-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 737-740
doi:10.5923/j.ajmms.20261602.72
Received: Jan. 6, 2026; Accepted: Feb. 5, 2026; Published: Feb. 26, 2026

Combined Pathomorphological Changes of the Menisci and Cruciate Ligaments of the Knee Joint in Post-Traumatic Gonarthrosis
Yodgorov Nodirjon Abdumazidovich1, Mahkamov Nosirjon Jo‘raevich2
1Independent Researcher, Fergana Institute of Public Health, Uzbekistan
2Professor, Andijan State Medical Institute, DSc., Uzbekistan
Correspondence to: Yodgorov Nodirjon Abdumazidovich, Independent Researcher, Fergana Institute of Public Health, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

Post-traumatic gonarthrosis is a progressive degenerative joint disorder in which damage to intra-articular stabilizing structures plays a critical role. This study aimed to investigate the combined pathomorphological changes of the knee joint menisci and cruciate ligaments in post-traumatic gonarthrosis. Histological examination revealed pronounced degenerative and dystrophic alterations in meniscal tissue, including disorganization and fragmentation of collagen fibers, decreased cellularity, mucoid degeneration, and focal necrotic areas. In advanced cases, fibrocartilaginous matrix disruption and cystic degeneration were observed. Pathomorphological analysis of the cruciate ligaments demonstrated collagen fiber loosening, hyalinization, fibroblast proliferation, and areas of fibrotic remodeling. Vascular alterations, such as neovascularization and vessel wall sclerosis, were frequently detected, accompanied by mild to moderate chronic inflammatory cell infiltration. The simultaneous involvement of menisci and cruciate ligaments resulted in structural instability of the knee joint and contributed to the progression of degenerative changes. The identified pathomorphological features reflect the key mechanisms underlying post-traumatic gonarthrosis development and emphasize the importance of combined damage to meniscal and ligamentous structures in disease progression.
Keywords: Post-traumatic gonarthrosis, Knee joint, Menisci, Cruciate ligaments, Pathomorphology, Degenerative changes
Cite this paper: Yodgorov Nodirjon Abdumazidovich, Mahkamov Nosirjon Jo‘raevich, Combined Pathomorphological Changes of the Menisci and Cruciate Ligaments of the Knee Joint in Post-Traumatic Gonarthrosis, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 737-740. doi: 10.5923/j.ajmms.20261602.72.
1. Introduction
- Post-traumatic gonarthrosis (PTOA) is a significant clinical and research concern as knee injuries such as anterior cruciate ligament (ACL) ruptures and meniscal tears are major risk factors for the development of osteoarthritis, often leading to progressive joint degeneration and functional impairment [1]. Traumatic meniscus damage disrupts the knee’s load distribution and shock-absorbing capacity, which accelerates cartilage breakdown and osteoarthritic changes over time [2]. Structural degeneration following meniscal trauma includes disorganization of collagen and extracellular matrix remodeling, which are characteristic morphological features preceding knee osteoarthritis [3]. Animal models show that combined ACL rupture and meniscal tears worsen meniscal, cartilage, and subchondral bone morphology, indicating a strong link between ligamentous and meniscal pathology in PTOA progression [4]. Systematic review evidence suggests that even after ACL reconstruction, structural changes in tibiofemoral joint components persist, contributing to long-term osteoarthritic risk [5]. Furthermore, clinical outcomes indicate that meniscal pathology in conjunction with ACL injury significantly alters joint stability and predisposes patients to degenerative changes [6]. Biomarker studies highlight that meniscal injury induces inflammatory responses associated with cartilage breakdown and impaired repair mechanisms in the knee joint, further underpinning the pathological relevance of meniscal degeneration in PTOA [7]. Epidemiological analyses demonstrate that the global research output on PTOA has increased markedly, reflecting growing recognition of its clinical burden and complex pathogenesis [8]. Additional studies confirm that meniscus injury leads to dynamic biomechanical perturbations of the knee joint, exacerbating joint instability and promoting degenerative cascades [9]. Collectively, these findings emphasize the pathological significance of combined meniscal and ligamentous degeneration in post-traumatic gonarthrosis and underline the importance of detailed pathomorphological investigation to improve diagnosis, prevention, and therapeutic strategies [10,13].Purpose of the StudyTo investigate the combined pathomorphological changes of the meniscus and cruciate ligaments in post-traumatic gonarthrosis, and to assess their impact on knee joint structure and function.
2. Materials and Methods
- For this study, tissue samples of the meniscus and cruciate ligaments of the knee joint were collected from patients with post-traumatic gonarthrosis who were treated at the Traumatology Hospitals of the Fergana Region between 2020 and 2024. A total of 50 samples were included in the analysis.For histopathological examination, the collected tissues were fixed in 10% formalin and embedded in paraffin. Sections were cut at a thickness of 5–7 μm using a microtome and stained with hematoxylin and eosin (H&E).During the analysis, structural changes in the meniscus and anterior cruciate ligament were assessed, including collagen composition, cell number and distribution, mucoid degeneration, necrosis, and morphological alterations of the ligaments. Each sample was evaluated using descriptive and semi-quantitative methods.All study procedures were approved by the Ethics Committee of the Traumatology Hospitals of the Fergana Region, and tissue samples were collected with the informed consent of the patients.
3. Research Results
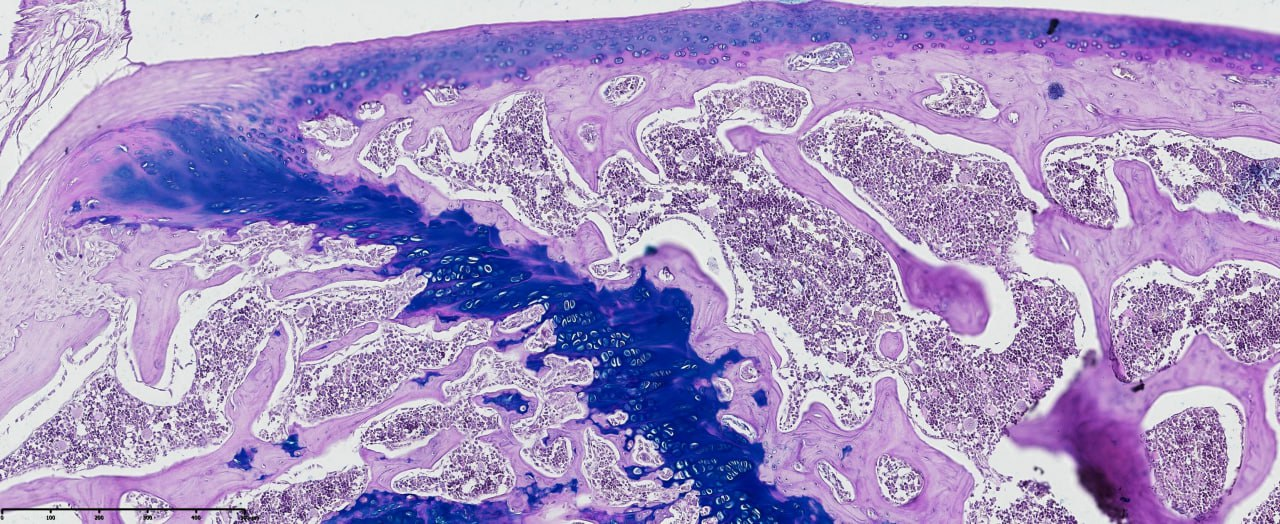
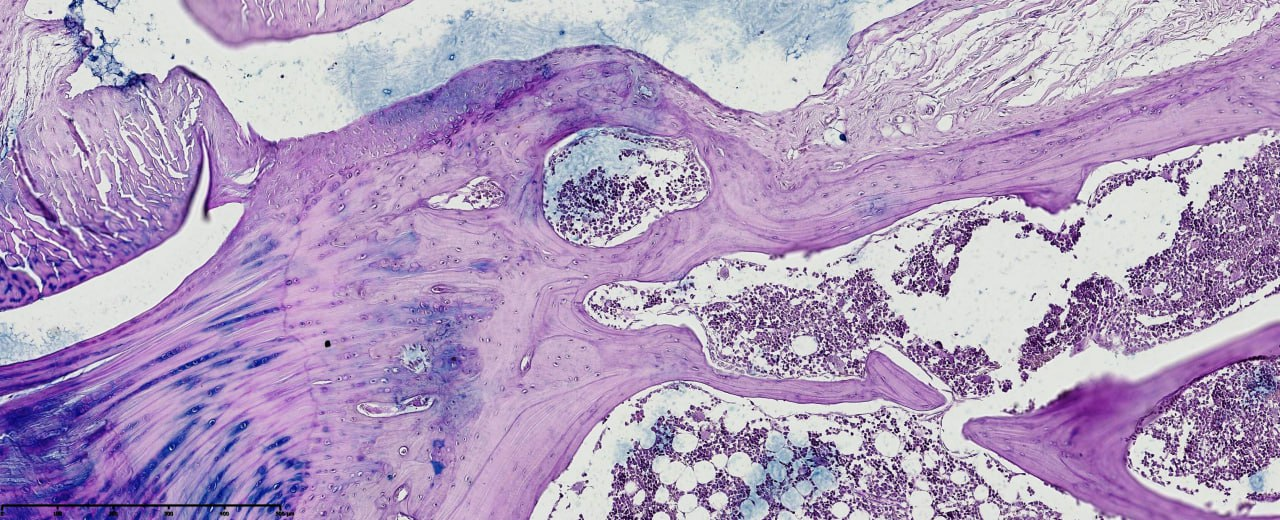
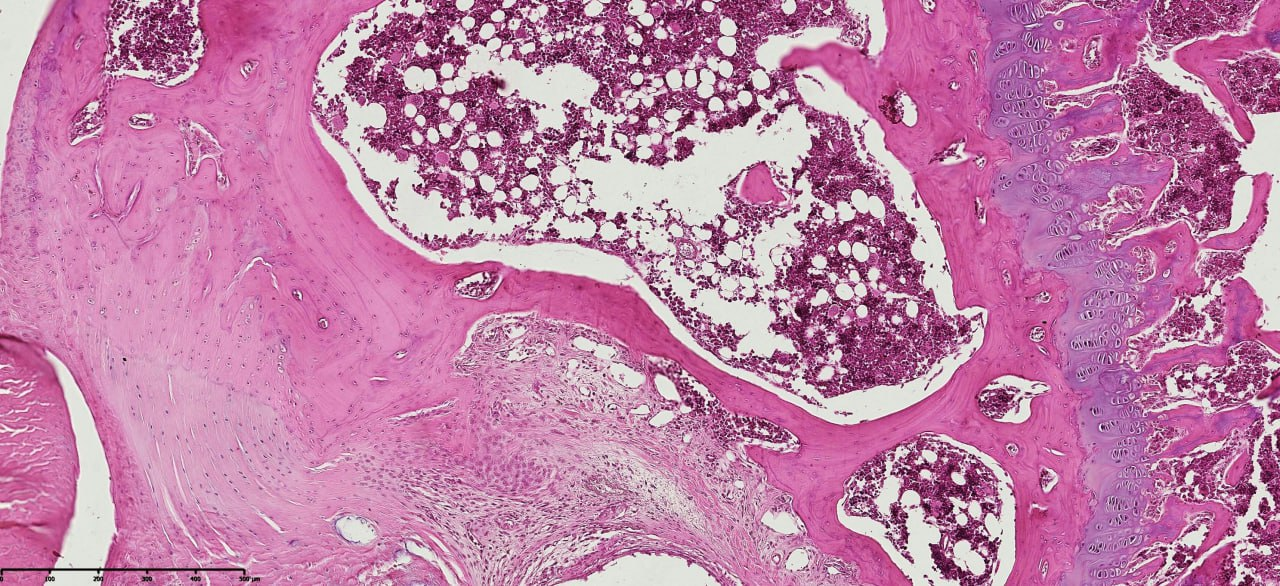
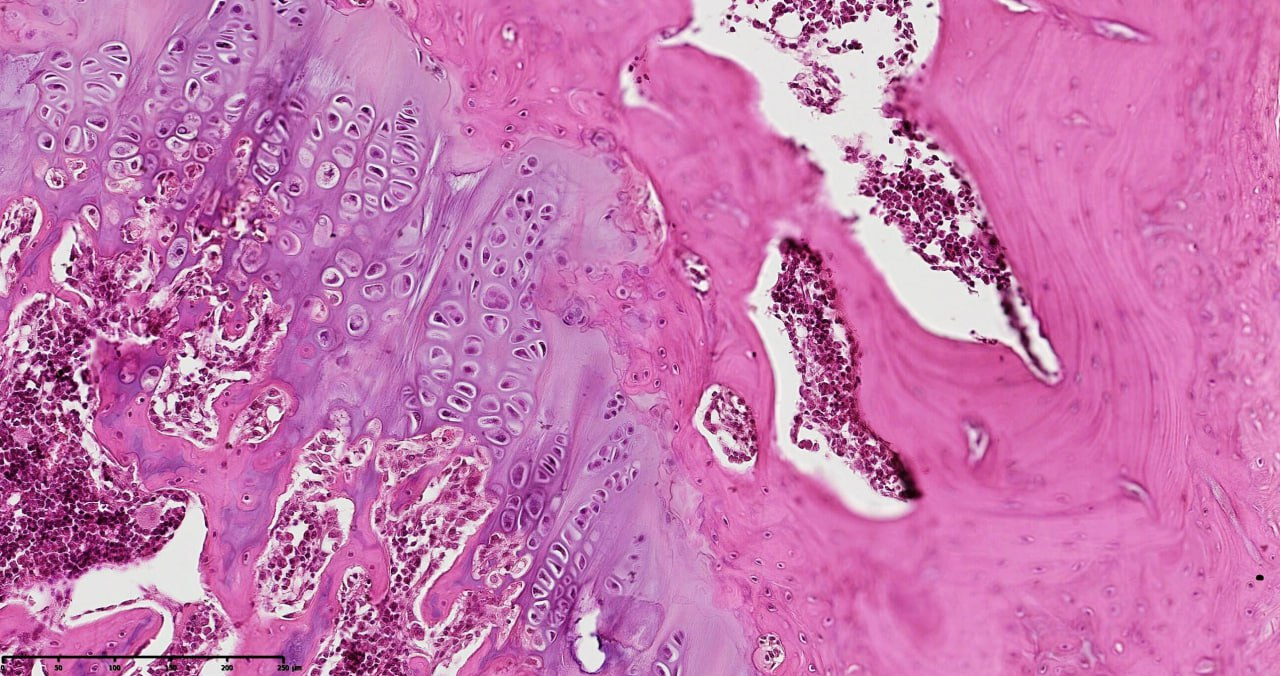
- Histopathological evaluation of meniscus samples from patients with post-traumatic gonarthrosis demonstrated prominent and region-specific degenerative alterations consistent with advanced meniscal pathology. On gross examination, the menisci showed surface irregularities, fragmentation along the inner border, and contour deformities, especially in the posterior horn, which is exposed to higher mechanical forces.Microscopically, there was substantial disruption of the collagen fiber network and extracellular matrix (ECM) structure. The collagen fibers exhibited disordered alignment and fragmentation, with circumferential and radial fibers irregularly oriented, impairing normal load transmission and shock absorption. Proteoglycan accumulation was detected in degenerated areas, indicating active matrix remodeling and changes in tissue biomechanics (see figure 1).
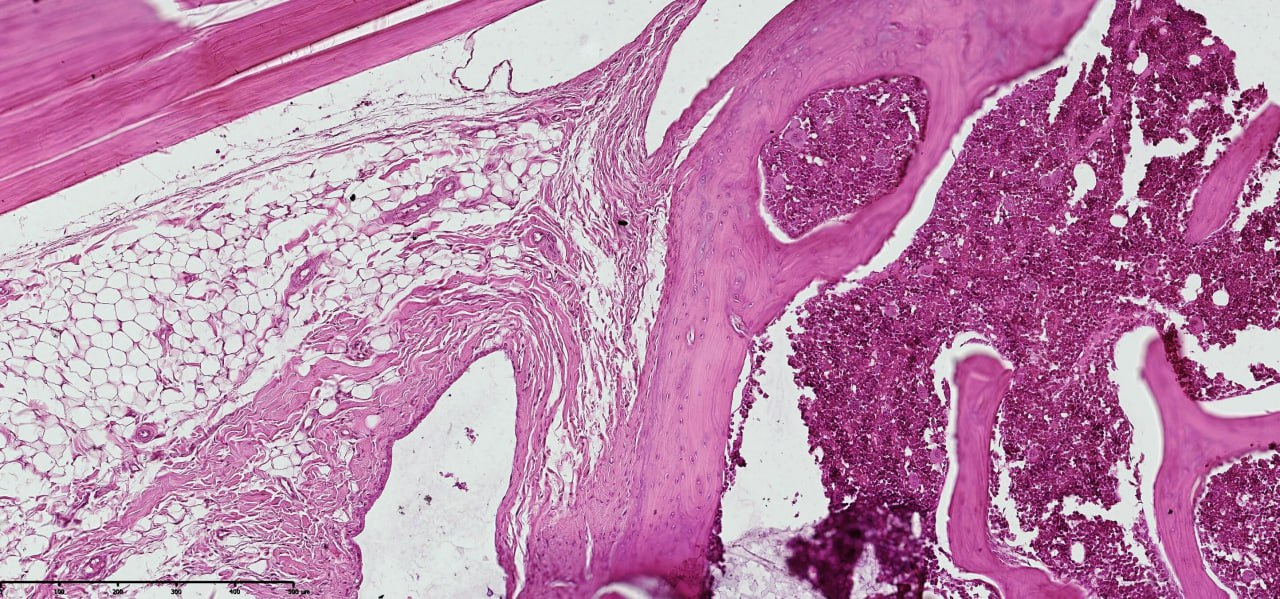 | Figure 3. Cruciate ligament section in post-traumatic gonarthrosis showing disrupted collagen fibers, ECM disorganization, and proteoglycan accumulation. Staining: Van Gieson. Magnification: 20x10 |
4. Conclusions
- The present study demonstrated extensive and region-specific pathomorphological alterations in the menisci and cruciate ligaments of the knee joint in patients with post-traumatic gonarthrosis. In the menisci, disorganization of collagen fibers and extracellular matrix (ECM), reduced cellularity, mucoid degeneration, and focal necrotic areas were observed, alongside fibrocartilaginous matrix disruption and cystic degeneration. In the cruciate ligaments, collagen fiber loosening, hyalinization, fibroblast proliferation, and fibrotic remodeling were identified, accompanied by peripheral vascular proliferation and neovascularization.These structural and cellular changes, particularly in association with ACL and meniscal pathology, adversely affected joint stability and contributed to the progressive development of post-traumatic gonarthrosis. Collagen and ECM disorganization, together with altered cellular phenotypes in the meniscus and ACL, compromised ligament and meniscal biomechanical function and joint stability. Furthermore, these morphological alterations possess clinical significance, as they reflect limited reparative potential and accelerate osteoarthritic progression.Overall, the pathomorphological analysis of menisci and cruciate ligaments in post-traumatic gonarthrosis provides critical insights into tissue function, reparative capacity, and has important implications for diagnostic, prognostic, and therapeutic strategies.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML