-
Paper Information
- Next Paper
- Previous Paper
- Paper Submission
-
Journal Information
- About This Journal
- Editorial Board
- Current Issue
- Archive
- Author Guidelines
- Contact Us
American Journal of Medicine and Medical Sciences
p-ISSN: 2165-901X e-ISSN: 2165-9036
2026; 16(2): 719-727
doi:10.5923/j.ajmms.20261602.69
Received: Jan. 2, 2026; Accepted: Jan. 22, 2026; Published: Feb. 26, 2026

Erosive and Ulcerative Gastroduodenal Bleeding in Patients with Comorbid Cardiovascular Pathology
Rasul I. Rakhimov1, Sherzod Sh. Zhanibekov2, Bobir I. Shukurov2, 3, Karimali S. Elmurodov3
1City Clinical Hospital No. 7, Tashkent, Uzbekistan
2Tashkent State Medical University, Tashkent, Uzbekistan
3Republican Research Center of Emergency Medicine, Tashkent, Uzbekistan
Correspondence to: Bobir I. Shukurov, Tashkent State Medical University, Tashkent, Uzbekistan.
| Email: |  |
Copyright © 2026 The Author(s). Published by Scientific & Academic Publishing.
This work is licensed under the Creative Commons Attribution International License (CC BY).
http://creativecommons.org/licenses/by/4.0/

This review is devoted to the problem of erosive and ulcerative gastroduodenal bleeding (EUGDB) in patients with comorbid cardiovascular pathology. EUGDB remains one of the most significant causes of emergency hospitalization, accounting for up to 90% of all gastrointestinal bleeding cases and being characterized by high recurrence rates and mortality. Particular attention is paid to the relationship between cardiovascular diseases, polypharmacy, and age-related changes with the risk of ulcerative lesions and their complications. Data are presented on the influence of antiplatelet agents, anticoagulants, nonsteroidal anti-inflammatory drugs, and Helicobacter pylori infection on the pathogenesis of EUGDB. Current evidence is summarized regarding the development of bleeding in patients with ischemic heart disease (IHD), chronic heart failure (CHF), and acute ischemic stroke (AIS), as well as their impact on prognosis and treatment outcomes. The importance of a multidisciplinary approach to diagnosis and therapy is emphasized, including prophylactic use of proton pump inhibitors, H. pylori eradication, and individualized selection of anticoagulant therapy. Special attention is given to new anticoagulants, particularly factor XI inhibitors, which demonstrate a promising efficacy and safety profile.
Keywords: Erosive and ulcerative gastroduodenal bleeding, Cardiovascular diseases, Comorbidity, Anticoagulant therapy, Helicobacter pylori, Ischemic heart disease, Acute ischemic stroke
Cite this paper: Rasul I. Rakhimov, Sherzod Sh. Zhanibekov, Bobir I. Shukurov, Karimali S. Elmurodov, Erosive and Ulcerative Gastroduodenal Bleeding in Patients with Comorbid Cardiovascular Pathology, American Journal of Medicine and Medical Sciences, Vol. 16 No. 2, 2026, pp. 719-727. doi: 10.5923/j.ajmms.20261602.69.
Article Outline
1. Introduction
- Erosive and ulcerative gastroduodenal bleeding (EUGDB) remains one of the most serious complications of many gastrointestinal disorders, accounting for 80–90% of all gastrointestinal bleeding (GIB) cases [1]. These bleedings are characterized by sudden onset, rapid progression, and high mortality rates [2]. In the United States, more than 320,000 patients are hospitalized annually due to EUGDB, with mortality rates ranging from 2% to 15%, and the economic burden associated with this type of bleeding exceeding 2.5 billion USD [3,4,5]. In Europe, between 48 and 147 individuals per 100,000 population are admitted to hospitals with gastroduodenal hemorrhage. Mortality directly attributed to bleeding or decompensation of comorbid conditions may reach 10% [6]. Despite substantial advances in surgery and anesthesiology, mortality from acute gastrointestinal bleeding remains at 5–14%, rising to 30–40% in cases of rebleeding, with no clear trend toward improvement [7]. Among patients with EUGDB, death directly related to blood loss occurs in approximately 18%, whereas the remaining fatal outcomes are associated with complications of underlying comorbidities [8].In recent years, the rising incidence of erosive and ulcerative gastroduodenal bleeding (EUGDB) in patients with cardiovascular diseases and other somatic disorders has acquired an epidemic character. According to various authors, the frequency of acute erosive and ulcerative lesions of the gastric and duodenal mucosa (GDM) in patients with therapeutic pathology reaches 80–90%, while EUGDB attributable to these lesions accounts for 45–55% of cases [9]. For example, analysis of 6,900 autopsies of patients who died from myocardial infarction and stroke in multidisciplinary hospitals demonstrated a nearly one-third increase in the detection rate of erosive and ulcerative GDM lesions over the past decade. Among patients with cardiovascular diseases, acute ulcers and erosions constitute 60–70% of all GDM mucosal lesions, whereas chronic peptic ulcers are diagnosed in 30–40% of cases. Acute ulcers predominate in individuals with acute cerebrovascular accidents and myocardial infarction, which is associated with stress-induced hypersecretory responses and impaired mucosal microcirculation, whereas chronic ulcers are more commonly observed in long-standing ischemic heart disease and prolonged use of antiplatelet agents [10].EUGDB of varying severity developed in 67% of deceased patients with ischemic heart disease (IHD) and in 55% of those with stroke; however, their contribution to mechanisms of fatal outcome differed. The incidence of fatal bleeding (EUGDB as the direct cause of death) was 3% in myocardial infarction, 2% in chronic forms of IHD accompanied by chronic heart failure (CHF), and 1% in stroke. At the same time, the frequency of acute erosive and ulcerative lesions of the GDM in deceased patients with myocardial infarction reaches 18%, in CHF – 10%, in stroke – 14%, and may rise to 21% when IHD is combined with cerebrovascular disease [11].The challenge of timely diagnosis and treatment of erosive and ulcerative GDM lesions – and consequently, prevention of EUGDB – is exacerbated by the fact that in 30–90% of patients with chronic somatic pathology, gastropathies are asymptomatic; in 46–58% of cases, typical clinical manifestations are absent; and in 25–42% of patients, symptoms of the underlying disease predominate. As a result, mucosal lesions of the GDM often remain undiagnosed until the development of life-threatening bleeding. In this patient population, EUGDB is the first clinical manifestation of gastroduodenal mucosal injury in 25–55% of cases, while in most instances this severe complication is identified for the first time at autopsy [8].On the other hand, several risk factors are becoming increasingly prevalent. Population aging has led to a higher incidence of cardiovascular diseases and other comorbid conditions, thereby increasing mortality risk in patients with EUGDB. In addition, the growing use of low-dose aspirin, NSAIDs, antiplatelet agents, and anticoagulants further contributes to this risk [12,13]. In this context, it is important to clarify that within the framework of this review, the term “cardiovascular diseases” encompasses both chronic forms of ischemic heart disease (stable angina, chronic heart failure) and acute vascular events – acute myocardial infarction and AIS. These conditions share common pathogenetic mechanisms, the use of antiplatelet and anticoagulant therapy, as well as similar risk factors for the development of erosive and ulcerative gastroduodenal lesions and bleeding.
2. The Relationship Between Comorbid Cardiovascular Pathology and Erosive–Ulcerative Gastroduodenal Bleeding
- The term “comorbidity” was first introduced in 1970 by the American epidemiologist A.R. Feinstein, who defined it as “any distinct additional clinical entity that has existed, exists, or may occur during the clinical course of a patient’s index disease” [14,15]. Comorbidity refers to the presence of two or more chronic diseases in a single patient, which may be etiopathogenetically interconnected or may simply coexist temporally, regardless of the level of activity of each condition. This concept implies that comorbid states arise due to shared or unified pathogenetic mechanisms.A comprehensive understanding of the relationship between comorbid pathology and EUGDB is a crucial component in improving outcomes of this severe urgent condition. It is well established that various chronic diseases significantly increase the risk of gastrointestinal bleeding, particularly EUGDB. For example, conditions such as liver cirrhosis and peptic ulcer disease show a strong association with the risk of gastrointestinal hemorrhage [16]. In addition, the elderly population faces additional risks due to factors such as polypharmacy and the long-term management of chronic diseases, especially when over-the-counter medications such as NSAIDs are used, which can exacerbate gastrointestinal complications [17].NSAID-associated ulcer bleeding accounts for more than 20,000 deaths annually in the United Kingdom and over 16,500 deaths in the United States [23]. Nevertheless, many patients take antiplatelet agents for years without pronounced adverse effects. Large cohort studies – Biobank (n=213,598) and ESTHER (n=7,737) – have confirmed that low-dose aspirin is an independent risk factor for peptic ulcer disease. Use of aspirin for less than one year was associated with a higher risk of ulcer formation compared with long-term use (more than one year); however, even during prolonged administration, aspirin maintains a statistically significant association with peptic ulcers [19].The combination of aspirin with proton pump inhibitors (PPIs) significantly reduces the risk of peptic ulcer disease and its complications. Moreover, this approach improves treatment tolerability and enhances patient adherence. In clinical practice, when IHD coexists with peptic ulcer disease, clopidogrel should be preferred as an antiplatelet agent due to its lower rate of gastrotoxic complications compared with aspirin. When dual antiplatelet therapy (aspirin + clopidogrel) is required, concomitant use of PPIs (omeprazole, esomeprazole) is mandatory. The use of ticlopidine and ticagrelor requires caution in patients with active ulcer disease, particularly in the presence of H. pylori infection, which should be eradicated prior to initiating therapy. The ESC (2020) and ACC/AHA (2019) guidelines emphasize that the choice of antiplatelet agent should take into account the risk of gastrointestinal bleeding, and in patients at high risk, clopidogrel monotherapy with PPI protection is preferred [18].Antiplatelet agents are not the only cause of ulcer formation and gastrointestinal bleeding. In a study involving 1,000 patients with stable IHD, erosive and ulcerative lesions of the gastric mucosa were observed even during PPI therapy [19]. Possible contributing factors include reduced mucosal blood supply due to atherosclerosis, adverse effects of PPIs, and H. pylori infection. According to REGATA-1 data, H. pylori infection was identified in 87.5% of patients with severe gastrointestinal bleeding [20].The impact of H. pylori on the pathogenesis of IHD remains a subject of ongoing investigation. Patients with H. pylori infection were more frequently diagnosed with coronary stenosis (7.6% vs. 2.9%, p=0.01) [18]. Proposed mechanisms of this association include systemic inflammation, vascular endothelial injury, oxidative stress, dyslipidemia, and enhanced platelet aggregation [21,22,24]. H. pylori eradication therapy has been shown to reduce the frequency of angina episodes and improve exercise tolerance [18,22].Patients with IHD who are receiving antiplatelet therapy should undergo regular screening for H. pylori infection followed by eradication, if detected. Long-term PPI use is recommended to reduce the likelihood of ulcer formation and bleeding [19,20].Analysis of the pathogenetic role of comorbid conditions allows for more accurate identification of high-risk groups among patients with EUGDB. This, in turn, enables the implementation of preventive strategies such as the use of proton pump inhibitors, correction of metabolic disturbances, and rational prescribing of medications, taking into account their adverse effects on the gastrointestinal mucosa (Table 1).
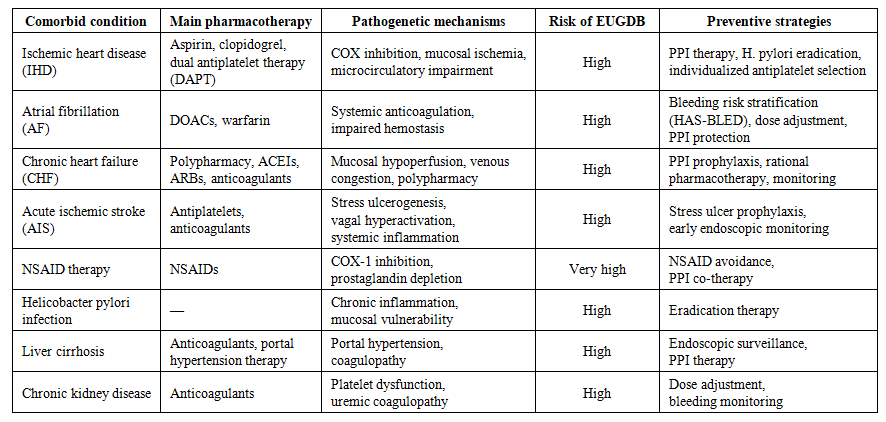 | Table 1. Comorbid cardiovascular and systemic conditions, pharmacological therapy, and risk of erosive and ulcerative gastroduodenal bleeding (EUGDB) |
3. Conclusions
- Gastrointestinal bleeding represents a serious clinical challenge, particularly when it is associated with comorbid conditions that complicate both diagnosis and treatment. Diseases such as cardiovascular disorders, hepatic dysfunction, and coagulopathies frequently coexist with gastrointestinal pathology, creating a complex interplay that can hinder effective management. The presence of these comorbid conditions may not only complicate the diagnosis of gastrointestinal bleeding but also increase the risk of severe complications, ultimately affecting patient outcomes.Comorbidities such as diabetes mellitus, arterial hypertension, chronic kidney disease, and metabolic disturbances significantly modify the pathogenesis of ulcerative lesions. They increase the vulnerability of the gastric and duodenal mucosa to injurious factors and impair regenerative and protective processes, thereby contributing to the development of complications. The presence of multiple comorbid conditions, as well as their pharmacological treatment (for example, the use of anticoagulants, antiplatelet agents, or nonsteroidal anti-inflammatory drugs), markedly increases the risk not only of primary bleeding but also of recurrent hemorrhage.This problem is further compounded by age-related physiological changes, as most patients with erosive and ulcerative gastroduodenal bleeding (EUGDB) are older than 60 years. These patients exhibit reduced mucosal resistance to acid and pepsin, decreased gastric motility, and other changes that heighten susceptibility to injury. It is also essential to recognize that comorbid conditions influence the clinical presentation of EUGDB, complicating diagnosis and necessitating a more detailed interdisciplinary approach to management.Identifying and addressing these interconnected health issues is crucial for clinicians when formulating an effective treatment strategy. Given the close association between EUGDB and comorbid diseases, examining the role of comorbidity becomes particularly important for developing effective approaches to prevention, diagnosis, and treatment.
 Abstract
Abstract Reference
Reference Full-Text PDF
Full-Text PDF Full-text HTML
Full-text HTML