The only limitation on BG biocompatibility is the possible effects of rapid ion release - excessively high local concentrations of alkaline and alkaline earth ions can damage cells. However, by selecting the appropriate composition and shape (for example, using porous granules instead of finely dispersed powder), these effects are minimized. In general, bioactive glasses are considered biocompatible and bioactive materials: they interact with tissues, eliciting a favorable response [7,8].In in vitro conditions, bioactive glasses exhibit interesting effects on cell cultures. Extracts obtained by dissolving bioactive glasses stimulate osteoblast proliferation and increase the expression of osteogenic genes (e.g., collagen I, osteocalcin, Runx2) compared to the control. It has been demonstrated that the addition of Si and Ca ions released by the glass accelerates the maturation of osteoblast-like cells. In direct co-culture with glass particles, dosage is important: a moderate amount of bioglass does not have a cytotoxic effect and even improves cell viability, while an excessive amount can temporarily decrease their number due to a sharp change in pH. For example, Lopes et al. showed that adding 7.5% 45S5-bioactive glass to β-TCP improves the survival of osteoblast-like cells, while 40% glass content caused an initial decrease in cell count during the first days of cultivation. Overall, the in vitro data confirm: ionic products of bioactive glasses have a beneficial effect on bone cells, stimulating their migration, proliferation, and differentiation. For instance, a recent study shows that bioactive 45S5 and S53P4 glasses accelerate preosteoblast migration without reducing their viability (in contrast to hydroxyapatite, which caused a short-term decrease in viability) [8].In animal experiments, bioactive glass has proven to be an effective stimulator of osteogenesis. In bone defects, BG granules quickly become coated with a layer of hydroxyapatite (within 1-2 weeks), ensuring the attachment of newly forming bone tissue. In vivo comparisons show that filling defects with bioactive glass can lead to faster bone mass recovery compared to using calcium phosphates alone. For example, in a rabbit osteoplasty model, new bone filled approximately 25% of the defect volume with BG versus about 16% with hydroxyapatite over the same period (approximate figures for illustration). Bioactive glass S53P4 demonstrates particularly remarkable results: when filling large human skull defects, a noticeable volume of new bone formation is observed after just a few months, as evidenced by radiological data. In vivo behavior of BG is also characterized by gradual resorption: after 6-12 months, a significant portion of the implant is replaced by the patient's own bone tissue. It is worth noting the antimicrobial effect of some bioactive glasses in vivo: in areas filled with S53P4, it is possible to contain and eliminate chronic osteomyelitic infections because the dissolution of the glass creates an unfavorable environment for bacteria (alkaline pH, increased osmolarity). This dual effect - simultaneous bone regeneration and infection control - makes bioactive glass a unique material for treating complex cases (for example, infected bone cavities). The disadvantage of bioglass in in vivo studies is its brittleness and relatively low strength: large 3D scaffolds made of glass alone can only bear limited loads and risk fragmentation. Therefore, in load-bearing bones, BG granules are often combined with stronger carriers or polymers.Bioactive glass has found applications in various fields of medicine. One of the first applications was in dentistry: Bioglass 45S5 powder (PerioGlas® preparation) has been used since the 1990s to fill periodontal pockets and bone defects around teeth, stimulating bone septum regeneration. In maxillofacial surgery, bioactive glass was used for the reconstruction of skull and jaw defects. In orthopedics, bioactive glass is used to fill post-resection bone cavities, bone cysts, and in the treatment of chronic osteomyelitis. Clinical trial reports indicate that S53P4 is effective and safe in treating chronic osteomyelitis, ensuring the elimination of infection and stimulating healthy bone growth in the defect cavity. Bioglass S53P4 has been approved for use in EU countries and by the FDA, and is successfully used to fill large defects in long bones after removal of infected necrotic tissues. Additionally, bioactive glass-ceramic materials are used as fillers in combination with other grafts; for example, in spinal surgery, there are bioactive glass composites with β-tricalcium phosphate and polymers for interbody spinal fusion. Overall, bioactive glass has proven itself clinically in the reconstruction of small and medium-sized bone defects, especially where rapid osseointegration is important or where an infectious process is present. The limitation is its use in areas of high load: due to their brittleness, large glass implants can break, so in load-bearing areas, they are used only in a mixture with stronger components or in small fragments. Bioactive glass interacts with the body through a multi-stage mechanism: (1) Immediately after implantation, alkaline and alkaline earth ions (Na+, Ca2+) begin to leach from the glass, replaced by H+ from the tissues, which leads to an increase in pH and the formation of a thin silica layer on the surface; (2) the silica (SiO2) layer reacts with ions from the surrounding fluid - Ca2+ and PO43− are adsorbed and concentrated on it; (3) Within a few hours, an amorphous calcium-phosphate layer forms on the surface, which then (4) crystallizes into a structure similar to hydroxyapatite - thus forming a surface layer of hydroxy-carbonate apatite (HCA), resembling bone mineral. (5) Bone matrix proteins (collagen, osteonectin, etc.) settle on this apatite layer, it is colonized by osteogenic cells, and the formation of new bone tissue, firmly bonded to the implant, begins. Simultaneously, (6) ions released from the dissolving glass affect cells and tissues: an increase in the concentration of Ca2+ and Si (OH) 4 in the extracellular environment stimulates osteoblast proliferation and the differentiation of mesenchymal cells into the osteogenic lineage. The mechanisms of this osteostimulating effect are being studied: it is hypothesized that calcium, phosphate, and silicon ions can act as signaling molecules, activating gene expression associated with osteogenesis. Additionally, the products of glass dissolution affect immune system cells: a moderate increase in pH activates macrophages along the "pro-regenerative" (M2) pathway, promoting healing and regeneration. Thus, bioactive glass acts in two ways - as a structural framework and as a source of chemical stimuli for regeneration. This complex mechanism distinguishes bioactive glass from inert materials. Essentially, bioactive glass in vivo transforms into a hybrid "quartz-apatite" layer, which osteoblasts perceive almost as a natural bone surface. This process underlies its high effectiveness in the repair of bone defects.Calcium phosphatesBesides hydroxyapatite, calcium phosphate-based osteoplastic materials include various solvated and unsolvated phases: β-tricalcium phosphate (β-TCP, Ca3 (PO4) 2), its high-temperature variant α-TCP, as well as HA/TCP mixtures called biphasic calcium phosphates (BCP). Also, calcium phosphate cements that form hydroxyapatite or brushite (CaHPO4·2H2O) in situ can be included here. These materials are similar to hydroxyapatite in that they contain the same Ca2+ and PO43− ions, but differ in their crystal chemistry, solubility, and biological behavior.Osteoconductivity and Osteoinductivity of CaP Materials. All calcium phosphates used in biomedicine are bioactive and osteoconductive: they are capable of directly bonding to bone and serve as a matrix for the growth of new bone tissue. For example, β-tricalcium phosphate in the body gradually becomes covered with an apatite-like layer and facilitates the attachment of osteoblasts and osteoconduction, similar to HA. However, like hydroxyapatite, pure calcium phosphates typically do not exhibit significant osteoinductivity. Without additional stimuli, they do not induce bone tissue formation outside of contact with the original bone. Nevertheless, certain forms can partially induce osteogenesis: for instance, studies have shown that highly porous ceramics made of β-TCP or BCP can initiate the formation of bone islands when implanted into animal muscles - presumably due to optimal microporosity (<10 μm), which allows for the adsorption of endogenous growth factors (BMPs). Overall, the osteoinductive potential of CaP bioceramics is lower than that of an autologous bone graft or demineralized bone matrix. To enhance osteoinduction, additives are often used - for example, β-TCP is combined with collagen, fibrin, or recombinant osteogenic factors (rhBMP-2, etc.) In the last decade, doped calcium phosphates have been developed, which reportedly exhibit more pronounced osteoinductivity, stimulating the differentiation of osteoprogenitor cells. However, basic (non-modified) CaP ceramics are mainly considered as osteoconductive fillers [5,13,14].Biocompatibility and biodegradation of CaP. Phosphate biomaterials exhibit excellent biocompatibility comparable to hydroxyapatite: they are not cytotoxic, do not cause immune rejection, and integrate with bone tissue. Studies have noted the absence of pronounced inflammation around β-TCP and BCP implants; only a mild transient foreign body reaction (giant cells) is observed, which is considered a normal response to a resorbable implant. An important advantage of many calcium phosphates is their resorbability. Unlike persistent hydroxyapatite, β-tricalcium phosphate dissolves relatively quickly and is replaced by new bone. In vivo, β-TCP can be resorbed over a period ranging from several months to 1-2 years, depending on particle size and conditions; during this time, under favorable circumstances, the defect has time to be filled with regenerating bone. According to data, β-TCP has higher biodegradability and resorption rate compared to HA, which causes the β-TCP implant to gradually decrease in volume as it is replaced by bone tissue. However, β-tricalcium phosphate is resorbed more slowly than the less stable α-tricalcium phosphate: the latter phase hydrolyzes so rapidly that it is practically not used for osteoplasty in its pure form. Biphasic calcium phosphates (BCP), representing a mixture of HA and β-TCP, allow for the regulation of resorption rate by combining both components. The higher the proportion of β-TCP in BCP, the faster the material is resorbed; conversely, an increase in the proportion of HA slows down degradation. For example, BCP with a 20% HA/80% β-TCP ratio is resorbed almost as quickly as pure β-TCP, while BCP with a 60% HA/40% β-TCP ratio is resorbed significantly slower, closer to hydroxyapatite. This allows for the selection of material for the required bone regeneration rate. Calcium phosphate biodegradation products - calcium and phosphate ions - are physiological and easily absorbed by the body; they can even stimulate mineralization (by modulating local ion balance and pH). However, excessively rapid resorption of the implant is undesirable: if β-TCP granules dissolve before the bone has filled the space, a cavity may form in the defect. Therefore, a correspondence between the rate of resorption and the rate of osteogenesis is considered optimal. Fortunately, most commercial β-TCP and BCP products have moderate dissolution rates comparable to bone remodeling rates [3,4].In cell cultures, materials based on β-TCP and BCP serve as good substrates for osteogenic cells. For example, the nanoporous structure of β-TCP promotes osteoblast adhesion, proliferation, and mineralization in vitro. Studies show that in the presence of dissolving β-TCP, cells receive additional calcium and phosphate, which stimulates their functional activity. Comparative in vitro viability tests of BCP compositions versus pure HA revealed the advantage of the compositions: adding a soluble component (TCP) to inert HA increases metabolic activity and the number of osteoblast-like cells. However, an excess of soluble phases is also undesirable: as mentioned, too rapid dissolution (e.g., pure α-TCP) can locally disrupt the environment for cells. Nevertheless, most in vitro data indicate that calcium phosphates support the growth and function of osteogenic cells at a level not inferior to hydroxyapatite.In animal experiments, tricalcium phosphate and BCP have also demonstrated their effectiveness as bone substitutes. Porous β-TCP implants in bone defects facilitate the formation of new bone comparable in volume to hydroxyapatite, but with the distinction that the implant gradually disappears. Several months after implantation of β-TCP material, forming bone is observed in the defect, interspersed with remaining material fragments surrounded by osteoclasts (a sign of resorption). By one year, typically, most of the β-TCP is replaced by bone tissue. Studies show that the rate and completeness of regeneration largely depend on pore size: macroporous structures (pores >100 μm) allow vessels and bone trabeculae to penetrate deep into the implant, ensuring uniform growth. For instance, in the work of Daculsi et al. (2010s), biphasic ceramics of 60% HA/40% TCP with interporous channels >100-150 μm demonstrated almost complete filling of pores with newly formed bone by the 12th week, while in fine-pored samples, bone grew only on the surface layers (clearly illustrating the role of porosity). Osteoinduction (bone formation outside of contact with the original bone) for β-TCP was noted by some authors in mouse and rabbit models, but the reproducibility of this effect is low and requires specific conditions (large implant volume, increased osteoclast activity). Overall, in vivo β-TCP and BCP provide reliable osteoconduction comparable to hydroxyapatite and are simultaneously gradually resorbed, making room for newly forming bone. This makes them valuable materials for cases where complete resorption of the implant is desirable after it fulfills its function as a guiding matrix [9,10,11].Clinical ApplicationsTricalcium phosphate (especially β-phase) and biphasic HA/TCP ceramics are widely used as commercial synthetic bone substitutes. For example, β-TCP is a component of ChronOSTM, Cerasorb®, and other materials used for filling bone cavities, treating bone cysts, osteotomy defects, and fractures. BCP is implemented in materials such as MBCP®, Bio-Oss® (partially sintered bone mineral similar to BCP) and many others. Calcium phosphate cements are a separate category: they are pastes that harden in situ with the formation of apatite or brushite. Cements based on calcium phosphates are used to fill defects in orthopedics, neurosurgery, and dentistry. Their advantage is the ability to introduce material in liquid form, filling defects of any shape, and hardening directly in the bone. The disadvantage of cements can be brittleness and relatively low osteoconduction if the cement hardens without pores. Nevertheless, existing cement formulations often become porous during resorption, and bone gradually grows into them. Rapidly soluble brushite cements attract attention as materials that fully resorb within a few months without inflammation, however, their application is limited to non-load-bearing areas due to their rapid decrease in strength after implantation. In clinical practice, the choice between hydroxyapatite, β-TCP, and BCP is based on the required bone replacement rate: for slowly regenerating areas, more stable HA or BCP with high HA content is preferred, and for rapidly healing defects, resorbable β-TCP or cement is preferred [10,11].Mechanisms of interaction with bone tissue. Calcium phosphate materials interact with bone similarly to hydroxyapatite, as they essentially transform into hydroxyapatite in vivo (either directly or through dissolution and re-mineralization). For example, β-TCP, when in contact with physiological solutions, partially hydrolyzes to acidic calcium phosphate and then reorganizes into carbonate hydroxyapatite - that is, the material spontaneously transitions to a more stable apatite form on the surface. The resulting apatite layer is similar to bone mineral and ensures the attachment of bone cells (as described earlier for HA). Simultaneously, the dissolution of β-TCP releases Ca2+ and PO43− ions, which increase the local mineral saturation of the bone defect, facilitating subsequent mineralization of the osteoid matrix. Thus, calcium phosphate resorption stimulates osteogenesis in two ways: (1) through the creation of an apatite osteoconductive layer and (2) through the supply of mineralizing ions for growing bone. Moreover, the interaction between phosphate implants and the body is mediated by cells: osteoclasts resorb the implant, while simultaneously releasing matrix proteins and factors that attract osteoblasts (a typical biological "coupling" of bone resorption and formation). Immune cells (macrophages) also respond to dissolving CaP particles by releasing cytokines that stimulate regeneration. Recent studies note that the immune system plays an important role in the osteoinductivity of CaP bioceramics: a moderate M2-type inflammatory response to material degradation can create a microenvironment favorable for osteogenesis. This aspect formed the basis of the concept of "osteoimmune modulation" of phosphate implants. Nevertheless, the foundation of interaction is still the formation of a biomineralized phase on the implant, which is then layered with bone tissue. For tricalcium phosphate and BCP, the formation of the same hydroxyapatite (HCA) layer has been observed as for bioactive glass, although it occurs somewhat slower and less abundantly. Perhaps it is precisely the relative scarcity of the de novo apatite layer on pure CaP that explains why the binding strength of such implants to bones is somewhat lower than that of bioactive glass: in glass, the entire implant surface transforms into a bone-like mineral, while in HA/TCP ceramics, the material itself is already a mineral, and the additional layer formed is minimal. Consequently, the bone-implant bond in the case of CaP depends more on the mechanical ingrowth of trabeculae into the pores than on chemical adhesion. Nevertheless, the qualitative outcome is the same: bone tissue gradually occupies the volume of the implant, and the implant itself or most of it disappears over time.Comparative characteristics of materialsThe materials described above - hydroxyapatite, bioactive glass, and calcium phosphates (including β-TCP/BCP) - have different property profiles. For clarity, Table 1 provides a comparative summary of their key parameters.
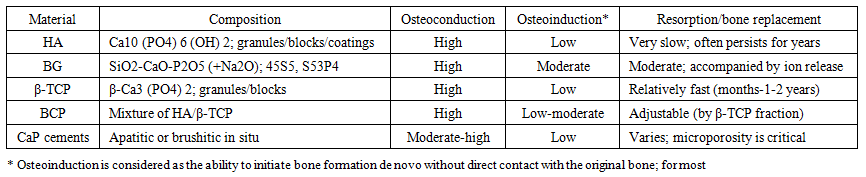 | Table 1. Comparison of materials by biological activity and resorption |
4. Conclusions
- Synthetic osteoplastic materials - hydroxyapatite, bioactive glass, and various calcium phosphates - have become an integral tool in reconstructive bone surgery in recent decades. Each of them has its own strengths and weaknesses. Hydroxyapatite is notable for its exceptional biocompatibility and ability to integrate directly with bone; however, its inertness and lack of resorption can limit the rate of bone remodeling. Beta-tricalcium phosphate and related calcium phosphate materials offer a solution to this problem - they resorb ahead of bone formation, allowing bone to gradually occupy the entire defect area, but with excessively rapid degradation, there may be a risk of incomplete defect filling. Bioactive glass represents a unique class: it is simultaneously biodegradable and bioactive, meaning it not only disappears as healing progresses but also accelerates the healing process itself by releasing osteostimulating ions. Mechanical reliability remains a limiting factor for glasses: due to their brittleness, they are more often used in the form of granules or in combination with other materials.When selecting material for a specific clinical case, it is necessary to consider the nature of the defect, the required recovery rate, the presence of infection, and other conditions. Hydroxyapatite is advisable when long-term volume maintenance is required and there is no need for complete implant resorption (for example, when augmenting bone in dentistry). Bioactive glass is indicated when rapid osseointegration and regeneration stimulation are important (for example, when treating infected defects, where its antibacterial properties and osteostimulation are especially valuable). Biphasic calcium phosphates and β-TCP are well-suited for cases where gradual replacement of the implant with the patient's own bone tissue is desirable - they serve as a temporary matrix, completely giving way to the regenerated bone after a certain time.Scientific research continues to focus on improving these materials. Composites designed to combine the best qualities are being developed - bioglass can compensate for the insufficient osteoinductivity of hydroxyapatite (HA), while HA provides strength and structural support to the glass. Materials with adjustable porosity and dissolution profiles, capable of adapting to the rate of osteogenesis, are being created. Ionic modification (doping) expands biological activity: it has already been shown that replacing a portion of Ca2+ with Sr2+ enhances osteogenic effects, and the introduction of Si4+ into the HA lattice accelerates bone formation. The use of tissue engineering constructs - seeding osteoplastic materials with cells and growth factors - opens the way to obtaining osteoinductive synthetic grafts that are not inferior in effectiveness to autologous bone.In conclusion, the choice between hydroxyapatite, bioactive glass, and calcium phosphates depends on the specific task at hand, and often their combination becomes the optimal solution. Composite materials combining HA and bioactive glass already demonstrate improved osteogenesis results due to the synergy of properties.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML